Abstract
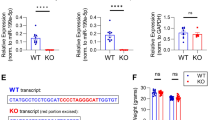
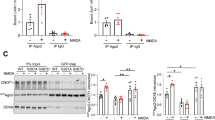
The microRNA pathway has been implicated in the regulation of synaptic protein synthesis and ultimately in dendritic spine morphogenesis, a phenomenon associated with long-lasting forms of memory. However, the particular microRNAs (miRNAs) involved are largely unknown. Here we identify specific miRNAs that function at synapses to control dendritic spine structure by performing a functional screen. One of the identified miRNAs, miR-138, is highly enriched in the brain, localized within dendrites and negatively regulates the size of dendritic spines in rat hippocampal neurons. miR-138 controls the expression of acyl protein thioesterase 1 (APT1), an enzyme regulating the palmitoylation status of proteins that are known to function at the synapse, including the α13 subunits of G proteins (Gα13). RNA-interference-mediated knockdown of APT1 and the expression of membrane-localized Gα13 both suppress spine enlargement caused by inhibition of miR-138, suggesting that APT1-regulated depalmitoylation of Gα13 might be an important downstream event of miR-138 function. Our results uncover a previously unknown miRNA-dependent mechanism in neurons and demonstrate a previously unrecognized complexity of miRNA-dependent control of dendritic spine morphogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others

References
Hering, H. & Sheng, M. Dendritic spines: structure, dynamics and regulation. Nature Rev. Neurosci. 2, 880–888 (2001).
Nimchinsky, E. A., Sabatini, B. L. & Svoboda, K. Structure and function of dendritic spines. Annu. Rev. Physiol. 64, 313–353 (2002).
Matsuzaki, M., Honkura, N., Ellis-Davies, G. C. & Kasai, H. Structural basis of long-term potentiation in single dendritic spines. Nature 429, 761–766 (2004).
Nagerl, U. V, Eberhorn, N., Cambridge, S. B. & Bonhoeffer, T. Bidirectional activity-dependent morphological plasticity in hippocampal neurons. Neuron 44, 759–767 (2004).
Bagni, C. & Greenough, W. T. From mRNP trafficking to spine dysmorphogenesis: the roots of fragile X syndrome. Nature Rev. Neurosci. 6, 376–387 (2005).
Okamoto, K., Nagai, T., Miyawaki, A. & Hayashi, Y. Rapid and persistent modulation of actin dynamics regulates postsynaptic reorganization underlying bidirectional plasticity. Nature Neurosci. 7, 1104–1112 (2004).
Yi, J. J. & Ehlers, M. D. Ubiquitin and protein turnover in synapse function. Neuron 47, 629–632 (2005).
Shi, S. H et al. Rapid spine delivery and redistribution of AMPA receptors after synaptic NMDA receptor activation. Science 284, 1811–1816 (1999).
Zhou, Z. et al. Brain-specific phosphorylation of MeCP2 regulates activity-dependent Bdnf transcription, dendritic growth, and spine maturation. Neuron 52, 255–269 (2006).
Bingol, B. & Schuman, E. M. Synaptic protein degradation by the ubiquitin proteasome system. Curr. Opin. Neurobiol. 15, 536–541 (2005).
Steward, O. & Schuman, E. M. Protein synthesis at synaptic sites on dendrites. Annu. Rev. Neurosci. 24, 299–325 (2001).
Martin, K. C., Barad, M. & Kandel, E. R. Local protein synthesis and its role in synapse-specific plasticity. Curr. Opin. Neurobiol. 10, 587–592 (2000).
Eberwine, J., Miyashiro, K., Kacharmina, J. E. & Job, C. Local translation of classes of mRNAs that are targeted to neuronal dendrites. Proc. Natl Acad. Sci. USA 98, 7080–7085 (2001).
Ostroff, L. E., Fiala, J. C., Allwardt, B. & Harris, K. M. Polyribosomes redistribute from dendritic shafts into spines with enlarged synapses during LTP in developing rat hippocampal slices. Neuron 35, 535–545 (2002).
Wells, D. G., Richter, J. D. & Fallon, J. R. Molecular mechanisms for activity-regulated protein synthesis in the synapto-dendritic compartment. Curr. Opin. Neurobiol. 10, 132–137 (2000).
Kiebler, M. A. & Bassell, G. J. Neuronal RNA granules: movers and makers. Neuron 51, 685–690 (2006).
Krichevsky, A. M. & Kosik, K. S. Neuronal RNA granules: a link between RNA localization and stimulation-dependent translation. Neuron 32, 683–696 (2001).
Ashraf, S. I., McLoon, A. L., Sclarsic, S. M. & Kunes, S. Synaptic protein synthesis associated with memory is regulated by the RISC pathway in Drosophila. Cell 124, 191–205 (2006).
Schratt, G. M. et al. A brain-specific microRNA regulates dendritic spine development. Nature 439, 283–289 (2006).
Ambros, V. The functions of animal microRNAs. Nature 431, 350–355 (2004).
Bartel, D. P. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116, 281–297 (2004).
Filipowicz, W., Bhattacharyya, S. N. & Sonenberg, N. Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nature Rev. Genet. 9, 102–114 (2008).
Kosik, K. S. The neuronal microRNA system. Nature Rev. Neurosci. 7, 911–920 (2006).
Miska, E. A. et al. Microarray analysis of microRNA expression in the developing mammalian brain. Genome Biol. 5, R68 (2004).
Sempere, L. F. et al. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 5, R13 (2004).
Kim, J. et al. Identification of many microRNAs that copurify with polyribosomes in mammalian neurons. Proc. Natl Acad. Sci. USA 101, 360–365 (2004).
Yeh, D. C., Duncan, J. A., Yamashita, S. & Michel, T. Depalmitoylation of endothelial nitric-oxide synthase by acyl-protein thioesterase 1 is potentiated by Ca2+-calmodulin. J. Biol. Chem. 274, 33148–33154 (1999).
Rao, A. & Steward, O. Evidence that protein constituents of postsynaptic membrane specializations are locally synthesized: analysis of proteins synthesized within synaptosomes. J. Neurosci. 11, 2881–2895 (1991).
Meister, G., Landthaler, M., Dorsett, Y. & Tuschl, T. Sequence-specific inhibition of microRNA- and siRNA-induced RNA silencing. RNA 10, 544–550 (2004).
Zito, K., Knott, G., Shepherd, G. M., Shenolikar, S. & Svoboda, K. Induction of spine growth and synapse formation by regulation of the spine actin cytoskeleton. Neuron 44, 321–334 (2004).
Mansfield, J. H. et al. MicroRNA-responsive 'sensor' transgenes uncover Hox-like and other developmentally regulated patterns of vertebrate microRNA expression. Nature Genet. 36, 1079–1083 (2004).
Vo, N. et al. A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proc. Natl Acad. Sci. USA 102, 16426–16431 (2005).
Rehmsmeier, M., Steffen, P., Hochsmann, M. & Giegerich, R. Fast and effective prediction of microRNA/target duplexes. RNA 10, 1507–1517 (2004).
Kruger, J. & Rehmsmeier, M. RNAhybrid: microRNA target prediction easy, fast and flexible. Nucleic Acids Res. 34, W451–W454 (2006).
Rodenas-Ruano, A., Perez-Pinzon, M. A., Green, E. J., Henkemeyer, M. & Liebl, D. J. Distinct roles for ephrinB3 in the formation and function of hippocampal synapses. Dev. Biol. 292, 34–45 (2006).
Etournay, R. et al. PHR1, an integral membrane protein of the inner ear sensory cells, directly interacts with myosin 1c and myosin VIIa. J. Cell Sci. 118, 2891–2899 (2005).
Ozaki, N. et al. cAMP-GEFII is a direct target of cAMP in regulated exocytosis. Nature Cell Biol. 2, 805–811 (2000).
Husseini Ael, D. et al. Synaptic strength regulated by palmitate cycling on PSD-95. Cell 108, 849–863 (2002).
Linder, M. E. & Deschenes, R. J. Palmitoylation: policing protein stability and traffic. Nature Rev. Mol. Cell Biol. 8, 74–84 (2007).
Kang, R. et al. Neural palmitoyl-proteomics reveals dynamic synaptic palmitoylation. Nature 456, 904–909 (2008).
Bhattacharyya, R. & Wedegaertner, P. B. Gα13 requires palmitoylation for plasma membrane localization, Rho-dependent signaling, and promotion of p115-RhoGEF membrane binding. J. Biol. Chem. 275, 14992–14999 (2000).
Kye, M. J. et al. Somatodendritic microRNAs identified by laser capture and multiplex RT-PCR. RNA 13, 1224–1234 (2007).
Lugli, G., Torvik, V. I., Larson, J. & Smalheiser, N. R. Expression of microRNAs and their precursors in synaptic fractions of adult mouse forebrain. J. Neurochem. 106, 650–661 (2008).
Bernard, O. Lim kinases, regulators of actin dynamics. Int. J. Biochem. Cell Biol. 39, 1071–1076 (2007).
Kurose, H. Gα12 and Gα13 as key regulatory mediator in signal transduction. Life Sci. 74, 155–161 (2003).
Tada, T. & Sheng, M. Molecular mechanisms of dendritic spine morphogenesis. Curr. Opin. Neurobiol. 16, 95–101 (2006).
Obernosterer, G., Leuschner, P. J., Alenius, M. & Martinez, J. Post-transcriptional regulation of microRNA expression. RNA 12, 1161–1167 (2006).
Schratt, G. M., Nigh, E. A., Chen, W. G., Hu, L. & Greenberg, M. E. BDNF regulates the translation of a select group of mRNAs by a mammalian target of rapamycin-phosphatidylinositol 3-kinase-dependent pathway during neuronal development. J. Neurosci. 24, 9366–9377 (2004).
Paradis, S. et al. An RNAi-based approach identifies molecules required for glutamatergic and GABAergic synapse development. Neuron 53, 217–232 (2007).
Obernosterer, G., Martinez, J. & Alenius, M. Locked nucleic acid-based in situ detection of microRNAs in mouse tissue sections. Nature Protocols 2, 1508–1514 (2007).
Acknowledgements
We thank O. Rocks and P. Wedegaertner for providing reagents; R. Heinen for suggestions on the luciferase reporter assays; T. Wüst for technical assistance; and M. Alenius and A. Keene for critically reading the manuscript. This work was supported by the Deutsche Forschungsgemeinschaft (SFB488; to G.M.S.), the Human Frontier Science Program (Career Development Award; to G.M.S.), the National Institute on Drug Abuse (1R21DA025102-01; to G.M.S.), the National Institutes of Health (NS045500; to M.E.G.) the Bioinformatics Initiative (to C.D. and M.R.), the Austrian Academy of Sciences (to J.M. and S.B. (DOC-fFORTE-fellowship)) and the Austrian Government, GEN-AU initiative (to G.O.). C.H. acknowledges the Alexander von Humboldt foundation for a postdoctoral fellowship. F.D. is a recipient of a TALENT-stipendium from the Netherlands Organization for Scientific Research (NWO) and a Marie-Curie fellowship from the European Union.
Author information
Authors and Affiliations
Contributions
G.M.S. performed microRNA profiling, M.C. analysed the microarray data, G.O., R.F. and C.J.B. performed in situ experiments, G.S., S.B., G.M.S. and C.K. performed and analysed spine measurements, M.O. and A.D. performed and analysed electrophysiology experiments, G.S. and G.M.S. performed and analysed immunostaining, G.S., G.O. and P.J.L. designed and performed the target validation experiments, G.O., C.D. and M.R. carried out bioinformatics, G.S. performed biochemistry, F.D., C.H., B.R. and H.W. were involved in the design and synthesis of specific APT1 inhibitors, R.F. performed sensor assays, G.S, S.B., M.C. and S.Kh. performed RNA expression analysis, K.H. and S.Ka provided reagents, M.E.G. participated in the initial stages of project planning, J.M. and G.M.S. directed the research and G.M.S. coordinated the project and wrote the manuscript.
Corresponding authors
Supplementary information
Supplementary Information
Supplementary Information (PDF 1708 kb)
Supplementary Information
Supplementary Table 1 (XLS 1262 kb)
Rights and permissions
About this article
Cite this article
Siegel, G., Obernosterer, G., Fiore, R. et al. A functional screen implicates microRNA-138-dependent regulation of the depalmitoylation enzyme APT1 in dendritic spine morphogenesis. Nat Cell Biol 11, 705–716 (2009). https://doi.org/10.1038/ncb1876
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1876
This article is cited by
-
An essential role for the RNA helicase DDX6 in NMDA receptor-dependent gene silencing and dendritic spine shrinkage
Scientific Reports (2024)
-
miR-483-5p offsets functional and behavioural effects of stress in male mice through synapse-targeted repression of Pgap2 in the basolateral amygdala
Nature Communications (2023)
-
MicroRNAs and Synaptic Plasticity: From Their Molecular Roles to Response to Therapy
Molecular Neurobiology (2022)
-
Roles of palmitoylation in structural long-term synaptic plasticity
Molecular Brain (2021)
-
MicroRNA-138 suppresses glioblastoma proliferation through downregulation of CD44
Scientific Reports (2021)