Abstract
Objective:
To identify the emotional and motivational processes that reinstate palatable food intake following removal of high-fat diet (HFD) and associated neuroadaptations tied to neurochemical and behavioural changes underlying dopaminergic function.
Methods:
Adult male C57Bl6 mice were placed on a HFD (58% kcal fat) or ingredient-matched, low-fat diet (LFD; 11% kcal fat) for 6 weeks. At the end of diet-regimen mice were either maintained on their respective diets, or HFD and LFD were replaced with normal chow (withdrawal). Effort-based operant responding for sucrose and high-fat food rewards was measured along with basal and stress-induced corticosterone levels and anxiety (elevated-plus maze). Protein levels for tyrosine hydroxylase (TH), corticosterone releasing factor type 1 receptor (CRF-R1), brain-derived neurotrophic factor (BDNF), phospho-CREB (pCREB) and ΔFosB (truncated splice variant of FosB) were assessed in the amygdala, nucleus accumbens (NAc) and ventral tegmental area (VTA) via western immunoblotting.
Results:
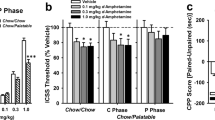
Six weeks of HFD resulting in significant weight gain elicited sucrose anhedonia, anxiety-like behaviour and hypothalamic-pituitary-adrenocortical axis (HPA) hypersensitivity to stress. Withdrawal from HFD but not LFD-potentiated anxiety and basal corticosterone levels and enhanced motivation for sucrose and high-fat food rewards. Chronic high-fat feeding reduced CRF-R1 and increased BDNF and pCREB protein levels in the amygdala and reduced TH and increased ΔFosB protein in NAc and VTA. Heightened palatable food reward in mice withdrawn from HFD coincided with increased BDNF protein levels in NAc and decreased TH and pCREB expression in the amygdala.
Conclusion:
Anhedonia, anxiety and sensitivity to stressors develops during the course of HFD and may have a key role in a vicious cycle that perpetuates high-fat feeding and the development of obesity. Removal of HFD enhances stress responses and heightens vulnerability for palatable foods by increasing food-motivated behaviour. Lasting changes in dopamine and plasticity-related signals in reward circuitry may promote negative emotional states, overeating and palatable food relapse.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gold MS, Graham NA, Cocores JA, Nixon SJ . Food addiction? J Addict Med 2009; 3: 42–45.
Nair SG, Adams-Deutsch T, Epstein DH, Shaham Y . The neuropharmacology of relapse to food seeking: methodology, main findings, and comparison with relapse to drug seeking. Prog Neurobiol 2009; 89: 18–45.
Ghitza UE, Gray SM, Epstein DH, Rice KC, Shaham Y . The anxiogenic drug yohimbine reinstates palatable food seeking in a rat relapse model: a role of CRF1 receptors. Neuropsychopharmacology 2006; 31: 2188–2196.
Nair SG, Navarre BM, Cifani C, Pickens CL, Bossert JM, Shaham Y . Role of dorsal medial prefrontal cortex dopamine D1-family receptors in relapse to high-fat food seeking induced by the anxiogenic drug yohimbine. Neuropsychopharmacology 2011; 36: 497–510.
Ifland JR, Preuss HG, Marcus MT, Rourke KM, Taylor WC, Burau K et al. Refined food addiction: a classic substance use disorder. Med Hypotheses 2009; 72: 518–526.
Fulton S . Appetite and reward. Front Neuroendocrinol 2010; 31: 85–103.
Carlezon WA, Thomas MJ . Biological substrates of reward and aversion: a nucleus accumbens activity hypothesis. Neuropharmacology 2009; 56 (Suppl 1): 122–132.
Etkin A, Prater KE, Schatzberg AF, Menon V, Greicius MD . Disrupted amygdalar subregion functional connectivity and evidence of a compensatory network in generalized anxiety disorder. Arch Gen Psychiatry 2009; 66: 1361–1372.
Tye KM, Prakash R, Kim SY, Fenno LE, Grosenick L, Zarabi H et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature 2011; 471: 358–362.
Hyman SE, Malenka RC, Nestler EJ . Neural mechanisms of addiction: the role of reward-related learning and memory. Annu Rev Neurosci 2006; 29: 565–598.
Russo SJ, Bolanos CA, Theobald DE, DeCarolis NA, Renthal W, Kumar A et al. IRS2-Akt pathway in midbrain dopamine neurons regulates behavioral and cellular responses to opiates. Nat Neurosci 2007; 10: 93–99.
Li Y, South T, Han M, Chen J, Wang R, Huang XF . High-fat diet decreases tyrosine hydroxylase mRNA expression irrespective of obesity susceptibility in mice. Brain Res 2009; 1268: 181–189.
South T, Westbrook F, Morris MJ . Neurological and stress related effects of shifting obese rats from a palatable diet to chow and lean rats from chow to a palatable diet. Physiol Behav 2012; 105: 1052–1057.
Vucetic Z, Carlin J, Totoki K, Reyes TM . Epigenetic dysregulation of the dopamine system in diet-induced obesity. J Neurochem 2012; 120: 891–898.
Sharma S, Fulton S . Diet-induced obesity promotes depressive-like behaviour that is associated with neural adaptations in brain reward circuitry. Int J Obes 2012. e-pub ahead of print 17 April 2012; doi:10:1038/ijo2012.48.
Cottone P, Sabino V, Steardo L, Zorrilla EP . Opioid-dependent anticipatory negative contrast and binge-like eating in rats with limited access to highly preferred food. Neuropsychopharmacology 2008; 33: 524–535.
Cottone P, Sabino V, Steardo L, Zorrilla EP . Intermittent access to preferred food reduces the reinforcing efficacy of chow in rats. Am J Physiol Regul Integr Comp Physiol 2008; 295: R1066–R1076.
Cottone P, Sabino V, Steardo L, Zorrilla EP . Consummatory, anxiety-related and metabolic adaptations in female rats with alternating access to preferred food. Psychoneuroendocrinology 2009; 34: 38–49.
Cottone P, Sabino V, Roberto M, Bajo M, Pockros L, Frihauf JB et al. CRF system recruitment mediates dark side of compulsive eating. Proc Natl Acad Sci USA 2009; 106: 20016–20020.
Pickering C, Alsiö J, Hulting A-L, Schiöth H . Withdrawal from free-choice high-fat high-sugar diet induces craving only in obesity-prone animals. Psychopharmacology (Berl) 2009; 204: 431–443.
Richardson NR, Roberts DC . Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J Neurosci Methods 1996; 66: 1–11.
Sharma S, Hryhorczuk C, Fulton S . Progressive-ratio Responding for Palatable High-fat and High-sugar Food inMice. J Vis Exp 2012; 63: e3754.
Carobrez AP, Bertoglio LJ . Ethological and temporal analyses of anxiety-like behavior: The elevated plus-maze model 20 years on. Neuroscience & Biobehavioral Reviews 2005; 29: 1193–1205.
Papp M, Willner P, Muscat R . An animal model of anhedonia: attenuation of sucrose consumption and place preference conditioning by chronic unpredictable mild stress. Psychopharmacology (Berl) 1991; 104: 255–259.
Teegarden SL, Bale TL . Decreases in Dietary Preference Produce Increased Emotionality and Risk for Dietary Relapse. Biol Psychiatry 2007; 61: 1021–1029.
George O, Ghozland S, Azar MR, Cottone P, Zorrilla EP, Parsons LH et al. CRF-CRF1 system activation mediates withdrawal-induced increases in nicotine self-administration in nicotine-dependent rats. Proc Natl Acad Sci U S A 2007; 104: 17198–17203.
Goeders NE, Guerin GF . Effects of the CRH receptor antagonist CP-154,526 on intravenous cocaine self-administration in rats. Neuropsychopharmacology 2000; 23: 577–586.
Koob GF . Brain stress systems in the amygdala and addiction. Brain Res 2009; 1293: 61–75.
Richter RM, Weiss F . In vivo CRF release in rat amygdala is increased during cocaine withdrawal in self-administering rats. Synapse 1999; 32: 254–261.
Roberto M, Cruz MT, Gilpin NW, Sabino V, Schweitzer P, Bajo M et al. Corticotropin releasing factor-induced amygdala gamma-aminobutyric Acid release plays a key role in alcohol dependence. Biol Psychiatry 2010; 67: 831–839.
Graham DL, Edwards S, Bachtell RK, DiLeone RJ, Rios M, Self DW . Dynamic BDNF activity in nucleus accumbens with cocaine use increases self-administration and relapse. Nat Neurosci 2007; 10: 1029–1037.
Krishnan B, Centeno M, Pollandt S, Fu Y, Genzer K, Liu J et al. Dopamine receptor mechanisms mediate corticotropin-releasing factor-induced long-term potentiation in the rat amygdala following cocaine withdrawal. Eur J Neurosci 2010; 31: 1027–1042.
Nestler EJ, Kelz MB, Chen J . DeltaFosB: a molecular mediator of long-term neural and behavioral plasticity. Brain Res 1999; 835: 10–17.
Russo SJ, Mazei-Robison MS, Ables JL, Nestler EJ . Neurotrophic factors and structural plasticity in addiction. Neuropharmacology 2009; 56 (Suppl 1): 73–82.
Robison AJ, Nestler EJ . Transcriptional and epigenetic mechanisms of addiction. Nat Rev Neurosci 2011; 12: 623–637.
Koob GF . Stress, corticotropin-releasing factor, and drug addiction. Ann N Y Acad Sci 1999; 897: 27–45.
Sarnyai Z . Neurobiology of stress and cocaine addiction. Studies on corticotropin-releasing factor in rats, monkeys, and humans. Ann N Y Acad Sci 1998; 851: 371–387.
Muscat R, Papp M, Willner P . Reversal of stress-induced anhedonia by the atypical antidepressants, fluoxetine and maprotiline. Psychopharmacology (Berl) 1992; 109: 433–438.
Papp M, Willner P, Muscat R . Behavioural sensitization to a dopamine agonist is associated with reversal of stress-induced anhedonia. Psychopharmacology (Berl) 1993; 110: 159–164.
Avena NM, Bocarsly ME, Rada P, Kim A, Hoebel BG . After daily bingeing on a sucrose solution, food deprivation induces anxiety and accumbens dopamine/acetylcholine imbalance. Physiol Behav 2008; 94: 309–315.
Colantuoni C, Rada P, McCarthy J, Patten C, Avena NM, Chadeayne A et al. Evidence that intermittent, excessive sugar intake causes endogenous opioid dependence. Obesity 2002; 10: 478–488.
Bailey A, Davis L, Lesscher HMB, Kelly MDW, Ledent C, Hourani SMO et al. Enhanced morphine withdrawal and μ-opioid receptor G-protein coupling in A2A adenosine receptor knockout mice. J Neurochem 2004; 88: 827–834.
Shepard JD, Bossert JM, Liu SY, Shaham Y . The anxiogenic drug yohimbine reinstates methamphetamine seeking in a rat model of drug relapse. Biol Psychiatry 2004; 55: 1082–1089.
Sinha R, Fox HC, Hong KA, Bergquist K, Bhagwagar Z, Siedlarz KM . Enhanced negative emotion and alcohol craving, and altered physiological responses following stress and cue exposure in alcohol dependent individuals. Neuropsychopharmacology 2009; 34: 1198–1208.
Zhao LY, Shi J, Zhang XL, Epstein DH, Zhang XY, Liu Y et al. Stress enhances retrieval of drug-related memories in abstinent heroin addicts. Neuropsychopharmacology 2010; 35: 720–726.
Ghitza UE, Nair SG, Golden SA, Gray SM, Uejima JL, Bossert JM et al. Peptide YY3-36 decreases reinstatement of high-fat food seeking during dieting in a rat relapse model. J Neurosci 2007; 27: 11522–11532.
Nair SG, Adams-Deutsch T, Pickens CL, Smith DG, Shaham Y . Effects of the MCH1 receptor antagonist SNAP 94847 on high-fat food-reinforced operant responding and reinstatement of food seeking in rats. Psychopharmacology (Berl) 2009; 205: 129–140.
Fulton S, Pissios P, Manchon RP, Stiles L, Frank L, Pothos EN et al. Leptin regulation of the mesoaccumbens dopamine pathway. Neuron 2006; 51: 811–822.
Geiger BM, Haburcak M, Avena NM, Moyer MC, Hoebel BG, Pothos EN . Deficits of mesolimbic dopamine neurotransmission in rat dietary obesity. Neuroscience 2009; 159: 1193–1199.
Davis JF, Tracy AL, Schurdak JD, Tschop MH, Lipton JW, Clegg DJ et al. Exposure to elevated levels of dietary fat attenuates psychostimulant reward and mesolimbic dopamine turnover in the rat. Behav Neurosci 2008; 122: 1257–1263.
Davis C, Patte K, Levitan R, Reid C, Tweed S, Curtis C . From motivation to behaviour: a model of reward sensitivity, overeating, and food preferences in the risk profile for obesity. Appetite 2007; 48: 12–19.
Stice E, Yokum S, Blum K, Bohon C . Weight gain is associated with reduced striatal response to palatable food. J Neurosci 2010; 30: 13105–13109.
Stice E, Yokum S, Burger KS, Epstein LH, Small DM . Youth at risk for obesity show greater activation of striatal and somatosensory regions to food. J Neurosci 2011; 31: 4360–4366.
Volkow ND, Wang GJ, Baler RD . Reward, dopamine and the control of food intake: implications for obesity. Trends Cogn Sci 2011; 15: 37–46.
Jang S, Kim D, Lee Y, Moon S, Oh S . Modulation of Sphingosine 1-Phosphate and Tyrosine Hydroxylase in the Stress-Induced Anxiety. Neurochem Res 2011; 36: 258–267.
Auvinen HE, Romijn JA, Biermasz NR, Pijl H, Havekes L, Smit JW et al. The Effects of high fat diet on the basal activity of the hypothalamus-pituitary-adrenal axis in mice. J Endocrinol 2012; 214: 191–197.
Bale TL, Picetti R, Contarino A, Koob GF, Vale WW, Lee K-F . Mice deficient for both corticotropin-releasing factor receptor 1 (crfr1) and crfr2 have an impaired stress response and display sexually dichotomous anxiety-like behavior. The Journal of Neuroscience 2002; 22: 193–199.
Smith GW, Aubry J-M, Dellu F, Contarino A, Bilezikjian LM, Gold LH et al. Corticotropin releasing factor receptor 1–deficient mice display decreased anxiety, impaired stress response, and aberrant neuroendocrine development. Neuron 1998; 20: 1093–1102.
Carlezon WA, Thome J, Olson VG, Lane-Ladd SB, Brodkin ES, Hiroi N et al. Regulation of cocaine reward by CREB. Science 1998; 282: 2272–2275.
Edwards S, Graham DL, Bachtell RK, Self DW . Region-specific tolerance to cocaine-regulated cAMP-dependent protein phosphorylation following chronic self-administration. Eur J Neurosci 2007; 25: 2201–2213.
Pandey SC, Roy A, Xu T, Mittal N . Effects of protracted nicotine exposure and withdrawal on the expression and phosphorylation of the CREB gene transcription factor in rat brain. J Neurochem 2001; 77: 943–952.
Cordeira JW, Frank L, Sena-Esteves M, Pothos EN, Rios M . Brain-derived neurotrophic factor regulates hedonic feeding by acting on the mesolimbic dopamine system. J Neurosci 2010; 30: 2533–2541.
Lobo MK, Covington HE, Chaudhury D, Friedman AK, Sun H, Damez-Werno D et al. Cell type-specific loss of BDNF signaling mimics optogenetic control of cocaine reward. Science 2010; 330: 385–390.
Vialou V, Robison AJ, LaPlant QC, Covington HE, Dietz DM, Ohnishi YN et al. [Delta]FosB in brain reward circuits mediates resilience to stress and antidepressant responses. Nat Neurosci 2010; 13: 745–752.
Acknowledgements
This project was supported by grants to SF from the Natural Sciences and Engineering Research Council of Canada (No.355881), Canadian Diabetes Association (OG-2-09-2835-SF) and Canadian Foundation for Innovation. SS is supported by a postdoctoral fellowship from the CIHR Neuroinflammation Training Program and MFF by a PhD fellowship from the Canadian Diabetes Association.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Sharma, S., Fernandes, M. & Fulton, S. Adaptations in brain reward circuitry underlie palatable food cravings and anxiety induced by high-fat diet withdrawal. Int J Obes 37, 1183–1191 (2013). https://doi.org/10.1038/ijo.2012.197
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2012.197
Keywords
This article is cited by
-
Obese-associated gut microbes and derived phenolic metabolite as mediators of excessive motivation for food reward
Microbiome (2023)
-
Transcriptomics in the nucleus accumbens shell reveal sex- and reinforcer-specific signatures associated with morphine and sucrose craving
Neuropsychopharmacology (2022)
-
Cannabinoid CB1 receptor in dorsal telencephalic glutamatergic neurons drives overconsumption of palatable food and obesity
Neuropsychopharmacology (2021)
-
AMPK in the gut-liver-brain axis and its influence on OP rats in an HSHF intake and WTD rat model
Pflügers Archiv - European Journal of Physiology (2021)
-
Palatable high-fat diet intake influences mnemonic and emotional aspects in female rats in an estrous cycle-dependent manner
Metabolic Brain Disease (2021)