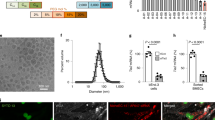
Abstract
It is well established that specific interaction between adhesion molecules of endothelial cells and receptors on leukocytes can separate and recruit leukocytes from the bloodstream to sites of inflammation and coagulation. Previously, we showed that P-selectin can be absorbed onto the surface of a blood-compatible microrenathane tube, and the P-selectin-coated surface could successfully capture P-selectin receptor-positive stem cells from physiological shear flow in vitro and from the bloodstream in vivo. In this paper, P-selectin was covalently attached to the surface of nanoscale liposomes to create targeting nanoparticles (NPs). Small interfering RNA (siRNA) was encapsulated by these nanoscale liposomes, and the liposomes were stabilized by PEGylation with DSPE-PEG2000. Experiments showed that these P-selectin-, PEGylated-, nanoscale-liposomes (PS-DSPE-PEG NPs) could be absorbed onto the inner surface of microrenathane tubing. The coated surface could specifically capture targeted cells from physiological shear flow, efficiently deliver encapsulated siRNA into adherent cells and dramatically silence the targeted gene neutrophil elastase. With this device, we create a high localized concentration for siRNA delivery in the circulatory system, providing circulating target cells adequate time to interact with therapeutic materials. SiRNA is efficaciously delivered into specific target cells, thereby providing a powerful tool for highly efficient siRNA transfection and other therapeutic materials delivery in circulation. The method should prove especially useful for diseases derived from disorders of blood cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Dorsett Y, Tuschl T . siRNAs: applications in functional genomics and potential as therapeutics. Nat Rev Drug Discov 2004; 3: 318–329.
Chapman EJ, Carrington JC . Specialization and evolution of endogenous small RNA pathways. Nat Rev Genet 2007; 8: 884–896.
McManus MT, Sharp PA . Gene silencing in mammals by small interfering RNAs. Nat Rev Genet 2002; 3: 737–747.
Grunweller A, Hartmann RK . RNA interference as a gene-specific approach for molecular medicine. Curr Med Chem 2005; 12: 3143–3161.
Campbell TN, Choy FY . RNA interference: past, present and future. Curr Issues Mol Biol 2005; 7: 1–6.
Davidson BL, Boudreau RL . RNA interference: a tool for querying nervous system function and an emerging therapy. Neuron 2007; 53: 781–788.
Kim DH, Rossi JJ . Strategies for silencing human disease using RNA interference. Nat Rev Genet 2007; 8: 173–184.
Mittal V . Improving the efficiency of RNA interference in mammals. Nat Rev Genet 2004; 5: 355–365.
Peer D, Park EJ, Morishita Y, Carman CV, Shimaoka M . Systemic leukocyte-directed siRNA delivery revealing cyclin D1 as an anti-inflammatory target. Science 2008; 319: 627–630.
Samad A, Sultana Y, Aqil M . Liposomal drug delivery systems: an update review. Curr Drug Deliv 2007; 4: 297–305.
Immordino ML, Dosio F, Cattel L . Stealth liposomes: review of the basic science, rationale, and clinical applications, existing and potential. Int J Nanomedicine 2006; 1: 297–315.
Discher BM, Won YY, Ege DS, Lee JC, Bates FS, Discher DE et al. Polymersomes: tough vesicles made from diblock copolymers. Science 1999; 284: 1143–1146.
Moghimi SM, Szebeni J . Stealth liposomes and long circulating nanoparticles: critical issues in pharmacokinetics, opsonization and protein-binding properties. Prog Lipid Res 2003; 42: 463–478.
Torchilin VP . Recent advances with liposomes as pharmaceutical carriers. Nat Rev Drug Discov 2005; 4: 145–160.
Papahadjopoulos D, Allen TM, Gabizon A, Mayhew E, Matthay K, Huang SK et al. Sterically stabilized liposomes: improvements in pharmacokinetics and antitumor therapeutic efficacy. Proc Natl Acad Sci USA 1991; 88: 11460–11464.
Kreuter J, Higuchi T . Improved delivery of methoxsalen. J Pharm Sci 1979; 68: 451–454.
Li SD, Huang L . Targeted delivery of antisense oligodeoxynucleotide and small interference RNA into lung cancer cells. Mol Pharm 2006; 3: 579–588.
Narasipura SD, Wojciechowski JC, Duffy BM, Liesveld JL, King MR . Purification of CD45+ hematopoietic cells directly from human bone marrow using a flow-based P-selectin-coated microtube. Am J Hematol 2008; 83: 627–629.
Wojciechowski JC, Narasipura SD, Charles N, Mickelsen D, Rana K, Blair ML et al. Capture and enrichment of CD34-positive haematopoietic stem and progenitor cells from blood circulation using P-selectin in an implantable device. Br J Haematol 2008; 140: 673–681.
Hrachovinova I, Cambien B, Hafezi-Moghadam A, Kappelmayer J, Camphausen RT, Widom A et al. Interaction of P-selectin and PSGL-1 generates microparticles that correct hemostasis in a mouse model of hemophilia A. Nat Med 2003; 9: 1020–1025.
Tomita H, Iwata Y, Ogawa F, Komura K, Shimizu K, Yoshizaki A et al. P-selectin glycoprotein ligand-1 contributes to wound healing predominantly as a P-selectin ligand and partly as an E-selectin ligand. J Invest Dermatol 2009 (e-pub ahead of print 29 January 2009; doi:10.1038/jid.2008.446).
Moore KL, Patel KD, Bruehl RE, Li F, Johnson DA, Lichenstein HS et al. P-selectin glycoprotein ligand-1 mediates rolling of human neutrophils on P-selectin. J Cell Biol 1995; 128: 661–671.
Marathe DD, Chandrasekaran EV, Lau JT, Matta KL, Neelamegham S . Systems-level studies of glycosyltransferase gene expression and enzyme activity that are associated with the selectin binding function of human leukocytes. FASEB J 2008; 22: 4154–4167.
Moore KL, Varki A, McEver RP . GMP-140 binds to a glycoprotein receptor on human neutrophils: evidence for a lectin-like interaction. J Cell Biol 1991; 112: 491–499.
Narasipura SD, Wojciechowski JC, Charles N, Liesveld JL, King MR . P-Selectin coated microtube for enrichment of CD34+ hematopoietic stem and progenitor cells from human bone marrow. Clin Chem 2008; 54: 77–85.
Moore KL, Stults NL, Diaz S, Smith DF, Cummings RD, Varki A et al. Identification of a specific glycoprotein ligand for P-selectin (CD62) on myeloid cells. J Cell Biol 1992; 118: 445–456.
Aigner S, Ramos CL, Hafezi-Moghadam A, Lawrence MB, Friederichs J, Altevogt P et al. CD24 mediates rolling of breast carcinoma cells on P-selectin. FASEB J 1998; 12: 1241–1251.
Collins SJ, Ruscetti FW, Gallagher RE, Gallo RC . Terminal differentiation of human promyelocytic leukemia cells induced by dimethyl sulfoxide and other polar compounds. Proc Natl Acad Sci USA 1978; 75: 2458–2462.
Greenberg AW, Hammer DA . Cell separation mediated by differential rolling adhesion. Biotechnol Bioeng 2001; 73: 111–124.
Charles N, Liesveld JL, King MR . Investigating the feasibility of stem cell enrichment mediated by immobilized selectins. Biotechnol Prog 2007; 23: 1463–1472.
Ma YQ, Plow EF, Geng JG . P-selectin binding to P-selectin glycoprotein ligand-1 induces an intermediate state of alphaMbeta2 activation and acts cooperatively with extracellular stimuli to support maximal adhesion of human neutrophils. Blood 2004; 104: 2549–2556.
Mazella J, Vincent JP . Internalization and recycling properties of neurotensin receptors. Peptides 2006; 27: 2488–2492.
Brigger I, Dubernet C, Couvreur P . Nanoparticles in cancer therapy and diagnosis. Adv Drug Deliv Rev 2002; 54: 631–651.
Maruyama K . [PEG-liposome in DDS and clinical studies]. Nippon Rinsho 1998; 56: 632–637.
Sasaki K, Ikeda K, Nagai M, Shima H, Nagao M, Takahara J et al. Protein phosphatase 2A alpha involvement in granulocytic differentiation of HL-60 cells. Hematol Oncol 1993; 11: 1–5.
Schumacher I, Margalit R . Liposome-encapsulated ampicillin: physicochemical and antibacterial properties. J Pharm Sci 1997; 86: 635–641.
Huang Z, Nie L, Xu M, Sun XH . Notch-induced E2A degradation requires CHIP and Hsc70 as novel facilitators of ubiquitination. Mol Cell Biol 2004; 24: 8951–8962.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, Z., King, M. An immobilized nanoparticle-based platform for efficient gene knockdown of targeted cells in the circulation. Gene Ther 16, 1271–1282 (2009). https://doi.org/10.1038/gt.2009.76
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2009.76
Keywords
This article is cited by
-
Nanotechnology for the detection and kill of circulating tumor cells
Nanoscale Research Letters (2014)
-
Glycomechanics of the Metastatic Cascade: Tumor Cell–Endothelial Cell Interactions in the Circulation
Annals of Biomedical Engineering (2012)