Abstract
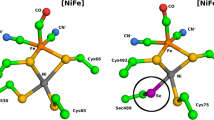
We studied the mechanism of aerobic inactivation of Desulfovibrio fructosovorans nickel-iron (NiFe) hydrogenase by quantitatively examining the results of electrochemistry, EPR and FTIR experiments. They suggest that, contrary to the commonly accepted mechanism, the attacking O2 is not incorporated as an active site ligand but, rather, acts as an electron acceptor. Our findings offer new ways toward the understanding of O2 inactivation and O2 tolerance in NiFe hydrogenases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Fernandez, V.M., Hatchikian, E.C., Patil, D.S. & Cammack, R. Biochim. Biophys. Acta 883, 145–154 (1986).
Volbeda, A. et al. Nature 373, 580–587 (1995).
Higuchi, Y., Yagi, T. & Yasuoka, N. Structure 5, 1671–1680 (1997).
Ogata, H. et al. Structure 13, 1635–1642 (2005).
Pandelia, M.E., Ogata, H. & Lubitz, W. ChemPhysChem 11, 1127–1140 (2010).
Lamle, S.E., Albracht, S.P. & Armstrong, F.A. J. Am. Chem. Soc. 126, 14899–14909 (2004).
Fritsch, J. et al. Nature 479, 249–252 (2011).
Pandelia, M.E. et al. Proc. Natl. Acad. Sci. USA 108, 6097–6102 (2011).
Volbeda, A. et al. Proc. Natl. Acad. Sci. USA 109, 5305–5310 (2012).
Shomura, Y., Yoon, K.S., Nishihara, H. & Higuchi, Y. Nature 479, 253–256 (2011).
Millo, D. et al. J. Phys. Chem. B 113, 15344–15351 (2009).
De Lacey, A.L., Fernandez, V.M., Rousset, M. & Cammack, R. Chem. Rev. 107, 4304–4330 (2007).
van der Zwaan, J.W., Albracht, S.P., Fontijn, R.D. & Slater, E.C. FEBS Lett. 179, 271–277 (1985).
Liebgott, P.P. et al. J. Am. Chem. Soc. 133, 986–997 (2011).
Bleijlevens, B. et al. J. Biol. Inorg. Chem. 9, 743–752 (2004).
Liebgott, P.P. et al. Nat. Chem. Biol. 6, 63–70 (2010).
van der Zwaan, J.W., Coremans, J.M., Bouwens, E.C. & Albracht, S.P. Biochim. Biophys. Acta 1041, 101–110 (1990).
Volbeda, A. et al. J. Biol. Inorg. Chem. 10, 239–249 (2005).
Ogata, H., Kellers, P. & Lubitz, W. J. Mol. Biol. 402, 428–444 (2010).
Carepo, M. et al. J. Am. Chem. Soc. 124, 281–286 (2002).
Van Gastel, M. Appl. Magn. Reson. 37, 207–218 (2010).
Kumar, M., Colpas, G.J., Day, R.O. & Maroney, M.J. J. Am. Chem. Soc. 111, 8323–8325 (1989).
Chohan, B.S., Shoner, S.C., Kovacs, J.A. & Maroney, M.J. Inorg. Chem. 43, 7726–7734 (2004).
Reddie, K.G. & Carroll, K.S. Curr. Opin. Chem. Biol. 12, 746–754 (2008).
Pandelia, M.E. et al. J. Am. Chem. Soc. 132, 6991–7004 (2010).
Leroux, F. et al. Experimental approaches to kinetics of gas diffusion in hydrogenase. Proc. Natl. Acad. Sci. USA 105, 11188–11193 (2008).
Liebgott, P.P. et al. Relating diffusion along the substrate tunnel and oxygen sensitivity in hydrogenase. Nat. Chem. Biol. 6, 63–70 (2010).
Léger, C., Dementin, S., Bertrand, P., Rousset, M. & Guigliarelli, B. Inhibition and aerobic inactivation kinetics of Desulfovibrio fructosovorans NiFe hydrogenase studied by protein film voltammetry. J. Am. Chem. Soc. 126, 12162–12172 (2004).
Liebgott, P.P. et al. Original design of an oxygen-tolerant [NiFe] hydrogenase: major effect of a valine-to-cysteine mutation near the active site. J. Am. Chem. Soc. 133, 986–997 (2011).
Rudiger, O., Abad, J.M., Hatchikian, E.C., Fernandez, V.M. & De Lacey, A.L. Oriented immobilization of Desulfovibrio gigas hydrogenase onto carbon electrodes by covalent bonds for nonmediated oxidation of H2 . J. Am. Chem. Soc. 127, 16008–16009 (2005).
Volbeda, A. et al. Structure of the [NiFe] hydrogenase active site: evidence for biologically uncommon Fe ligands. J. Am. Chem. Soc. 118, 12989–12996 (1996).
Acknowledgements
We thank J. Hadj-Saïd for performing preliminary electrochemical experiments and P. Bertrand, V. Fourmond and V. Fernández for helpful discussions. This work was funded by the CNRS, the Agence Nationale de la Recherche, the Aix-Marseille Université, the City of Marseilles and the Spanish Ministerio de Ciencia e Innovación (MICINN) (project CTQ2009-12649) and was supported by the Pôle de Compétitivité Capénergies. A.A.H. thanks the CNRS and the Région Provence-Alpes Côte d'Azur for financial support. O.G.-S. thanks the MICINN for a Formación de Personal Investigador (FPI) grant.
Author information
Authors and Affiliations
Contributions
A.A.H. carried out the enzyme purification, electrochemical and EPR experiments, and analyzed the data with the support of S.D., M.R., C.B., C.L., B.B. and B.G. O.G.-S. performed FTIR experiments and analyzed the data with the support of A.L.D.L. P.-P.L. performed preliminary electrochemical experiments and analyzed the data. The research was designed by A.A.H., B.B., M.R., B.G., C.L. and S.D. S.D. and C.L. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Results (PDF 874 kb)
Rights and permissions
About this article
Cite this article
Abou Hamdan, A., Burlat, B., Gutiérrez-Sanz, O. et al. O2-independent formation of the inactive states of NiFe hydrogenase. Nat Chem Biol 9, 15–17 (2013). https://doi.org/10.1038/nchembio.1110
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1110
This article is cited by
-
Biomimetic asymmetric catalysis
Science China Chemistry (2023)
-
Reversible catalysis
Nature Reviews Chemistry (2021)
-
Theoretical insights into [NiFe]-hydrogenases oxidation resulting in a slowly reactivating inactive state
JBIC Journal of Biological Inorganic Chemistry (2017)
-
Crystallographic studies of [NiFe]-hydrogenase mutants: towards consensus structures for the elusive unready oxidized states
JBIC Journal of Biological Inorganic Chemistry (2015)
-
A redox hydrogel protects hydrogenase from high-potential deactivation and oxygen damage
Nature Chemistry (2014)