Abstract
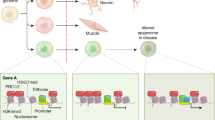
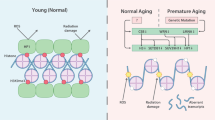
Epigenetic modifications of DNA and histones might be crucial for understanding the molecular basis of complex phenotypes. One reason for this is that epigenetic factors are sometimes malleable and plastic enough to react to cues from the external and internal environments. Such induced epigenetic changes can be solidified and propagated during cell division, resulting in permanent maintenance of the acquired phenotype. In addition, the finding that there is partial epigenetic stability in somatic and germline cells allows insight into the molecular mechanisms of heritability. Epigenetics can provide a new framework for the search of aetiological factors in complex traits and diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hemminki, K., Lorenzo Bermejo, J. & Forsti, A. The balance between heritable and environmental aetiology of human disease. Nature Rev. Genet. 7, 958–965 (2006).
Taubes, G. Epidemiology faces its limits. Science 269, 164–169 (1995).
Austin, J. Schizophrenia: an update and review. J. Genet. Couns. 14, 329–340 (2005).
Kendler, K. S. & Baker, J. H. Genetic influences on measures of the environment: a systematic review. Psychol. Med. 37, 615–626 (2007).
Alberg, A. J. & Samet, J. M. Epidemiology of lung cancer. Chest 123, 21S–49S (2003).
Kendler, K. S., Thornton, L. M. & Pedersen, N. L. Tobacco consumption in Swedish twins reared apart and reared together. Arch. Gen. Psychiatry 57, 886–892 (2000).
Kendler, K. S. & Karkowski-Shuman, L. Stressful life events and genetic liability to major depression: genetic control of exposure to the environment? Psychol. Med. 27, 539–547 (1997).
Plomin, R. & Daniels, D. Why are children in the same family so different from one another? Behav. Brain Sci. 14, 373–427 (1987).
Plomin, R. Environment and genes. Determinants of behavior. Am. Psychol. 44, 105–111 (1989).
Bouchard, T. J. Jr & McGue, M. Genetic and environmental influences on human psychological differences. J. Neurobiol. 54, 4–45 (2003).
Lichtenstein, P. et al. Environmental and heritable factors in the causation of cancer — analyses of cohorts of twins from Sweden, Denmark, and Finland. N. Engl. J. Med. 343, 78–85 (2000).
Harris, J. R. The Nurture Assumption: Why Children Turn Out the Way They Do 462 (Touchstone, 1999).
Turkheimer, E. & Waldron, M. Nonshared environment: a theoretical, methodological, and quantitative review. Psychol. Bull. 126, 78–108 (2000).
Gartner, K. & Baunack, E. Is the similarity of monozygotic twins due to genetic factors alone? Nature 292, 646–647 (1981). In this study, the phenotypic variation among isogenic monozygotic twins in mice was compared with that of dizygotic twins, and the authors concluded that a significant proportion of phenotypic variation cannot be explained by DNA sequences and environmental factors.
Edwards, J. L. et al. Cloning adult farm animals: a review of the possibilities and problems associated with somatic cell nuclear transfer. Am. J. Reprod. Immunol. 50, 113–123 (2003).
Rhind, S. M. et al. Cloned lambs — lessons from pathology. Nature Biotechnol. 21, 744–745 (2003).
Yanagimachi, R. Cloning: experience from the mouse and other animals. Mol. Cell. Endocrinol. 187, 241–248 (2002).
Martin, N., Boomsma, D. & Machin, G. A twin-pronged attack on complex traits. Nature Genet. 17, 387–392 (1997).
Boomsma, D., Busjahn, A. & Peltonen, L. Classical twin studies and beyond. Nature Rev. Genet. 3, 872–882 (2002).
Jockin, V., McGue, M. & Lykken, D. T. Personality and divorce: a genetic analysis. J. Pers. Soc. Psychol. 71, 288–299 (1996).
Turkheimer, E., Haley, A., Waldron, M., D'Onofrio, B. & Gottesman, I. I. Socioeconomic status modifies heritability of IQ in young children. Psychol. Sci. 14, 623–628 (2003).
Visscher, P. M., Hill, W. G. & Wray, N. R. Heritability in the genomics era — concepts and misconceptions. Nature Rev. Genet. 9, 255–266 (2008). This paper discusses the complexities in interpreting heritability.
Heath, A. C., Eaves, L. J. & Martin, N. G. Interaction of marital status and genetic risk for symptoms of depression. Twin Res. 1, 119–122 (1998).
Stokstad, E. Biophysics: DNA on the big screen. Science 275, 1882 (1997).
Gottesman, I. I. Twins: en route to QTLs for cognition. Science 276, 1522–1523 (1997).
Maher, B. Personal genomes: the case of the missing heritability. Nature 456, 18–21 (2008).
Feuk, L., Carson, A. R. & Scherer, S. W. Structural variation in the human genome. Nature Rev. Genet. 7, 85–97 (2006).
Frazer, K. A., Murray, S. S., Schork, N. J. & Topol, E. J. Human genetic variation and its contribution to complex traits. Nature Rev. Genet. 10, 241–251 (2009).
Vogel, F. & Motulsky, A. Human Genetics: Problems and Approaches 851 (Springer, 1997).
Richards, E. J. Inherited epigenetic variation — revisiting soft inheritance. Nature Rev. Genet. 7, 395–401 (2006). This review provides an informative and balanced summary of epigenetic heritability across generations and its possible role in evolution.
Jirtle, R. L. & Skinner, M. K. Environmental epigenomics and disease susceptibility. Nature Rev. Genet. 8, 253–262 (2007). This review summarizes the evidence that environmental factors can change the epigenetic regulation of genes, as well as that certain environmentally induced epigenetic modifications can be heritable.
Weaver, I. C. et al. Epigenetic programming by maternal behavior. Nature Neurosci. 7, 847–854 (2004).
Collins, A. et al. Exercise improves cognitive responses to psychological stress through enhancement of epigenetic mechanisms and gene expression in the dentate gyrus. PLoS ONE 4, e4330 (2009).
Fagiolini, M., Jensen, C. L. & Champagne, F. A. Epigenetic influences on brain development and plasticity. Curr. Opin. Neurobiol. 19, 207–212 (2009).
Ushijima, T. et al. Fidelity of the methylation pattern and its variation in the genome. Genome Res. 13, 868–874 (2003).
Wong, A. H., Gottesman, I. I. & Petronis, A. Phenotypic differences in genetically identical organisms: the epigenetic perspective. Hum. Mol. Genet. 14, R11–R18 (2005).
Bouchard, T. J. Jr, Lykken, D. T., McGue, M., Segal, N. L. & Tellegen, A. Sources of human psychological differences: the Minnesota Study of Twins Reared Apart. Science 250, 223–228 (1990). This landmark study in human research challenges ideas about the importance of environment on several physical and psychological traits, which were investigated in pairs of monozygotic and dizygotic twins who had been reared apart and together.
Finch, C. E. & Kirkwood, T. Chance, Development, and Aging (Oxford Univ. Press, 2000).
Gartner, K. A third component causing random variability beside environment and genotype. A reason for the limited success of a 30 year long effort to standardize laboratory animals? Lab. Anim. 24, 71–77 (1990).
Blewitt, M. E., Vickaryous, N. K., Paldi, A., Koseki, H. & Whitelaw, E. Dynamic reprogramming of DNA methylation at an epigenetically sensitive allele in mice. PLoS Genet. 2, e49 (2006).
Flanagan, J. M. et al. Intra- and interindividual epigenetic variation in human germ cells. Am. J. Hum. Genet. 79, 67–84 (2006).
Haaf, T. Methylation dynamics in the early mammalian embryo: implications of genome reprogramming defects for development. Curr. Top. Microbiol. Immunol. 310, 13–22 (2006).
Mayer, W., Niveleau, A., Walter, J., Fundele, R. & Haaf, T. Demethylation of the zygotic paternal genome. Nature 403, 501–502 (2000).
Rideout, W. M., Eggan, K. & Jaenisch, R. Nuclear cloning and epigenetic reprogramming of the genome. Science 293, 1093–1098 (2001).
Kaminsky, Z. A. et al. DNA methylation profiles in monozygotic and dizygotic twins. Nature Genet. 41, 240–245 (2009).
Kerkel, K. et al. Genomic surveys by methylation-sensitive SNP analysis identify sequence-dependent allele-specific DNA methylation. Nature Genet. 40, 904–908 (2008).
Heijmans, B. T., Kremer, D., Tobi, E. W., Boomsma, D. I. & Slagboom, P. E. Heritable rather than age-related environmental and stochastic factors dominate variation in DNA methylation of the human IGF2/H19 locus. Hum. Mol. Genet. 16, 547–554 (2007).
Hajkova, P. et al. Chromatin dynamics during epigenetic reprogramming in the mouse germ line. Nature 452, 877–881 (2008).
Surani, M. A., Durcova-Hills, G., Hajkova, P., Hayashi, K. & Tee, W. W. Germ line, stem cells, and epigenetic reprogramming. Cold Spring Harb. Symp. Quant. Biol. 73, 9–15 (2008).
Johannes, F. et al. Assessing the impact of transgenerational epigenetic variation on complex traits. PLoS Genet. 5, e1000530 (2009).
Macleod, D., Clark, V. H. & Bird, A. Absence of genome-wide changes in DNA methylation during development of the zebrafish. Nature Genet. 23, 139–140 (1999).
Lane, N. et al. Resistance of IAPs to methylation reprogramming may provide a mechanism for epigenetic inheritance in the mouse. Genesis 35, 88–93 (2003).
Silva, A. J. & White, R. Inheritance of allelic blueprints for methylation patterns. Cell 54, 145–152 (1988).
Youngson, N. A. & Whitelaw, E. Transgenerational epigenetic effects. Annu. Rev. Genomics Hum. Genet. 9, 233–257 (2008).
Chong, S., Youngson, N.A. & Whitelaw, E. Heritable germline epimutation is not the same as transgenerational epigenetic inheritance. Nature Genet. 39, 574–575 (2007).
Suter, C. M. & Martin, D. I. K. Inherited epimutation or a haplotypic basis for the propensity to silence? Nature Genet. 39, 573 (2007).
Suter, C. M. & Martin, D. I. K. Reply to “Heritable germline epimutation is not the same as transgenerational epigenetic inheritance”. Nature Genet. 39, 575–576 (2007).
Pilia, G. et al. Heritability of cardiovascular and personality traits in 6,148 Sardinians. PLoS Genet. 2, e132 (2006).
Hong, L. E. et al. Sensory gating endophenotype based on its neural oscillatory pattern and heritability estimate. Arch. Gen. Psychiatry 65, 1008–1016 (2008).
Allen, N. D., Norris, M. L. & Surani, M. A. Epigenetic control of transgene expression and imprinting by genotype-specific modifiers. Cell 61, 853–861 (1990).
Petronis, A. & Kennedy, J. L. Unstable genes — unstable mind? Am. J. Psychiatry 152, 164–172 (1995).
Timshel, S., Therkildsen, C., Bendahl, P. O., Bernstein, I. & Nilbert, M. An effect from anticipation also in hereditary nonpolyposis colorectal cancer families without identified mutations. Cancer Epidemiol. 33, 231–234 (2009).
McFaul, C. D. et al. Anticipation in familial pancreatic cancer. Gut 55, 252–258 (2006).
Petronis, A., Kennedy, J. L. & Paterson, A. D. Genetic anticipation: fact or artifact, genetics or epigenetics? Lancet 350, 1403–1404 (1997).
Sollars, V. et al. Evidence for an epigenetic mechanism by which Hsp90 acts as a capacitor for morphological evolution. Nature Genet. 33, 70–74 (2003).
Jablonka, E. & Lamb, M.J. Precis of evolution in four dimensions. Behav. Brain Sci. 30, 353–365; 365–389 (2007).
Pigliucci, M. Modelling phenotypic plasticity. II. Do genetic correlations matter? Heredity 77, 453–460 (1996).
Pal, C. & Miklos, I. Epigenetic inheritance, genetic assimilation and speciation. J. Theor. Biol. 200, 19–37 (1999).
De Vries, H. Species and Varieties: Their Origin by Mutation (Open Court, 1904).
Kuhn, T. S. The Structure of Scientific Revolutions 172 (Univ. Chicago Press, 1962).
Petronis, A. Human morbid genetics revisited: relevance of epigenetics. Trends Genet. 17, 142–146 (2001).
Pauling, L. et al. Sickle cell anemia, a molecular disease. Science 109, 543–548 (1949).
Risch, N. Genetic linkage and complex diseases, with special reference to psychiatric disorders. Genet. Epidemiol. 7, 3–16; 17–45 (1990).
Laird, P. W. Principles and challenges of genome-wide DNA methylation analysis. Nature Rev. Genet. 11, 191–203 (2010).
Lister, R. et al. Human DNA methylomes at base resolution show widespread epigenomic differences. Nature 462, 315–322 (2009).
Acknowledgements
I thank A. Wong, I. Gottesman, A. Paterson and C. Ptak for editorial suggestions, A. Schumacher for figure design, and the reviewers for their comments. Work in my laboratory has been supported by the Canadian Institutes of Health Research (grants 199170 and 186007) and the US National Institutes of Health (grants MH074127, MH088413, DP3DK085698 and HG004535). I am Tapscott Chair in Schizophrenia Studies at the University of Toronto and a senior fellow of the Ontario Mental Health Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Additional information
Reprints and permissions information is available at http://www.nature.com/reprints.
Rights and permissions
About this article
Cite this article
Petronis, A. Epigenetics as a unifying principle in the aetiology of complex traits and diseases. Nature 465, 721–727 (2010). https://doi.org/10.1038/nature09230
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09230
This article is cited by
-
Gene body hypomethylation of pyroptosis-related genes NLRP7, NLRP2, and NLRP3 facilitate non-invasive surveillance of hepatocellular carcinoma
Functional & Integrative Genomics (2023)
-
Cell-type-specific DNA methylation analysis of the frontal cortices of mutant Polg1 transgenic mice with neuronal accumulation of deleted mitochondrial DNA
Molecular Brain (2022)
-
Assessing Differential Variability of High-Throughput DNA Methylation Data
Current Environmental Health Reports (2022)
-
The Role of Alcohol Dependence and Depressive Disorder in Childhood Adversities and Reproductive History Traits in Inpatient Women: a Mediation Analyses
International Journal of Mental Health and Addiction (2022)
-
DNA methylation biomarkers of myocardial infarction and cardiovascular disease
Clinical Epigenetics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.