Abstract
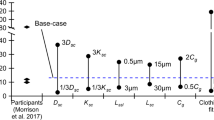
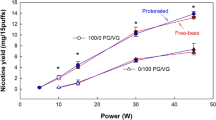
Nicotine plus flavorings in a propylene glycol (PG) vehicle are the components of electronic cigarette liquids (e-liquids), which are vaporized and inhaled by the user. Dermal exposure to nicotine and e-liquids may occur among workers in mixing and filling of e-cigarettes in the manufacturing process. Inadvertent skin contact among consumers is also a concern. In vitro nicotine permeation studies using heat-separated human epidermis were performed with surrogate and two commercial e-liquids, neat and aqueous nicotine donor formulations. Steady-state fluxes (Jss), and lag times (tlag) were measured for each formulation. In addition, transient (4 h) exposure and finite dose (1–10 μl/cm2) experiments were undertaken using one commercial e-liquid. Average Jss (μg/cm2/h) from formulations were: nicotine in PG (24 mg/ml): 3.97; commercial e-liquid containing menthol (25 mg/ml nicotine): 10.2; commercial e-liquid containing limonene (25 mg/ml nicotine): 23.7; neat nicotine: 175. E-liquid lag times ranged from 5 to 10 h. Absorbed fraction of nicotine from finite doses was ≈0.3 at 48 h. The data were applied to transient exposure and finite dose dermal exposure assessment models and to a simple pharmacokinetic model. Three illustrative exposure scenarios demonstrate use of the data to predict systemic uptake and plasma concentrations from dermal exposure. The data demonstrate the potential for significant nicotine absorption through skin contact with e-cigarette refill solutions and the neat nicotine used to mix them.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Grana R, Benowitz N, Glantz SA . E-cigarettes: a scientific review. Circulation 2014; 129: 1972–1986.
CDC. E-cigarette use triples among middle and high school students in just one year. Centers for Disease Control Press Release (2015). Available at http://www.cdc.gov/media/releases/2015/p0416-e-cigarette-use.html. Accessed 9 February 2015.
FDA. FDA and Public Health Experts Warn About Electronic Cigarettes. Food and Drug Administration Press Release. Available at 2009. http://www.fda.gov/newsevents/newsroom/pressannouncements/ucm173222.htm Accessed 21 November 2016.
Konstantinos EF, Polosa R . Safety evaluation and risk assessment of electronic cigarettes as tobacco cigarette substitutes: a systematic review. Ther Adv Drug Saf 2014; 5 (2): 67–86.
Chatham-Stephens K, Law R, Taylor E, Melstrom P, Bunnell R, Wang B et al. Notes from the field: calls to Poison centers for exposures to electronic cigarettes — United States, September 2010–February 2014. MMWR 2014; 63: 292–293.
Richtel M, Selling a poison by the barrel: liquid nicotine for e-cigarettes. New York Times, 23 March 2014. Available at http://www.nytimes.com/2014/03/24/business/selling-a-poison-by-the-barrel-liquid-nicotine-for-e-cigarettes.html?_r=0. Accessed 21 November 2016.
Glatter R, The real dangers of liquid nicotine. Forbes, 24 March 2014. Available at http://www.forbes.com/sites/robertglatter/2014/03/24/the-real-danger-of-liquid-nicotine/. Accessed 21 November 2016.
OSHA. Occupational Safety and Health Administration Regional News Release, 2013. Available at https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=NEWS_RELEASES&p_id=23844. Accessed 21 November 2016.
Williams AC, Barry BW . Penetration enhancers. Adv Drug Deliver Rev 2004; 56: 603–618.
Faulkner JM . Nicotine poisoning by absorption through the skin. JAMA 1933; 100: 1664–1665.
Benowitz NL, Lake T, Keller KH, Lee BL . Prolonged absorption with development of tolerance to toxic effects after cutaneous exposure to nicotine. Clin Pharmacol Ther 1987; 42: 119–120.
Davis P, Levy S, Pahari A, Martinez D . Case report: acute nicotine poisoning associated with a traditional remedy for eczema. Arch Dis Child 2001; 85: 500–502.
Aungst BJ . Nicotine skin penetration characteristics using aqueous and non-aqueous vehicles, anionic polymers, and silicone matrices. Drug Dev Ind Pharm 1988; 14: 1481–1494.
Zorin S, Kuylenstierna F, Thulin H . In vitro test of nicotine’s permeability through human skin. Risk evaluation and safety aspects. Ann Occup Hyg 1999; 43: 405–413.
Qvist MH, Hoeck U, Kreilgaard B, Madsen F, Frokjaer S . Evaluation of the Göttingen minipig skin for transdermal in vitro permeation studies. Eur J Pharm Sci 2000; 11: 59–68.
Kuswahyuning R, Roberts MS . Concentration dependency in nicotine skin penetration flux from aqueous solutions reflects vehicle induced changes in nicotine stratum corneum retention. Pharm Res 2014; 31: 1501–1511.
Barbero AM, Frasch HF . Effect of frozen human epidermis storage duration and cryoprotectant on barrier function using two model compounds. Skin Pharmacol Physiol 2016; 29: 31–40.
Frasch HF, Barbero AM . Application of solid-phase microextraction to in vitro skin permeation experiments: example using diethyl phthalate. Toxicol In Vitro 2005; 19: 253–259.
Crank J . The Mathematics of Diffusion, 2nd edn. Clarendon Press: Oxford. 1975; p 51.
OECD OECD Guideline for the Testing of Chemicals. Organization for Economic Co-Operation and Development: Paris, 2004.
Lim PFC, Liu XY, Kang L, Ho PCL, Chan YW, Chan SY . Limonene GP1/PG organogel as a vehicle in transdermal delivery of haloperidol. Int J Pharm 2006; 311: 157–164.
Candrashekar NS, Hiremath SR . In vivo immunomodulatory, cumulative skin irritation, sensitization and effect of d-limonene on permeation of 6-mercaptopurine through transdermal drug delivery. Biol Pharm Bull 2008; 31: 656–661.
Frasch HF, Bunge AL . The transient dermal exposure II: post-exposure absorption and evaporation of volatile compounds. J Pharm Sci 2015; 104: 1499–1507.
Hansen S, Selzer D, Schaefer UF, Kasting GB . An extended database of keratin binding. J Pharm Sci 2011; 100: 1712–1726.
Benowitz NL, Jacob P, 3rd . Daily intake of nicotine during cigarette smoking. Clin Pharmacol Ther 1984; 35: 499–504.
Gori GB, Lynch CJ . Analytical cigarette yields as predictors of smoke bioavailability. Regul Toxicol Pharmacol 1985; 5: 314–326.
Molander L, Hansson A, Lunell E . Pharmacokinetics of nicotine in healthy elderly people. Clin Pharmacol Ther 2001; 69: 57–65.
Mayer B . How much nicotine kills a human? Tracing back the generally accepted lethal dose to dubious self-experiments in the nineteenth century. Arch Toxicol 2014; 88: 5–7.
Hukkanen J, Jacob P, 3rd, Benowitz NL . Metabolism and disposition kinetics of nicotine. Pharmacol Rev 2005; 57: 79–115.
Woolf A, Burkhart K, Caraccio T, Litovitz T . Self-poisoning among adults using multiple transdermal nicotine patches. J Toxicol Clin Toxicol 1996; 34: 691–698.
Murai M, Lau HK, Pereira BP, Pho RW . A cadaver study on volume and surface area of the fingertip. J Hand Surg Am 1997; 22 (5): 935–941.
EPA A Laboratory Method to Determine the Retention of Liquids on the Surface of Hands. US Environmental Protection Agency. 1992 EPA/747/R-92/003.
Engineering Toolbox. Liquids kinematic viscosities. The Engineering ToolBox http://www.engineeringtoolbox.com/kinematic-viscosity-d_397.html. Accessed 21 November 2016.
EPA. Dermal Exposure FactorsExposure Factors Handbook: 2011 Edition. Ch. 7 National Center for Environmental Assessment: Washington, DC. 2011 EPA/600/R-09/052F.
Acknowledgements
Intramural funding for this research was provided by the National Institute for Occupational Safety and Health, which is part of the Centers for Disease Control. We thank Professor Annette L. Bunge of the Colorado School of Mines for helpful comments and suggestions to incorporate estimates of plasma nicotine concentrations for the assessment of risk from dermal contact. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Frasch, H., Barbero, A. In vitro human epidermal permeation of nicotine from electronic cigarette refill liquids and implications for dermal exposure assessment. J Expo Sci Environ Epidemiol 27, 618–624 (2017). https://doi.org/10.1038/jes.2016.68
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2016.68
Keywords
This article is cited by
-
An update of skin permeability data based on a systematic review of recent research
Scientific Data (2024)
-
Structural, optical and gas sensing properties of Zn-doped CuO nanostructure thin films for benzene gas sensing applications
Journal of Materials Science: Materials in Electronics (2024)
-
Oral nicotine pouches with an aftertaste? Part 2: in vitro toxicity in human gingival fibroblasts
Archives of Toxicology (2023)
-
In vitro human skin permeation of benzene in gasoline: Effects of concentration, multiple dosing and skin preparation
Journal of Exposure Science & Environmental Epidemiology (2018)