Abstract
Background Cardiac tissue engineering might be useful in treatment of diseased myocardium or cardiac malformations. The creation of functional, biocompatible contractile tissues, however, remains challenging. We hypothesized that coupling of arginine–glycine–aspartic acid–serine (RGD+) adhesion peptides would improve cardiomyocyte viability and differentiation and contractile performance of collagen-cell scaffolds.
Methods Clinically approved collagen scaffolds were functionalized with RGD+ cells and seeded with cardiomyocytes. Contractile performance, cardiomyocyte viability and differentiation were analyzed at days 1 and 8 and/or after culture for 1 month.
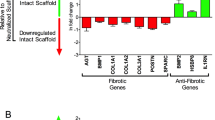
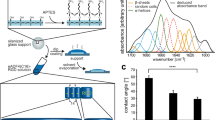
Results The method used for the RGD+ cell–collagen scaffold coupling enabled the following features: high coupling yields and complete washout of excess reagent and by-products with no need for chromatography; spectroscopic quantification of RGD+ coupling; a spacer arm of 36Å, a length reported as optimal for RGD+-peptide presentation and favorable for integrin-receptor clustering and subsequent activation. Isotonic and isometric mechanical parameters, either spontaneous or electrostimulated, exhibited good performance in RGD+ constructs. Cell number and viability was increased in RGD+ scaffolds, and we saw good organization of cell contractile apparatus with occurrence of cross-striation.
Conclusions We report a novel method of engineering a highly effective collagen-cell scaffold based on RGD+ peptides cross-linked to a clinically approved collagen matrix. The main advantages were cell contractile performance, cardiomyocyte viability and differentiation.
Key Points
-
We propose a new method for tissue engineering to create an efficient and safe contractile tissue by improving the interaction of contractile cells and their surrounding three-dimensional collagen environment
-
The approach increases the functionality of collagen templates by covalent binding and optimal presentation of molecules, such as hormones, growth factors, cytokines and adhesive ligand molecules (i.e. arginine–glycine–aspartic acid peptides)
-
We applied several tools for assessment of myocyte contractility in vitro, in three-dimensional cellular architectures under isometric and isotonic conditions
-
This scaffold might also serve as a platform for improving cellular transplantation in the heart or for engineering a true myocardium equivalent
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Zimmermann WH and Eschenhagen T (2003) Cardiac tissue engineering for replacement therapy. Heart Fail Rev 8: 259–269
Zimmermann WH et al. (2006) Engineered heart tissue grafts improve systolic and diastolic function in infarcted rat hearts. Nat Med 12: 452–458 3
Akhyari P et al. (2002) Mechanical stretch regimen enhances the formation of bioengineered autologous cardiac muscle grafts. Circulation 106: I137–I142
Fink C et al. (2000) Chronic stretch of engineered heart tissue induces hypertrophy and functional improvement. Faseb J 14: 669–6795
Radisic M et al. (2004) Functional assembly of engineered myocardium by electrical stimulation of cardiac myocytes cultured on scaffolds. Proc Natl Acad Sci U S A 101: 18129–18134
Radisic M et al. (2004) Medium perfusion enables engineering of compact and contractile cardiac tissue. Am J Physiol Heart Circ Physiol 286: H507–H516
Papadaki M et al. (2001) Tissue engineering of functional cardiac muscle: molecular, structural, and electrophysiological studies. Am J Physiol Heart Circ Physiol 280: H168–H178
Giraud MN et al. (2007) Current state of the art in myocardial tissue engineering. Tissue Eng 13: 1825–1836
Caplice NM (2006) The future of cell therapy for acute myocardial infarction. Nat Clin Pract Cardiovasc Med 3 (Suppl 1): S129–S132
Yacoub M et al. (2006) The future of regenerative therapy in patients with chronic heart failure. Nat Clin Pract Cardiovasc Med 3 (Suppl 1): S133–S135
Torella D et al. (2006) Resident human cardiac stem cells: role in cardiac cellular homeostasis and potential for myocardial regeneration. Nat Clin Pract Cardiovasc Med 3 (Suppl 1): S8–S13
Adler ED and Maddox TM (2007) Cell therapy for cardiac disease: where do we go from here? Nat Clin Pract Cardiovasc Med 4: 2–3
Aldhous P (2007) Stem-cell research: after the gold rush. Nature 434: 694–696
Nadal-Ginard B and Fuster V (2007) Myocardial cell therapy at the crossroads. Nat Clin Pract Cardiovasc Med 4: 1
Akhyari P et al. (2008) Myocardial tissue engineering: the extracellular matrix. Eur J Cardiothorac Surg 34: 229–241
Masuda S et al. (2008) Cell sheet engineering for heart tissue repair. Adv Drug Deliv Rev 60: 277–285
Radisic M et al. (2008) Cardiac tissue engineering using perfusion bioreactor systems. Nat Protoc 3: 719–738
Christman KL and Lee RJ (2006) Biomaterials for the treatment of myocardial infarction. J Am Coll Cardiol 48: 907–913
Fuster V and Sanz J (2007) Gene therapy and stem cell therapy for cardiovascular diseases today: a model for translational research. Nat Clin Pract Cardiovasc Med 4 (Suppl 1): S1–S8
Pucéat M and Ballis A (2007) Embryonic stem cells: from bench to bedside. Clin Pharmacol Ther 82: 337–339
Samarel AM (2005) Costameres, focal adhesions, and cardiomyocyte mechanotransduction. Am J Physiol Heart Circ Physiol 289: H2291–H2301
Griffith LG and Swartz MA (2006) Capturing complex 3D tissue physiology in vitro. Nat Rev Mol Cell Biol 7: 211–224
Parker KK et al. (2008) Myofibrillar architecture in engineered cardiac myocytes. Circ Res 103: 340–342
Ross RS and Borg TK (2001) Integrins and the myocardium. Circ Res 88: 1112–1119
Srivastava D and Yu S (2006) Stretching to meet needs: integrin-linked kinase and the cardiac pump. Genes Dev 20: 2327–2331
Kuhn B et al. (2007) Periostin induces proliferation of differentiated cardiomyocytes and promotes cardiac repair. Nat Med 13: 962–969
Brancaccio M et al. (2006) Integrin signalling: the tug-of-war in heart hypertrophy. Cardiovasc Res 70: 422–433
Darribère T et al. (2000) Integrins: regulators of embryogenesis. Biol Cell 92: 5–25
Shimazaki M et al. (2008) Periostin is essential for cardiac healing after acute myocardial infarction. J Exp Med 205: 295–303
Knowlton KU et al. (1991) Co-regulation of the atrial natriuretic factor and cardiac myosin light chain-2 genes during alpha-adrenergic stimulation of neonatal rat ventricular cells. Identification of cis sequences within an embryonic and a constitutive contractile protein gene which mediate inducible expression. J Biol Chem 266: 7759–7768
Dar A et al. (2002) Optimization of cardiac cell seeding and distribution in 3D porous alginate scaffolds. Biotechnol Bioeng 80: 305–312
Zimmermann WH et al. (2002) Cardiac grafting of engineered heart tissue in syngenic rats. Circulation 106: I151–I157
Mandarim-De-Lacerda CA and Meirelles Pereira LM (2000) Numerical density of cardiomyocytes in chronic nitric oxide synthesis inhibition. Pathobiology 68: 36–42
Borenfreund E and Puerner JA (1985) Toxicity determined in vitro by morphological alterations and neutral red absorption. Toxicol Lett 24: 119–124
Guellich A et al. (2007) Role of oxidative stress in cardiac dysfunction of PPARα−/− mice. Am J Physiol Heart Circ Physiol 293: H93–H102
Lecarpentier Y and Coirault C (1998) Mechanics, energetics, and crossbridge kinetics of rabbit diaphragm during congestive heart failure. Faseb J 12: 981–989
Vogel V and Sheetz M (2006) Local force and geometry sensing regulate cell functions. Nat Rev Mol Cell Biol 7: 265–275
Wood MA et al. (2008) The β integrins and cytoskeletal nanoimprinting. Exp Cell Res 314: 927–935
Myles JL et al. (2000) Modification of the adhesive properties of collagen by covalent grafting with RGD peptides. J Biomater Sci Polym Ed 11: 69–86
Beer JH et al. (1992) Immobilized Arg-Gly-Asp (RGD) peptides of varying lengths as structural probes of the platelet glycoprotein IIb/IIIa receptor. Blood 79: 117–128
Craig WS et al. (1995) Concept and progress in the development of RGD-containing peptide pharmaceuticals. Biopolymers 37: 157–175
Kofidis T et al. (2003) Clinically established hemostatic scaffold (tissue fleece) as biomatrix in tissue- and organ-engineering research. Tissue Eng 9: 517–523
Lecarpentier Y (1985) Real-time kinetics of sarcomere relaxation by laser diffraction. Circ Res 56: 331–339
Valencik ML et al. (2006) Integrin activation in the heart: a link between electrical and contractile dysfunction? Circ Res 99: 1403–1410
Chachques JC et al. (2006) Myocardial Assistance by Grafting a New Bioartificial Upgraded Myocardium (MAGNUM Trial): clinical feasability study. Circulation 114 (Suppl II): II-566
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Schussler, O., Coirault, C., Louis-Tisserand, M. et al. Use of arginine–glycine–aspartic acid adhesion peptides coupled with a new collagen scaffold to engineer a myocardium-like tissue graft. Nat Rev Cardiol 6, 240–249 (2009). https://doi.org/10.1038/ncpcardio1451
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpcardio1451
This article is cited by
-
SPECT and PET to optimize cardiac stem cell therapy
Journal of Nuclear Cardiology (2012)
-
Collagen scaffolds with or without the addition of RGD peptides support cardiomyogenesis after aggregation of mouse embryonic stem cells
In Vitro Cellular & Developmental Biology - Animal (2011)