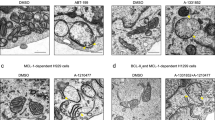
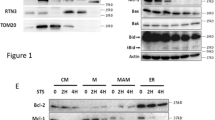
Abstract
Mitochondrial proteins function as essential regulators in apoptosis. Here, we show that mitochondrial adenylate kinase 2 (AK2) mediates mitochondrial apoptosis through the formation of an AK2–FADD–caspase-10 (AFAC10) complex. Downregulation of AK2 attenuates etoposide- or staurosporine-induced apoptosis in human cells, but not that induced by tumour-necrosis-factor-related apoptosis-inducing ligand (TRAIL) or Fas ligand (FasL). During intrinsic apoptosis, AK2 translocates to the cytoplasm, whereas this event is diminished in Apaf-1 knockdown cells and prevented by Bcl-2 or Bcl-XL. Addition of purified AK2 protein to cell extracts first induces activation of caspase-10 via FADD and subsequently caspase-3 activation, but does not affect caspase-8. AFAC10 complexes are detected in cells undergoing intrinsic cell death and AK2 promotes the association of caspase-10 with FADD. In contrast, AFAC10 complexes are not detected in several etoposide-resistant human tumour cell lines. Taken together, these results suggest that, acting in concert with FADD and caspase-10, AK2 mediates a novel intrinsic apoptotic pathway that may be involved in tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chinnaiyan, A. M., O'Rourke, K., Tewari, M. & Dixit, V. M. FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis. Cell 81, 505–512 (1995).
Kischkel, F. C., Hellbardt, S., Behrmann, I., Germer, M., Pawlita, M., Krammer, P. H. & Peter, M. E. Cytotoxicity-dependent APO-1 (Fas/CD95)-associated proteins form a death-inducing signaling complex (DISC) with the receptor. EMBO J. 14, 5579–5588 (1995).
Zhang, J. & Winoto, A. A mouse Fas-associated protein with homology to the human Mort1/FADD protein is essential for Fas-induced apoptosis. Mol. Cell. Biol. 16, 2756–2763 (1996).
Micheau, O., Solary, E., Hammann, A. & Dimanche-Boitrel, M. T. Fas ligand-independent, FADD-mediated activation of the Fas death pathway by anticancer drugs. J. Biol. Chem. 274, 7987–7992 (1999).
Shimada, K., Matsuyoshi, S., Nakamura, M., Ishida, E., Kishi, M. & Konishi, N. Phosphorylation of Fas-associated death domain contributes to enhancement of etoposide-induced apoptosis in prostate cancer cells. Jpn. J. Cancer Res. 93, 1164–1174 (2002).
Kawahara, A., Ohsawa, Y., Matsumura, H., Uchiyama, Y. & Nagata, S. Caspase-independent cell killing by Fas-associated protein with death domain. J. Cell Biol. 143, 1353–1360 (1998).
Matsumura, H., Shimizu, Y., Ohsawa, Y., Kawahara, A., Uchiyama, Y. & Nagata, S. Necrotic death pathway in Fas receptor signaling. J. Cell Biol. 151, 1247–1256 (2000).
Scaffidi, C., Volkland, J., Blomberg, I., Hoffmann, I., Krammer, P. H. & Peter, M. E. Phosphorylation of FADD/ MORT1 at serine 194 and association with a 70-kDa cell cycle-regulated protein kinase. J. Immunol. 164, 1236–1242 (2000).
Hua, Z. C., Sohn, S. J., Kang, C., Cado, D. & Winoto, A. A function of Fas-associated death domain protein in cell cycle progression localized to a single amino acid at its C-terminal region. Immunity 18, 513–521 (2003).
Alappat, E. C. et al. Phosphorylation of FADD at serine 194 by CKIα regulates its nonapoptotic activities. Mol. Cell 19, 321–332 (2005).
Zhang, J., Cado, D., Chen, A., Kabra, N. H. & Winoto, A. Fas-mediated apoptosis and activation-induced T-cell proliferation are defective in mice lacking FADD/Mort1. Nature 392, 296–300 (1998).
Yeh, W. C. et al. FADD: essential for embryo development and signaling from some, but not all, inducers of apoptosis. Science 279, 1954–1958 (1998).
Wang, J. et al. Inherited human Caspase 10 mutations underlie defective lymphocyte and dendritic cell apoptosis in autoimmune lymphoproliferative syndrome type II. Cell 98, 47–58 (1999).
Teitz, T. et al. Caspase 8 is deleted or silenced preferentially in childhood neuroblastomas with amplification of MYCN. Nature Med. 6, 529–535 (2000).
Shin, M. S. et al. Inactivating mutations of CASP10 gene in non-Hodgkin lymphomas. Blood 99, 4094–4099 (2002).
Single, B., Leist, M. & Nicotera, P. Simultaneous release of adenylate kinase and cytochrome c in cell death. Cell Death Differ. 5, 1001–1003 (1998).
Samali, A., Cai, J., Zhivotovsky, B., Jones, D. P. & Orrenius, S. Presence of a pre-apoptotic complex of pro-caspase-3, Hsp60 and Hsp10 in the mitochondrial fraction of jurkat cells. EMBO J. 18, 2040–2048 (1999).
Kluck, R. M. et al. The pro-apoptotic proteins, Bid and Bax, cause a limited permeabilization of the mitochondrial outer membrane that is enhanced by cytosol. J. Cell Biol. 147, 809–822 (1999).
Wang, J., Chun, H. J., Wong, W., Spencer, D. M. & Lenardo, M. J. Caspase-10 is an initiator caspase in death receptor signaling. Proc. Natl Acad. Sci. USA 98, 13884–13888 (2001).
Schlauderer, G. J. & Schulz, G. E. The structure of bovine mitochondrial adenylate kinase: comparison with isoenzymes in other compartments. Protein Sci. 5, 434–441 (1996).
Noma, T., Song, S., Yoon, Y. S., Tanaka, S. & Nakazawa, A. cDNA cloning and tissue-specific expression of the gene encoding human adenylate kinase isozyme 2. Biochim. Biophys. Acta. 1395, 34–39 (1998).
Chandra, D., Choy, G., Deng, X., Bhatia, B., Daniel, P. & Tang, D. G. Association of active caspase 8 with the mitochondrial membrane during apoptosis: potential roles in cleaving BAP31 and caspase 3 and mediating mitochondrion–endoplasmic reticulum cross talk in etoposide-induced cell death. Mol. Cell. Biol. 24, 6592–6607 (2004).
Micheau, O., Solary, E., Hammann, A. & Dimanche-Boitrel, M. T. Fas ligand-independent, FADD-mediated activation of the Fas death pathway by anticancer drugs. J. Biol. Chem. 274, 7987–7992 (1999).
Kim, P. K. et al. Hepatocyte Fas-associating death domain protein/mediator of receptor-induced toxicity (FADD/MORT1) levels increase in response to pro-apoptotic stimuli. J. Biol. Chem. 277, 38855–38862 (2002).
Park, S. J., Wu, C. H., Gordon, J. D., Zhong, X., Emami, A. & Safa, A. R. Taxol induces caspase-10-dependent apoptosis. J. Biol. Chem. 279, 51057–51067 (2004).
Filomenko, R. et al. Caspase-10 involvement in cytotoxic drug-induced apoptosis of tumor cells. Oncogene 25, 7635–7645 (2006).
Fujita, E., Egashira, J., Urase, K., Kuida, K. & Momoi, T. Caspase-9 processing by caspase-3 via a feedback amplification loop in vivo. Cell Death Differ. 8, 335–344 (2001).
Zou, H. et al. Regulation of the Apaf-1/Caspase-9 apoptosome by Caspase-3 and XIAP. J. Biol. Chem. 278, 8091–8098 (2003).
Lassus, P., Opitz-Arraya, X. & Lazebnik, Y. Requirement for caspase-2 in stress-induced apoptosis before mitochondrial permeabilization. Science 297, 1352–1354 (2002).
Slee, E. A. et al. Ordering the cytochrome c-initiated caspase cascade: hierarchical activation of caspases-2, -3, -6, -7, -8 and -10 in a caspase-9-dependent manner. J. Cell Biol. 144, 281–292 (1999).
Acknowledgements
The authors thank: J. Blenis (Harvard University, Boston, MA) for providing Jurkat cells (A3, I2-1, I9-2); J. Yuan (Harvard University) for HeLa/Bcl-2, HeLa/Bcl-XL and HeLa/CrmA cells; and H. D. Ryoo (New York University, New York, NY) for critical reading of this manuscript. J.-O. P. and S.-h. H. were in part supported by the Korean Research Foundation (KRF-2005-201-C00039) and by the BK21 programme. This work was supported by grants from the Functional Human Genome Project (FG06-2-7) and the Brain Research Center of the 21st Century Frontier Research Programme and the Ubiquitome project funded by the Korea Science and Engineering Foundation (KOSEF) of the Korea government (MOST).
Author information
Authors and Affiliations
Contributions
H.J.L., J.O.P. and H.J.K. performed flow cytometry analysis, IP assays and cell line characterization. H.J.L., Y.O., S.H.H., H.K., D.H.C., H.N.W. and J.H.N. performed siRNA generation and fractionation and RT–PCR analysis. H.J.L. and Y.J.J. performed in vitro pull-down assays. S.S. performed functional assays of AK2 and FADD in the mouse system. Hyo Joon Kim and K.S.K. performed antibody generation and mutagenesis. Y.K.J. guided the project and data interpretation. H.J.L. and Y.K.J. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1, S2, S3, S4, S5 and Methods (PDF 5724 kb)
Rights and permissions
About this article
Cite this article
Lee, HJ., Pyo, JO., Oh, Y. et al. AK2 activates a novel apoptotic pathway through formation of a complex with FADD and caspase-10. Nat Cell Biol 9, 1303–1310 (2007). https://doi.org/10.1038/ncb1650
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1650
This article is cited by
-
AK2 is an AMP-sensing negative regulator of BRAF in tumorigenesis
Cell Death & Disease (2022)
-
Quantitative proteomic analysis reveals AK2 as potential biomarker for late normal tissue radiotoxicity
Radiation Oncology (2019)
-
Caspase-10 inhibits ATP-citrate lyase-mediated metabolic and epigenetic reprogramming to suppress tumorigenesis
Nature Communications (2019)
-
Flavonoids of Rosa roxburghii Tratt offers protection against radiation induced apoptosis and inflammation in mouse thymus
Apoptosis (2018)
-
E2-25K SUMOylation inhibits proteasome for cell death during cerebral ischemia/reperfusion
Cell Death & Disease (2016)