Abstract
Objective:
Induction of heme oxygenase-1 (HO-1) has been demonstrated to result in chronic weight loss in several rodent models of obesity. However, the specific contribution of the HO metabolite, carbon monoxide (CO) to this response remains unknown. In this study, we determined the effect of chronic low level administration of a specific CO donor on the progression of obesity and its effects on metabolism and adipocyte biology in mice fed a high-fat diet.
Design:
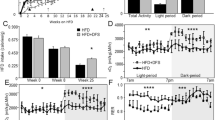
Experiments were performed on C57BL/6J mice fed a high-fat diet (60%) from 4 weeks until 30 weeks of age. Mice were administered either the CO donor, carbon monoxide releasing molecules (CORM)-A1 (5 mg kg−1, intraperitoneally every other day) or the inactive form of the drug (iCORM-A1). Body weights were measured weekly and fasted blood glucose, insulin as well as body composition were measured every 6 weeks. Food intake, O2 consumption, CO2 production, activity and body heat production were measured at 28 weeks after start of the experimental protocol.
Results:
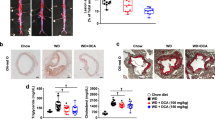
Chronic CORM-A1 attenuated the development of high fat induced obesity from 18 weeks until the end of the study. Chronic CORM-A1 treatment in mice fed a high-fat diet resulted in significant decreases in fasted blood glucose, insulin and body fat and increased O2 consumption and heat production as compared with mice treated with iCORM-A1. Chronic CORM-A1 treatment also resulted in a significant decrease in adipocyte size and an increase in adipocyte number and in NRF-1, PGC-1α and UCP1 protein levels in epidydmal fat.
Conclusion:
Our results demonstrate that chronic CO treatment prevents the development of high-fat diet induced obesity via stimulation of metabolism and remodeling of adipocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Eckel RH, Grundy SM, Zimmet PZ . The metabolic syndrome. Lancet 2005; 365: 1415–1428.
Hotamisligil GS . Inflammation and metabolic disorders. Nature 2006; 444: 860–867.
Verma A, Hirsch DJ, Glatt CE, Ronnett GV, Snyder SH . Carbon monoxide: a putative neural messenger. Science 1993; 259: 381–384.
Johnson RA, Colombari E, Colombari DS, Lavesa M, Talman WT, Nasjletti A . Role of endogenous carbon monoxide in central regulation of arterial pressure. Hypertension 1997; 30: 962–967.
Morse D, Sethi J, Choi AM . Carbon monoxide-dependent signaling. Crit Care Med 2001; 30: S12–S17.
Abraham NG, Kappas A . Pharmacological and clinical aspects of heme oxygenase. Pharmacol Rev 2008; 60: 79–127.
Kaide JI, Zhang F, Wei Y, Jiang H, Yu C, Wang WH et al. Carbon monoxide of vascular origin attenuates the sensitivity of renal arterial vessels to vasoconstrictors. J Clin Invest 2001; 107: 1163–1171.
Parfenova H, Neff RA 3rd, Alonso JS, Shlopov BV, Jamal CN, Sarkisova SA et al. Cerebral vascular endothelial heme oxygenase: expression, localization, and activation by glutamate. Am J Physiol Cell Physiol 2001; 281: C1954–C1963.
Peterson SJ, Drummond G, Kim DH, Li M, Kruger AL, Ikehara S et al. L-4F treatment reduces adiposity, increases adiponectin levels, and improves insulin sensitivity in obese mice. J Lipid Res 2008; 49: 1658–1669.
Li M, Kim DH, Tsenovoy PL, Peterson SJ, Rezzani R, Rodella LF et al. Treatment of obese diabetic mice with a heme oxygenase inducer reduces visceral and subcutaneous adiposity, increases adiponectin levels, and improves insulin sensitivity and glucose tolerance. Diabetes 2008; 57: 1526–1535.
Burgess A, Li M, Vanella L, Kim DH, Rezzani R, Rodella L et al. Adipocyte heme oxygenase-1 induction attenuates metabolic syndrome in both male and female obese mice. Hypertension 2010; 56: 1124–1130.
Csongradi E, Docarmo JM, Dubinion JH, Vera T, Stec DE . Chronic HO-1 induction with cobalt protoporphyrin (CoPP) treatment increases oxygen consumption, activity, heat production and lowers body weight in obese melanocortin-4 receptor-deficient mice. Int J Obes (Lond) 2011; 36: 244–253.
Cao J, Peterson SJ, Sodhi K, Vanella L, Barbagallo I, Rodella LF et al. Heme oxygenase gene targeting to adipocytes attenuates adiposity and vascular dysfunction in mice fed a high-fat diet. Hypertension 2012; 60: 467–475.
Pitchumony TS, Spingler B, Motterlini R, Alberto R . Syntheses, structural characterization and CO releasing properties of boranocarbonate [H3BCO2H]- derivatives. Org Biomol Chem 2010; 8: 4849–4854.
Vummaleti SV, Branduardi D, Masetti M, De Vivo M, Motterlini R, Cavalli A . Theoretical insights into the mechanism of carbon monoxide (CO) release from CO-releasing molecules. Chemistry 2012; 18: 9267–9275.
Motterlini R, Clark JE, Foresti R, Sarathchandra P, Mann BE, Green CJ . Carbon monoxide-releasing molecules: characterization of biochemical and vascular activities. Circ Res 2002; 90: E17–E24.
Clark JE, Naughton P, Shurey S, Green CJ, Johnson TR, Mann BE et al. Cardioprotective actions by a water-soluble carbon monoxide-releasing molecule. Circ Res 2003; 93: e2–e8.
Motterlini R, Otterbein LE . The therapeutic potential of carbon monoxide. Nat Rev Drug Discov 2010; 9: 728–743.
Motterlini R, Sawle P, Hammad J, Bains S, Alberto R, Foresti R et al. CORM-A1: a new pharmacologically active carbon monoxide-releasing molecule. FASEB J 2005; 19: 284–286.
Vera T, Henegar JR, Drummond HA, Rimoldi JM, Stec DE . Protective effect of carbon monoxide-releasing compounds in ischemia-induced acute renal failure. J Am Soc Nephrol 2005; 16: 950–958.
Ryan MJ, Jernigan NL, Drummond HA, McLemore GR Jr, Rimoldi JM, Poreddy SR et al. Renal vascular responses to CORM-A1 in the mouse. Pharmacol Res 2006; 54: 24–29.
Csongradi E, Juncos LA, Drummond HA, Vera T, Stec DE . Role of carbon monoxide in kidney function: is a little carbon monoxide good for the kidney? Curr Pharm Biotechnol 2012; 13: 819–826.
Suliman HB, Carraway MS, Tatro LG, Piantadosi CA . A new activating role for CO in cardiac mitochondrial biogenesis. J Cell Sci 2007; 120: 299–308.
Suliman HB, Carraway MS, Ali AS, Reynolds CM, Welty-Wolf KE, Piantadosi CA . The CO/HO system reverses inhibition of mitochondrial biogenesis and prevents murine doxorubicin cardiomyopathy. J Clin Invest 2007; 117: 3730–3741.
Rhodes MA, Carraway MS, Piantadosi CA, Reynolds CM, Cherry AD, Wester TE et al. Carbon monoxide, skeletal muscle oxidative stress, and mitochondrial biogenesis in humans. Am J Physiol Heart Circ Physiol 2009; 297: H392–H399.
Piantadosi CA, Carraway MS, Babiker A, Suliman HB . Heme oxygenase-1 regulates cardiac mitochondrial biogenesis via Nrf2-mediated transcriptional control of nuclear respiratory factor-1. Circ Res 2008; 103: 1232–1240.
Almeida AS, Queiroga CS, Sousa MF, Alves PM, Vieira HL . Carbon monoxide modulates apoptosis by reinforcing oxidative metabolism in astrocytes: role of Bcl-2. J Biol Chem 2012; 287: 10761–10770.
Tsui TY, Obed A, Siu YT, Yet SF, Prantl L, Schlitt HJ et al. Carbon monoxide inhalation rescues mice from fulminant hepatitis through improving hepatic energy metabolism. Shock 2007; 27: 165–171.
Lancel S, Hassoun SM, Favory R, Decoster B, Motterlini R, Neviere R . Carbon monoxide rescues mice from lethal sepsis by supporting mitochondrial energetic metabolism and activating mitochondrial biogenesis. J Pharmacol Exp Ther 2009; 329: 641–648.
Lancel S, Montaigne D, Marechal X, Marciniak C, Hassoun SM, Decoster B et al. Carbon monoxide improves cardiac function and mitochondrial population quality in a mouse model of metabolic syndrome. PLoS One 2012; 7: e41836.
Sandouka A, Balogun E, Foresti R, Mann BE, Johnson TR, Tayem Y et al. Carbon monoxide-releasing molecules (CO-RMs) modulate respiration in isolated mitochondria. Cell Mol Biol (Noisy -le-grand) 2005; 51: 425–432.
Lo IL, Boczkowski J, Zini R, Salouage I, Berdeaux A, Motterlini R et al. A carbon monoxide-releasing molecule (CORM-3) uncouples mitochondrial respiration and modulates the production of reactive oxygen species. Free Radic Biol Med 2011; 50: 1556–1564.
Cypess AM, Kahn CR . Brown fat as a therapy for obesity and diabetes. Curr Opin Endocrinol Diabetes Obes 2010; 17: 143–149.
Tseng YH, Cypess AM, Kahn CR . Cellular bioenergetics as a target for obesity therapy. Nat Rev Drug Discov 2010; 9: 465–482.
Ziouzenkova O, Orasanu G, Sharlach M, Akiyama TE, Berger JP, Viereck J et al. Retinaldehyde represses adipogenesis and diet-induced obesity. Nat Med 2007; 13: 695–702.
Schwarz EJ, Reginato MJ, Shao D, Krakow SL, Lazar MA . Retinoic acid blocks adipogenesis by inhibiting C/EBPbeta-mediated transcription. Mol Cell Biol 1997; 17: 1552–1561.
Villarroya F, Iglesias R, Giralt M . Retinoids and retinoid receptors in the control of energy balance: novel pharmacological strategies in obesity and diabetes. Curr Med Chem 2004; 11: 795–805.
Kiefer FW, Vernochet C, O’Brien P, Spoerl S, Brown JD, Nallamshetty S et al. Retinaldehyde dehydrogenase 1 regulates a thermogenic program in white adipose tissue. Nat Med 2012; 18: 918–925.
Shukla V, Singh SN, Vats P, Singh VK, Singh SB, Banerjee PK . Ghrelin and leptin levels of sojourners and acclimatized lowlanders and high altitude. Nutr Neurosci 2005; 8: 161–165.
Vats P, Singh VK, Singh SN, Singh SB . High altitude induced anorexia: effect of changes in leptin and oxidative stress levels. Nutr Neurosci 2007; 10: 243–249.
Norese M, Lezón CE, Alippi RM, Martínez MP, Conti MI, Bozzini CE . Failure of polycythemia-induced increase in arterial oxygen content to supress the anorexic effect of simulated high altitude in the adult rat. High Alt Med Biol 2002; 3: 49–57.
Acknowledgements
The authors would like to acknowledge the support of grants from the National Heart, Lung and Blood Institute, PO1HL-51971, HL088421 (DES) and 1T32HL105324 (PAH). The authors gratefully acknowledge the Analytical and Assay Core Laboratory in the Department of Physiology and Biophysics at the University of Mississippi Medical Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Hosick, P., AlAmodi, A., Storm, M. et al. Chronic carbon monoxide treatment attenuates development of obesity and remodels adipocytes in mice fed a high-fat diet. Int J Obes 38, 132–139 (2014). https://doi.org/10.1038/ijo.2013.61
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2013.61
Keywords
This article is cited by
-
Independent and combined associations of solid-fuel use and smoking with obesity among rural Chinese adults
Environmental Science and Pollution Research (2021)
-
Association of biomass fuel use with reduced body weight of adult Ghanaian women
Journal of Exposure Science & Environmental Epidemiology (2020)
-
Progesterone receptor membrane associated component 1 enhances obesity progression in mice by facilitating lipid accumulation in adipocytes
Communications Biology (2020)
-
Inhalation of carbon monoxide is ineffective as a long-term therapy to reduce obesity in mice fed a high fat diet
BMC Obesity (2014)
-
Heme oxygenase-1: an emerging therapeutic target to curb cardiac pathology
Basic Research in Cardiology (2014)