Abstract
Context:
Patients with pseudohypoparathyroidism type 1a (PHP-1a) develop early-onset obesity. The abnormality in energy expenditure and/or energy intake responsible for this weight gain is unknown.
Objective:
The aim of this study was to evaluate energy expenditure in children with PHP-1a compared with obese controls.
Patients:
We studied 6 obese females with PHP-1a and 17 obese female controls. Patients were recruited from a single academic center.
Measurements:
Resting energy expenditure (REE) and thermogenic effect of a high fat meal were measured using whole room indirect calorimetry. Body composition was assessed using whole body dual energy x-ray absorptiometry. Fasting glucose, insulin, and hemoglobin A1C were measured.
Results:
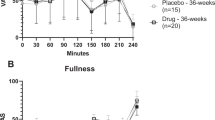
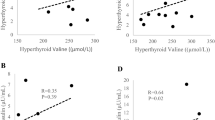
Children with PHP-1a had decreased REE compared with obese controls (P<0.01). After adjustment for fat-free mass, the PHP-1a group’s REE was 346.4 kcals day−1 less than obese controls (95% CI (−585.5–−106.9), P<0.01). The thermogenic effect of food (TEF), expressed as percent increase in postprandial energy expenditure over REE, was lower in PHP-1a patients than obese controls, but did not reach statistical significance (absolute reduction of 5.9%, 95% CI (−12.2–0.3%), P=0.06).
Conclusions:
Our data indicate that children with PHP-1a have decreased REE compared with the obese controls, and that may contribute to the development of obesity in these children. These patients may also have abnormal diet-induced thermogenesis in response to a high-fat meal. Understanding the causes of obesity in PHP-1a may allow for targeted nutritional or pharmacologic treatments in the future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Long DN, McGuire S, Levine MA, Weinstein LS, Germain-Lee EL . Body mass index differences in pseudohypoparathyroidism type 1a versus pseudopseudohypoparathyroidism may implicate paternal imprinting of Galpha(s) in the development of human obesity. J Clin Endocrinol Metab 2007; 92: 1073–1079.
Chen M, Gavrilova O, Liu J, Xie T, Deng C, Nguyen AT et al. Alternative Gnas gene products have opposite effects on glucose and lipid metabolism. Proc Natl Acad Sci USA 2005; 102: 7386–7391.
Farooqi IS, Keogh JM, Yeo GS, Lank EJ, Cheetham T, O’Rahilly S . Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N Engl J Med 2003; 348: 1085–1095.
Thearle MS, Muller YL, Hanson RL, Mullins M, Abdussamad M, Tran J et al. Greater impact of melanocortin-4 receptor deficiency on rates of growth and risk of type 2 diabetes mellitus during childhood compared with adulthood in Pima Indians. Diabetes 2011; Nov: 21.
Chen M, Wang J, Dickerson KE, Kelleher J, Xie T, Gupta D et al. Central nervous system imprinting of the G protein G(s)alpha and its role in metabolic regulation. Cell Metab 2009; 9: 548–555.
Fan W, Dinulescu DM, Butler AA, Zhou J, Marks DL, Cone RD . The central melanocortin system can directly regulate serum insulin levels. Endocrinology 2000; 141: 3072–3079.
Obici S, Feng Z, Tan J, Liu L, Karkanias G, Rossetti L . Central melanocortin receptors regulate insulin action. J Clin Invest 2001; 108: 1079–1085.
Butler AA, Marks DL, Fan W, Kuhn CM, Bartolome M, Cone RD . Melanocortin-4 receptor is required for acute homeostatic responses to increased dietary fat. Nat Neurosci 2001; 4: 605–611.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC . Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412–419.
Weir JB . New methods for calculating metabolic rate with special reference to protein metabolism. J Physiol 1949; 109: 1–9.
Molnar D, Jeges S, Erhardt E, Schutz Y . Measured and predicted resting metabolic rate in obese and nonobese adolescents. J Pediatr 1995; 127: 571–577.
Hofsteenge GH, Chinapaw MJ, Delemarre-van de Waal HA, Weijs PJ . Validation of predictive equations for resting energy expenditure in obese adolescents. Am J Clin Nutr 2010; 91: 1244–1254.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG . Research electronic data capture (REDCap)-a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 2009; 42: 377–381.
Huber P . The behavior of maximum likelihood estimates under nonstandard conditions. Proceedings of the Fifth Berkeley Symposium on Mathematical Statistics and Probability 1967; I: 221–233.
Ong KK, Amin R, Dunger DB . Pseudohypoparathyroidism--another monogenic obesity syndrome. Clin Endocrinol (Oxf) 2000; 52: 389–391.
Lomenick JP, El-Sayyid M, Smith WJ . Effect of levo-thyroxine treatment on weight and body mass index in children with acquired hypothyroidism. J Pediatr 2008; 152: 96–100.
Greenfield JR, Miller JW, Keogh JM, Henning E, Satterwhite JH, Cameron GS et al. Modulation of blood pressure by central melanocortinergic pathways. N Engl J Med 2009; 360: 44–52.
Nwosu BU, Lee MM . Pseudohypoparathyroidism type 1a and insulin resistance in a child. Nat Rev Endocrinol 2009; 5: 345–350.
Dekelbab BH, Aughton DJ, Levine MA . Pseudohypoparathyroidism type 1A and morbid obesity in infancy. Endocr Pract 2009; 15: 249–253.
Germain-Lee EL, Groman J, Crane JL, Jan de Beur SM, Levine MA . Growth hormone deficiency in pseudohypoparathyroidism type 1a: another manifestation of multihormone resistance. J Clin Endocrinol Metab 2003; 88: 4059–4069.
Mantovani G, Maghnie M, Weber G, De Menis E, Brunelli V, Cappa M et al. Growth hormone-releasing hormone resistance in pseudohypoparathyroidism type ia: new evidence for imprinting of the Gs alpha gene. J Clin Endocrinol Metab 2003; 88: 4070–4074.
Stenlof K, Sjostrom L, Lonn L, Bosaeus I, Kvist H, Tolli J et al. Effects of recombinant human growth hormone on basal metabolic rate in adults with pituitary deficiency. Metabolism 1995; 44: 67–74.
Chong PK, Jung RT, Scrimgeour CM, Rennie MJ, Paterson CR . Energy expenditure and body composition in growth hormone deficient adults on exogenous growth hormone. Clin Endocrinol (Oxf) 1994; 40: 103–110.
Karmisholt J, Andersen S, Laurberg P . Weight loss after therapy of hypothyroidism is mainly caused by excretion of excess body water associated with myxoedema. J Clin Endocrinol Metab 2011; 96: E99–103.
Kong WM, Sheikh MH, Lumb PJ, Naoumova RP, Freedman DB, Crook M et al. A 6-month randomized trial of thyroxine treatment in women with mild subclinical hypothyroidism. Am J Med 2002; 112: 348–354.
Krakoff J, Ma L, Kobes S, Knowler WC, Hanson RL, Bogardus C et al. Lower metabolic rate in individuals heterozygous for either a frameshift or a functional missense MC4R variant. Diabetes 2008; 57: 3267–3272.
Schutz Y, Bessard T, Jequier E . Diet-induced thermogenesis measured over a whole day in obese and nonobese women. Am J Clin Nutr 1984; 40: 542–552.
Camastra S, Bonora E, Del Prato S, Rett K, Weck M, Ferrannini E . Effect of obesity and insulin resistance on resting and glucose-induced thermogenesis in man. EGIR (European Group for the Study of Insulin Resistance). Int J Obes Relat Metab Disord 1999; 23: 1307–1313.
Segal KR, Albu J, Chun A, Edano A, Legaspi B, Pi-Sunyer FX . Independent effects of obesity and insulin resistance on postprandial thermogenesis in men. J Clin Invest 1992; 89: 824–833.
Farooqi IS, Yeo GS, Keogh JM, Aminian S, Jebb SA, Butler G et al. Dominant and recessive inheritance of morbid obesity associated with melanocortin 4 receptor deficiency. J Clin Invest 2000; 106: 271–279.
Henry CJ, Lightowler HJ, Marchini J . Intra-individual variation in resting metabolic rate during the menstrual cycle. Br J Nutr 2003; 89: 811–817.
Melanson KJ, Saltzman E, Russell R, Roberts SB . Postabsorptive and postprandial energy expenditure and substrate oxidation do not change during the menstrual cycle in young women. J Nutr 1996; 126: 2531–2538.
Acknowledgements
We thank Rachel Lippert for her assistance with the mutation analysis and Lauren Whitaker for her assistance with the indirect calorimetry. We would like to acknowledge all the patients and their families who made this research possible. This study was supported in part by NIH grants 5T32HD060554-02 (AHS), the Vanderbilt CTSA grant URL1 RR024975-01 from NCRR/NIH, and 5 M01 RR-000095 from the NCRR/NIH (AHS), RO1DK070332 (RDC), and by a Fellows Development Research Grant in Diabetes, Obesity and Fat Cell Biology from the Endocrine Fellows Foundation (AHS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Shoemaker, A., Lomenick, J., Saville, B. et al. Energy expenditure in obese children with pseudohypoparathyroidism type 1a. Int J Obes 37, 1147–1153 (2013). https://doi.org/10.1038/ijo.2012.200
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2012.200
Keywords
This article is cited by
-
Pseudohypoparathyroidism, acrodysostosis, progressive osseous heteroplasia: different names for the same spectrum of diseases?
Endocrine (2021)
-
An appraisal of whole-room indirect calorimeters and a metabolic cart for measuring resting and active metabolic rates
Scientific Reports (2020)
-
Diagnosis and management of pseudohypoparathyroidism and related disorders: first international Consensus Statement
Nature Reviews Endocrinology (2018)
-
Genetic and epigenetic alterations in the GNAS locus and clinical consequences in Pseudohypoparathyroidism: Italian common healthcare pathways adoption
Italian Journal of Pediatrics (2016)
-
Eating behaviors in obese children with pseudohypoparathyroidism type 1a: a cross-sectional study
International Journal of Pediatric Endocrinology (2014)