Abstract
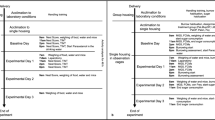
Laparoscopic surgery offers advantages for both animal welfare and quality of experimental data. Compared with laparotomy, laparoscopy is associated with less postoperative pain and faster recuperation in humans and is also associated with less postoperative pain in dogs. Postoperative pain associated with laparotomy and laparoscopy has not been compared in rodents, however. The authors used a validated pain grimace scale to evaluate postoperative pain in male Sprague Dawley rats after liver biopsy by laparotomy or laparoscopy. Rats that underwent laparoscopy showed fewer recognized signs of pain than did rats that underwent laparotomy. The authors suggest that laparoscopy could be used for repeated biopsies in rats, minimizing the number of animals used in pharmacological and toxicological studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Berguer, R., Gutt, C. & Stiegmann, G.V. Laparoscopic surgery in the rat. Surg. Endosc. 7, 345–347 (1993).
Baran, S.W., Perret-Gentil, M.I., Johnson, E.J., Miedel, E.L. & Kehler, J. Rodent laparoscopy, refinement for rodent drug studies and model development, and monitoring of neoplastic, inflammatory and metabolic diseases. Lab. Anim. 45, 231–239 (2011).
Fernandez-Pineda, I., Millan, A., Morcillo, J. & De Agustin, J.C. Laparoscopic surgery in a rat model. J. Laparoendosc. Adv. Surg. Tech. A 20, 575–576 (2010).
Galesso, M.P. et al. Laparoscopic orchiectomy, experimental rat model. Brazil J. Urol. 28, 143–146 (2002).
Perret-Gentil, M. et al. Videoendoscopy, an effective and efficient way to perform multiple visceral biopsies in small animals. J. Invest. Surg. 12, 157–165 (1999).
Shapira, Y. et al. Utilization of murine laparoscopy for continuous in-vivo assessment of the liver in multiple disease models. PLoS ONE 4, e4776 (2009).
Decadt, B. et al. Randomized clinical trial of early laparoscopy in the management of acute non-specific abdominal pain. Br. J. Surg. 86, 1383–1386 (1999).
Gower, S. & Mayhew, P. Canine laparoscopic and laparoscopic-assisted ovariohysterectomy and ovariectomy. Compend. Contin. Educ. Vet. 30, 430–440 (2008).
Kuntz, C. et al. Comparison of laparoscopic versus conventional technique in colonic and liver resection in a tumor-bearing small animal model. Surg. Endosc. 16, 1175–1181 (2002).
Matsumiya, L.C. et al. Using the mouse grimace scale to reevaluate the efficacy of postoperative analgesics in laboratory mice. J. Am. Assoc. Lab. Anim. Sci. 51, 42–49 (2012).
Sotocinal, S.G. et al. The Rat Grimace Scale, a partially automated method for quantifying pain in the laboratory rat via facial expressions. Mol. Pain 7, 55 (2011).
Berger, R., Cornelius, T. & Dalton, M. The optimum pneumoperitoneal pressure for laparoscopic surgery in the rat model. Surg. Endosc. 11, 915–918 (1997).
Gutt, C.N. et al. Standardized technique of laparoscopic surgery in the rat. Dig. Surg. 15, 135–139 (1998).
Gutt, C.N., Kim, Z.-G. & Krähenbühl, L. Training for advanced laparoscopic surgery. Eur. J. Surg. 168, 172–178 (2002).
Van Velthoven, R.F. & Hoffmann, P. Methods for laparoscopic training using animal models. Curr. Urol. Rep. 7, 114–119 (2006).
de Menezes Ettinger, J.E. et al. Laparoscopic gastric banding in the rat model as a means of videolaparoscopic training. Obesity Surg. 16, 903–907 (2006).
Karanika, S., Karantanos, T. & Theodoropoulos, G.E. Immune response after laparoscopic colectomy for cancer, a review. Gastroenterol Rep. 1, 85–94 (2013).
Filho, I.A. et al. Influence of laparoscopy and laparotomy on gasometry, leukocytes and cytokines in a rat abdominal sepsis model. Acta Cirurgica Brasil 21, 74–79 (2006).
Huang, S.G. et al. Laparotomy and laparoscopy diversely affect macrophage-associated antimicrobial activity in a murine model. BMC Immunol. 14, 27–34 (2013).
Watson, R.W. et al. Exposure of the peritoneal cavity to air regulates early inflammatory responses to surgery in a murine model. Br. J. Surg. 82, 1060–1065 (1995).
Tung, P.H. & Smith, C.D. Laparoscopic insufflation with room air causes exaggerated interleukin-6 response. Surg. Endosc. 13, 473–475 (1999).
Moehrlen, U. et al. Impact of carbon dioxide versus air pneumoperitoneum on peritoneal cell migration and cell fate. Surg. Endosc. 20, 1607–1613 (2006).
Gitzelmann, C.A. et al. Cell-mediated immune response is better preserved by laparoscopy than laparotomy. Surgery 127, 65–71 (2000).
Nickkholgh, A. et al. Signs of reperfusion injury following CO2 pneumoperitoneum, an in vivo microscopy study. Surg. Endosc. 22, 122–128 (2008).
Hazebroek, E.J. et al. Impact of carbon dioxide and helium insufflation on cardiorespiratory function during prolonged pneumoperitoneum in an experimental rat model. Surg. Endosc. 16, 1073–1078 (2002).
Gutt, C.N. et al. Carbon dioxide pneumoperitoneum is associated with increased liver metastases in a rat model. Surgery 127, 566–570 (2000).
Gutt, C.N. et al. Impact of laparoscopic surgery on experimental hepatic metastases. Br. J. Surg. 88, 371–375 (2001).
Thoolen, B. et al. Proliferative and nonproliferative lesions of the rat and mouse hepatobiliary system. Toxicol. Path. 38 (suppl. 7), 5S–81S (2010).
Kheirabadi, B.S. et al. Metabolic and hemodynamic effects of CO2 pneumoperitoneum in a controlled hemorrhage model. J. Trauma 50, 1031–1043 (2001).
Acknowledgements
We thank Guy Beauchamp (statistician working for the faculty of veterinary medicine of the University of Montreal) for statistical analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Préfontaine, L., Hélie, P. & Vachon, P. Postoperative pain in Sprague Dawley rats after liver biopsy by laparotomy versus laparoscopy. Lab Anim 44, 174–178 (2015). https://doi.org/10.1038/laban.731
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/laban.731


