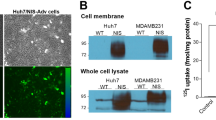
Abstract
In this study, we determined the in vitro and in vivo efficacy of sodium iodide symporter (NIS) gene transfer and the therapeutic potential of oncolytic virotherapy combined with radioiodine therapy using a conditionally replicating oncolytic adenovirus. For this purpose, we used a replication-selective adenovirus in which the E1a gene is driven by the mouse alpha-fetoprotein (AFP) promoter and the human NIS gene is inserted in the E3 region (Ad5-E1/AFP-E3/NIS). Human hepatocellular carcinoma cells (HuH7) infected with Ad5-E1/AFP-E3/NIS concentrated radioiodine at a level that was sufficiently high for a therapeutic effect in vitro. In vivo experiments demonstrated that 3 days after intratumoral (i.t.) injection of Ad5-E1/AFP-E3/NIS HuH7 xenograft tumors accumulated approximately 25% ID g−1 (percentage of the injected dose per gram tumor tissue) 123I as shown by 123I gamma camera imaging. A single i.t. injection of Ad5-E1/AFP-E3/NIS (virotherapy) resulted in a significant reduction of tumor growth and prolonged survival, as compared with injection of saline. Combination of oncolytic virotherapy with radioiodine treatment (radiovirotherapy) led to an additional reduction of tumor growth that resulted in markedly improved survival as compared with virotherapy alone. In conclusion, local in vivo NIS gene transfer using a replication-selective oncolytic adenovirus is able to induce a significant therapeutic effect, which can be enhanced by additional 131I application.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Forner A, Llovet JM, Bruix J . Hepatocellular carcinoma. Lancet 2012; 379: 1245–1255.
Siegel R, Naishadham D, Jemal A . Cancer statistics, 2012. CA Cancer J Clin 2012; 62: 10–29.
Cao H, Phan H, Yang LX . Improved chemotherapy for hepatocellular carcinoma. Anticancer Res 2012; 32: 1379–1386.
Carrasco N . Iodide transport in the thyroid gland. Biochim Biophys Acta 1993; 1154: 65–82.
Jhiang SM, Cho JY, Ryu KY, DeYoung BR, Smanik PA, McGaughy VR et al. An immunohistochemical study of Na+/I- symporter in human thyroid tissues and salivary gland tissues. Endocrinology 1998; 139: 4416–4419.
Hingorani M, Spitzweg C, Vassaux G, Newbold K, Melcher A, Pandha H et al. The biology of the sodium iodide symporter and its potential for targeted gene delivery. Curr Cancer Drug Targets 2010; 10: 242–267.
Spitzweg C, O'Connor MK, Bergert ER, Tindall DJ, Young CY, Morris JC . Treatment of prostate cancer by radioiodine therapy after tissue-specific expression of the sodium iodide symporter. Cancer Res 2000; 60: 6526–6530.
Scholz IV, Cengic N, Baker CH, Harrington KJ, Maletz K, Bergert ER et al. Radioiodine therapy of colon cancer following tissue-specific sodium iodide symporter gene transfer. Gene Therapy 2005; 12: 272–280.
Willhauck MJ, Sharif-Samani B, Senekowitsch-Schmidtke R, Wunderlich N, Goke B, Morris JC et al. Functional sodium iodide symporter expression in breast cancer xenografts in vivo after systemic treatment with retinoic acid and dexamethasone. Breast Cancer Res Treat 2008; 109: 263–272.
Klutz K, Russ V, Willhauck MJ, Wunderlich N, Zach C, Gildehaus FJ et al. Targeted radioiodine therapy of neuroblastoma tumors following systemic nonviral delivery of the sodium iodide symporter gene. Clin Cancer Res 2009; 15: 6079–6086.
Klutz K, Schaffert D, Willhauck MJ, Grunwald GK, Haase R, Wunderlich N et al. Epidermal growth factor receptor-targeted (131)I-therapy of liver cancer following systemic delivery of the sodium iodide symporter gene. Mol Ther 2011; 19: 676–685.
Richard-Fiardo P, Franken PR, Harrington KJ, Vassaux G, Cambien B . The use of molecular imaging of gene expression by radiotracers in gene therapy. Expert Opin Biol Ther 2011; 11: 1273–1285.
Chan DW, Kelsten M, Rock R, Bruzek D . Evaluation of a monoclonal immunoenzymometric assay for alpha-fetoprotein. Clin Chem 1986; 32: 1318–1322.
Johnson PJ . Role of alpha-fetoprotein in the diagnosis and management of hepatocellular carcinoma. J Gastroenterol Hepatol 1999; 14 (Suppl): S32–S36.
Watanabe K, Saito A, Tamaoki T . Cell-specific enhancer activity in a far upstream region of the human alpha-fetoprotein gene. J Biol Chem 1987; 262: 4812–4818.
Klutz K, Willhauck MJ, Wunderlich N, Zach C, Anton M, Senekowitsch-Schmidtke R et al. Sodium iodide symporter (NIS)-mediated radionuclide ((131)I, (188)Re) therapy of liver cancer after transcriptionally targeted intratumoral in vivo NIS gene delivery. Hum Gene Ther 2011; 22: 1403–1412.
Kakinuma H, Bergert ER, Spitzweg C, Cheville JC, Lieber MM, Morris JC . Probasin promoter (ARR(2)PB)-driven, prostate-specific expression of the human sodium iodide symporter (h-NIS) for targeted radioiodine therapy of prostate cancer. Cancer Res 2003; 63: 7840–7844.
Klutz K, Willhauck MJ, Dohmen C, Wunderlich N, Knoop K, Zach C et al. Image-guided tumor-selective radioiodine therapy of liver cancer after systemic nonviral delivery of the sodium iodide symporter gene. Hum Gene Ther 2011; 22: 1563–1574.
Spitzweg C, Baker CH, Bergert ER, O'Connor MK, Morris JC . Image-guided radioiodide therapy of medullary thyroid cancer after carcinoembryonic antigen promoter-targeted sodium iodide symporter gene expression. Hum Gene Ther 2007; 18: 916–924.
Spitzweg C, Dietz AB, O'Connor MK, Bergert ER, Tindall DJ, Young CY et al. In vivo sodium iodide symporter gene therapy of prostate cancer. Gene Therapy 2001; 8: 1524–1531.
Dingli D, Russell SJ, Morris JC . In vivo imaging and tumor therapy with the sodium iodide symporter. J Cell Biochem 2003; 90: 1079–1086.
Spitzweg C, Morris JC . The sodium iodide symporter: its pathophysiological and therapeutic implications. Clin Endocrinol (Oxf) 2002; 57: 559–574.
Spitzweg C, Zhang S, Bergert ER, Castro MR, McIver B, Heufelder AE et al. Prostate-specific antigen (PSA) promoter-driven androgen-inducible expression of sodium iodide symporter in prostate cancer cell lines. Cancer Res 1999; 59: 2136–2141.
Cengic N, Baker CH, Schutz M, Goke B, Morris JC, Spitzweg C . A novel therapeutic strategy for medullary thyroid cancer based on radioiodine therapy following tissue-specific sodium iodide symporter gene expression. J Clin Endocrinol Metab 2005; 90: 4457–4464.
Trujillo MA, Oneal MJ, Davydova J, Bergert E, Yamamoto M, Morris JC . Construction of an MUC-1 promoter driven, conditionally replicating adenovirus that expresses the sodium iodide symporter for gene therapy of breast cancer. Breast Cancer Res 2009; 11: R53.
Trujillo MA, Oneal MJ, McDonough S, Qin R, Morris JC . A probasin promoter, conditionally replicating adenovirus that expresses the sodium iodide symporter (NIS) for radiovirotherapy of prostate cancer. Gene Therapy 2010; 17: 1325–1332.
Willhauck MJ, Sharif Samani BR, Klutz K, Cengic N, Wolf I, Mohr L et al. Alpha-fetoprotein promoter-targeted sodium iodide symporter gene therapy of hepatocellular carcinoma. Gene Therapy 2008; 15: 214–223.
Zhang DE, Rabek JP, Hsieh CC, Torres-Ramos C, Papaconstantinou J . Functional analysis of the mouse alpha-fetoprotein enhancers and their subfragments in primary mouse hepatocyte cultures. J Biol Chem 1992; 267: 10676–10682.
Russell WC . Adenoviruses: update on structure and function. J Gen Virol 2009; 90: 1–20.
Shirakawa T . The current status of adenovirus-based cancer gene therapy. Mol Cells 2008; 25: 462–466.
Toth K, Dhar D, Wold WS . Oncolytic (replication-competent) adenoviruses as anticancer agents. Expert Opin Biol Ther 2010; 10: 353–368.
Kaminsky SM, Levy O, Salvador C, Dai G, Carrasco N . The Na+/I- symporter of the thyroid gland. Soc Gen Physiol Ser 1993; 48: 251–262.
Ma XJ, Huang R, Kuang AR . AFP promoter enhancer increased specific expression of the human sodium iodide symporter (hNIS) for targeted radioiodine therapy of hepatocellular carcinoma. Cancer Invest 2009; 27: 673–681.
Penheiter AR, Wegman TR, Classic KL, Dingli D, Bender CE, Russell SJ et al. Sodium iodide symporter (NIS)-mediated radiovirotherapy for pancreatic cancer. AJR Am J Roentgenol 2010; 195: 341–349.
Msaouel P, Iankov ID, Allen C, Aderca I, Federspiel MJ, Tindall DJ et al. Noninvasive imaging and radiovirotherapy of prostate cancer using an oncolytic measles virus expressing the sodium iodide symporter. Mol Ther 2009; 17: 2041–2048.
Peerlinck I, Merron A, Baril P, Conchon S, Martin-Duque P, Hindorf C et al. Targeted radionuclide therapy using a Wnt-targeted replicating adenovirus encoding the Na/I symporter. Clin Cancer Res 2009; 15: 6595–6601.
Merron A, Peerlinck I, Martin-Duque P, Burnet J, Quintanilla M, Mather S et al. SPECT/CT imaging of oncolytic adenovirus propagation in tumours in vivo using the Na/I symporter as a reporter gene. Gene Therapy 2007; 14: 1731–1738.
Singh R, Kostarelos K . Designer adenoviruses for nanomedicine and nanodiagnostics. Trends Biotechnol 2009; 27: 220–229.
Hart LS, Yannone SM, Naczki C, Orlando JS, Waters SB, Akman SA et al. The adenovirus E4orf6 protein inhibits DNA double strand break repair and radiosensitizes human tumor cells in an E1B-55K-independent manner. J Biol Chem 2005; 280: 1474–1481.
Hart LS, Ornelles D, Koumenis C . The adenoviral E4orf6 protein induces atypical apoptosis in response to DNA damage. J Biol Chem 2007; 282: 6061–6067.
Advani SJ, Mezhir JJ, Roizman B, Weichselbaum RR . ReVOLT: radiation-enhanced viral oncolytic therapy. Int J Radiat Oncol Biol Phys 2006; 66: 637–646.
Hingorani M, White CL, Merron A, Peerlinck I, Gore ME, Slade A et al. Inhibition of repair of radiation-induced DNA damage enhances gene expression from replication-defective adenoviral vectors. Cancer Res 2008; 68: 9771–9778.
Hingorani M, White CL, Zaidi S, Merron A, Peerlinck I, Gore ME et al. Radiation-mediated up-regulation of gene expression from replication-defective adenoviral vectors: implications for sodium iodide symporter gene therapy. Clin Cancer Res 2008; 14: 4915–4924.
Kumar S, Gao L, Yeagy B, Reid T . Virus combinations and chemotherapy for the treatment of human cancers. Curr Opin Mol Ther 2008; 10: 371–379.
Dingli D, Diaz RM, Bergert ER, O'Connor MK, Morris JC, Russell SJ . Genetically targeted radiotherapy for multiple myeloma. Blood 2003; 102: 489–496.
Goel A, Carlson SK, Classic KL, Greiner S, Naik S, Power AT et al. Radioiodide imaging and radiovirotherapy of multiple myeloma using VSV(Delta51)-NIS, an attenuated vesicular stomatitis virus encoding the sodium iodide symporter gene. Blood 2007; 110: 2342–2350.
Knoop K, Kolokythas M, Klutz K, Willhauck MJ, Wunderlich N, Draganovici D et al. Image-guided, tumor stroma-targeted 131I therapy of hepatocellular cancer after systemic mesenchymal stem cell-mediated NIS gene delivery. Mol Ther 2011; 19: 1704–1713.
Willhauck MJ, Sharif Samani BR, Gildehaus FJ, Wolf I, Senekowitsch-Schmidtke R, Stark HJ et al. Application of 188rhenium as an alternative radionuclide for treatment of prostate cancer after tumor-specific sodium iodide symporter gene expression. J Clin Endocrinol Metab 2007; 92: 4451–4458.
Maxon HR, Thomas SR, Samaratunga RC . Dosimetric considerations in the radioiodine treatment of macrometastases and micrometastases from differentiated thyroid cancer. Thyroid 1997; 7: 183–187.
Chiesa C, Castellani MR, Vellani C, Orunesu E, Negri A, Azzeroni R et al. Individualized dosimetry in the management of metastatic differentiated thyroid cancer. Q J Nucl Med Mol Imaging 2009; 53: 546–561.
Lassmann M, Reiners C, Luster M . Dosimetry and thyroid cancer: the individual dosage of radioiodine. Endocr Relat Cancer 2010; 17: R161–R172.
Barton KN, Stricker H, Elshaikh MA, Pegg J, Cheng J, Zhang Y et al. Feasibility of adenovirus-mediated hNIS gene transfer and 131I radioiodine therapy as a definitive treatment for localized prostate cancer. Mol Ther 2011; 19: 1353–1359.
Woller N, Knocke S, Mundt B, Gurlevik E, Struver N, Kloos A et al. Virus-induced tumor inflammation facilitates effective DC cancer immunotherapy in a Treg-dependent manner in mice. J Clin Invest 2011; 121: 2570–2582.
Weiss SJ, Philp NJ, Grollman EF . Iodide transport in a continuous line of cultured cells from rat thyroid. Endocrinology 1984; 114: 1090–1098.
Unterholzner S, Willhauck MJ, Cengic N, Schutz M, Goke B, Morris JC et al. Dexamethasone stimulation of retinoic acid-induced sodium iodide symporter expression and cytotoxicity of 131-I in breast cancer cells. J Clin Endocrinol Metab 2006; 91: 69–78.
Acknowledgements
We are grateful to SM Jhiang, Ohio State University, Columbus, OH, USA, for supplying the full-length human NIS complementary DNA; to M Geissler, Esslingen, Germany, for supplying the murine AFP promoter/enhancer fragment; and to JC Morris, Mayo Clinic, Rochester, MN, USA, for providing the NIS mouse monoclonal antibody. We also thank R Anderson, Viraquest, for the synthesis of the Ad5-E1/AFP-E3/NIS construct; K Mantwill, Technical University Munich, Germany for providing the qPCR primers and protocol as well as D Mayr, Ludwig-Maximilians University, Munich, Germany, for preparation of paraffin-embedded slides. This study was supported by grant SFB 824 (Sonderforschungsbereich 824) from the Deutsche Forschungsgemeinschaft, Bonn, Germany, and by a grant from the Wilhelm-Sander-Stiftung (2008.037.1) to C Spitzweg.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Grünwald, G., Klutz, K., Willhauck, M. et al. Sodium iodide symporter (NIS)-mediated radiovirotherapy of hepatocellular cancer using a conditionally replicating adenovirus. Gene Ther 20, 625–633 (2013). https://doi.org/10.1038/gt.2012.79
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2012.79
Keywords
This article is cited by
-
Dual-targeted NIS polyplexes—a theranostic strategy toward tumors with heterogeneous receptor expression
Gene Therapy (2019)
-
Sequence-defined cMET/HGFR-targeted Polymers as Gene Delivery Vehicles for the Theranostic Sodium Iodide Symporter (NIS) Gene
Molecular Therapy (2016)
-
Small activating RNA upregulates NIS expression: promising potential for hepatocellular carcinoma endoradiotherapy
Cancer Gene Therapy (2016)
-
Application of conditionally replicating adenoviruses in tumor early diagnosis technology, gene-radiation therapy and chemotherapy
Applied Microbiology and Biotechnology (2016)
-
A novel placental like alkaline phosphate promoter driven transcriptional silencing combined with single chain variable fragment antibody based virosomal delivery for neoplastic cell targeting
Journal of Translational Medicine (2015)