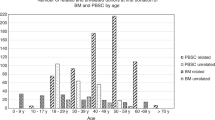
Abstract
Donation of haematopoietic stem cells, either through BM or PBSC collection, is a generally safe procedure for healthy donors although adverse reactions are a definite risk. The invaluable source of donation and its central role in transplantation implies that every effort should be made to alleviate possible difficulties the donor encounters. The physical and psychological reactions to donation have been established for some time, but less is known about the factors that are associated with a poorer donation experience. In this article, we provide an overview of the physical and psychological donation experience and focus attention on demographic, physical and psychological factors that may influence this donation experience. Understanding that toxicity profiles vary with certain donor characteristics is crucial as this knowledge could influence practice in numerous ways including the modification of joining and recruitment policies and the improvement of supportive measures and donor follow-up procedures. Although this review deals with both unrelated and related donors (RDs), there is a relative paucity of regulation of RD care and we call for more attention to this area. Owing to the relative rarity of donation in each country, a global effort to collect donor outcome data is needed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gratwohl A, Baldomero H, Aljurf M, Pasquini MC, Bouzas LF, Yoshimi A et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 2010; 303: 1617–1624.
Baldomero H, Gratwohl M, Gratwohl A, Tichelli A, Niederwieser D, Madrigal A et al. The EBMT activity survey 2009: trends over the past 5 years. Bone Marrow Transplant 2011; 46: 485–501.
Pasquini MC, Wang Z Current use and outcome of hematopoietic stem cell transplantation: CIBMTR Summary Slides. Available at http://www.cibmtr.org2012.
Boo M, van Walraven SM, Chapman J, Lindberg B, Schmidt AH, Shaw BE et al. Remuneration of hematopoietic stem cell donors: principles and perspective of the World Marrow Donor Association. Blood 2011; 117: 21–25.
Karlsson L, Quinlan D, Guo D, Brown C, Selinger S, Klassen J et al. Mobilized blood cells vs bone marrow harvest: experience compared in 171 donors with particular reference to pain and fatigue. Bone Marrow Transplant 2004; 33: 709–713.
de la Rubia J, de Arriba F, Arbona C, Pascual MJ, Zamora C, Insunza A et al. Follow-up of healthy donors receiving granulocyte colony-stimulating factor for peripheral blood progenitor cell mobilization and collection. Results of the Spanish Donor Registry. Haematologica 2008; 93: 735–740.
Pulsipher MA, Chitphakdithai P, Logan BR, Shaw BE, Wingard JR, Lazarus HM et al. Acute toxicities of unrelated bone marrow versus peripheral blood stem cell donation: results of a prospective trial from the National Marrow Donor Program. Blood 2013; 121: 197–206.
Pulsipher MA, Chitphakdithai P, Miller JP, Logan BR, King RJ, Rizzo JD et al. Adverse events among 2408 unrelated donors of peripheral blood stem cells: results of a prospective trial from the National Marrow Donor Program. Blood 2009; 113: 3604–3611.
Murata M, Harada M, Kato S, Takahashi S, Ogawa H, Okamoto S et al. Peripheral blood stem cell mobilization and apheresis: analysis of adverse events in 94 normal donors. Bone Marrow Transplant 1999; 24: 1065–1071.
Chen SH, Yang SH, Chu SC, Su YC, Chang CY, Chiu YW et al. The role of donor characteristics and post-granulocyte colony-stimulating factor white blood cell counts in predicting the adverse events and yields of stem cell mobilization. Int J Hematol 2011; 93: 652–659.
Miller JP, Perry EH, Price TH, Bolan CD Jr, Karanes C, Boyd TM et al. Recovery and safety profiles of marrow and PBSC donors: experience of the National Marrow Donor Program. Biol Blood Marrow Transplant 2008; 14 (9 Suppl): 29–36.
Holig K, Kramer M, Kroschinsky F, Bornhauser M, Mengling T, Schmidt AH et al. Safety and efficacy of hematopoietic stem cell collection from mobilized peripheral blood in unrelated volunteers: 12 years of single-center experience in 3928 donors. Blood 2009; 114: 3757–3763.
Fortanier C, Kuentz M, Sutton L, Milpied N, Michalet M, Macquart-Moulin G et al. Healthy sibling donor anxiety and pain during bone marrow or peripheral blood stem cell harvesting for allogeneic transplantation: results of a randomised study. Bone Marrow Transplant 2002; 29: 145–149.
Martino M, Console G, Dattola A, Callea I, Messina G, Moscato T et al. Short and long-term safety of lenograstim administration in healthy peripheral haematopoietic progenitor cell donors: a single centre experience. Bone Marrow Transplant 2009; 44: 163–168.
Pamphilon D, Nacheva E, Navarrete C, Madrigal A, Goldman J . The use of granulocyte-colony-stimulating factor in volunteer unrelated hemopoietic stem cell donors. Transfusion 2008; 48: 1495–1501.
Hirsch B, Oseth L, Cain M, Trader E, Pulkrabek S, Lindgren B et al. Effects of granulocyte-colony stimulating factor on chromosome aneuploidy and replication asynchrony in healthy peripheral blood stem cell donors. Blood 2011; 118: 2602–2608.
Gandini A, Roata C, Franchini M, Agostini E, Guizzardi E, Pontiero Giacometti P et al. Unrelated allogeneic bone marrow donation: short- and long-term follow-up of 103 consecutive volunteer donors. Bone Marrow Transplant 2001; 28: 369–374.
Machaczka M, Kalaitzakis E, Eleborg L, Ljungman P, Hagglund H . Comparison of general vs regional anaesthesia for BM harvesting: a retrospective study of anaesthesia-related complications. Bone Marrow Transplant 2010; 45: 53–61.
Nishimori M, Yamada Y, Hoshi K, Akiyama Y, Hoshi Y, Morishima Y et al. Health-related quality of life of unrelated bone marrow donors in Japan. Blood 2002; 99: 1995–2001.
Boulton FE, James V . Guidelines for policies on alternatives to allogeneic blood transfusion. 1. Predeposit autologous blood donation and transfusion. Transfus Med 2007; 17: 354–365.
Favre G, Beksac M, Bacigalupo A, Ruutu T, Nagler A, Gluckman E et al. Differences between graft product and donor side effects following bone marrow or stem cell donation. Bone Marrow Transplant 2003; 32: 873–880.
Stroncek DF, Holland PV, Bartch G, Bixby T, Simmons RG, Antin JH et al. Experiences of the first 493 unrelated marrow donors in the National Marrow Donor Program. Blood 1993; 81: 1940–1946.
Rowley SD, Donaldson G, Lilleby K, Bensinger WI, Appelbaum FR . Experiences of donors enrolled in a randomized study of allogeneic bone marrow or peripheral blood stem cell transplantation. Blood 2001; 97: 2541–2548.
Heldal D, Brinch L, Tjonnfjord G, Solheim BG, Egeland T, Gadeholt G et al. Donation of stem cells from blood or bone marrow: results of a randomised study of safety and complaints. Bone Marrow Transplant 2002; 29: 479–486.
Bredeson C, Leger C, Couban S, Simpson D, Huebsch L, Walker I et al. An evaluation of the donor experience in the canadian multicenter randomized trial of bone marrow versus peripheral blood allografting. Biol Blood Marrow Transplant 2004; 10: 405–414.
Siddiq S, Pamphilon D, Brunskill S, Doree C, Hyde C, Stanworth S . Bone marrow harvest versus peripheral stem cell collection for haemopoietic stem cell donation in healthy donors. Cochrane Database Syst Rev 2009; 21: Cd006406.
Pulsipher M Increased risk of severe adverse events after BM vs PBSC donation: Results of a prospective study of 9494 NMDP donors (2004-2009). http://www.worldmarrow.org/fileadmin/Meeting/IDRC_Presentations/2012-IDRC-Sydney/20120504-Pulsipher.pdf2012.
Halter J, Kodera Y, Ispizua AU, Greinix HT, Schmitz N, Favre G et al. Severe events in donors after allogeneic hematopoietic stem cell donation. Haematologica 2009; 94: 94–101.
Halter JP, van Walraven SM, Worel N, Bengtsson M, Hagglund H, Nicoloso de Faveri G et al. Allogeneic hematopoietic stem cell donation-standardized assessment of donor outcome data: a consensus statement from the Worldwide Network for Blood and Marrow Transplantation (WBMT). Bone Marrow Transplant 2013; 48: 220–225.
Anderlini P, Champlin RE . Biologic and molecular effects of granulocyte colony-stimulating factor in healthy individuals: recent findings and current challenges. Blood 2008; 111: 1767–1772.
D'Souza A, Jaiyesimi I, Trainor L, Venuturumili P . Granulocyte colony-stimulating factor administration: adverse events. Transfus Med Rev 2008; 22: 280–290.
McCullough J, Kahn J, Adamson J, Anderlini P, Benjamin R, Confer D et al. Hematopoietic growth factors—use in normal blood and stem cell donors: clinical and ethical issues. Transfusion 2008; 48: 2008–2025.
Kennedy GA, Morton J, Western R, Butler J, Daly J, Durrant S . Impact of stem cell donation modality on normal donor quality of life: a prospective randomized study. Bone Marrow Transplant 2003; 31: 1033–1035.
Confer DL, Shaw BE, Pamphilon DH . WMDA guidelines for subsequent donations following initial BM or PBSCs. Bone Marrow Transplant 2011; 46: 1409–1412.
Leitner GC, Baumgartner K, Kalhs P, Biener D, Greinix HT, Hoecker P et al. Regeneration, health status and quality of life after rhG-CSF-stimulated stem cell collection in healthy donors: a cross-sectional study. Bone Marrow Transplant 2009; 43: 357–363.
Wiersum-Osselton JC, van Walraven SM, Bank I, Lenselink AM, Fibbe WE, van der Bom JG et al. Clinical outcomes after peripheral blood stem cell donation by related donors: a Dutch single-center cohort study. Transfusion 2013; 53: 96–103.
Anderlini P, Rizzo JD, Nugent ML, Schmitz N, Champlin RE, Horowitz MM . Peripheral blood stem cell donation: an analysis from the International Bone Marrow Transplant Registry (IBMTR) and European Group for Blood and Marrow Transplant (EBMT) databases. Bone Marrow Transplant 2001; 27: 689–692.
O'Donnell PV, Pedersen TL, Confer DL, Rizzo JD, Pulsipher MA, Stroncek D et al. Practice patterns for evaluation, consent, and care of related donors and recipients at hematopoietic cell transplantation centers in the United States. Blood 2010; 115: 5097–5101.
Shaw BE . First do no harm. Blood 2010; 115: 4978–4979.
van Walraven SM, Nicoloso-de Faveri G, Axdorph-Nygell UA, Douglas KW, Jones DA, Lee SJ et al. Family donor care management: principles and recommendations. Bone Marrow Transplant 2010; 45: 1269–1273.
Horowitz MM, Confer DL . Evaluation of hematopoietic stem cell donors. Hematol Am Soc Hematol Educ Program 2005; 1: 469–475.
Human Tissue Authority EU Tissue and Cells Directive. Available at http://www.hta.gov.uk/legislationpoliciesandcodesofpractice/legislation/eutissueandcellsdirectives.cfm2007.
Chang G, McGarigle C, Spitzer TR, McAfee SL, Harris F, Piercy K et al. A comparison of related and unrelated marrow donors. Psychosom. Med 1998; 60: 163–167.
Pulsipher MA, Logan BR, shan Y RDSafe: a multi-institutional study of hematopoietic cell transplantation donor safety and quality of life http://www.cibmtr.org/Studies2010.
Petersdorf EW . The World Marrow Donor Association: 20 years of international collaboration for the support of unrelated donor and cord blood hematopoietic cell transplantation. Bone Marrow Transplant 2010; 45: 807–810.
Shaw BE, Ball L, Beksac M, Bengtsson M, Confer D, Diler S et al. Donor safety: the role of the WMDA in ensuring the safety of volunteer unrelated donors: clinical and ethical considerations. Bone Marrow Transplant 2010; 45: 832–838.
Lown RN, Shaw BE . 'First do no harm': where do we stand on unrelated hematopoietic cell donor safety? Expert Rev Hematol 2012; 5: 249–252.
King RJ, Confer DL, Greinix HT, Halter J, Horowitz M, Schmidt AH et al. Unrelated hematopoietic stem cell donors as research subjects. Bone Marrow Transplant 2011; 46: 10–13.
Switzer GE, Simmons RG, Dew MA . Helping unrelated strangers: physical and psychological reactions to the bone marrow donation process among anonymous donors1. J Appl Soc Psychol 1996; 26: 469–490.
Yuan S, Ziman A, Smeltzer B, Lu Q, Goldfinger D . Moderate and severe adverse events associated with apheresis donations: incidences and risk factors. Transfusion 2010; 50: 478–486.
Stroncek DF, Clay ME, Petzoldt ML, Smith J, Jaszcz W, Oldham FB et al. Treatment of normal individuals with granulocyte-colony-stimulating factor: donor experiences and the effects on peripheral blood CD34+ cell counts and on the collection of peripheral blood stem cells. Transfusion 1996; 36: 601–610.
Ruau D, Liu LY, Clark JD, Angst MS, Butte AJ . Sex differences in reported pain across 11 000 patients captured in electronic medical records. J Pain 2012; 13: 228–234.
Taenzer AH, Clark C, Curry CS . Gender affects report of pain and function after arthroscopic anterior cruciate ligament reconstruction. Anesthesiology 2000; 93: 670–675.
Rademaker M . Do women have more adverse drug reactions? Am J Clin Dermatol 2001; 2: 349–351.
Eder AF, Hillyer CD, Dy BA, Notari EPt, Benjamin RJ . Adverse reactions to allogeneic whole blood donation by 16- and 17-year-olds. JAMA 2008; 299: 2279–2286.
Wiltbank TB, Giordano GF, Kamel H, Tomasulo P, Custer B . Faint and prefaint reactions in whole-blood donors: an analysis of predonation measurements and their predictive value. Transfusion 2008; 48: 1799–1808.
Amrein K, Valentin A, Lanzer G, Drexler C . Adverse events and safety issues in blood donation-a comprehensive review. Blood Rev 2012; 26: 33–42.
Stroncek D, Shawker T, Follmann D, Leitman SF . G-CSF-induced spleen size changes in peripheral blood progenitor cell donors. Transfusion 2003; 43: 609–613.
Platzbecker U, Prange-Krex G, Bornhauser M, Koch R, Soucek S, Aikele P et al. Spleen enlargement in healthy donors during G-CSF mobilization of PBPCs. Transfusion 2001; 41: 184–189.
Spitzer TR, Areman EM, Cirenza E, Yu M, Dickerson S, Kotula PL et al. The impact of harvest center on quality of marrows collected from unrelated donors. J Hematother 1994; 3: 65–70.
Lavi A, Efrat R, Feigin E, Kadari A . Regional versus general anesthesia for bone marrow harvesting. J Clin Anesth 1993; 5: 204–206.
Knudsen LM, Johnsen HE, Gaarsdal E, Jensen L . Spinal versus general anaesthesia for bone marrow harvesting. Bone Marrow Transplant 1995; 15: 486–487.
Burmeister MA, Standl T, Brauer P, Ramsperger K, Kroger N, Zander A et al. Safety and efficacy of spinal vs general anaesthesia in bone marrow harvesting. Bone Marrow Transplant 1998; 21: 1145–1148.
Bosi A, Bartolozzi B . Safety of bone marrow stem cell donation: a review. Transplant Proc 2010; 42: 2192–2194.
Butterworth VA, Simmons RG, Bartsch G, Randall B, Schimmel M, Stroncek DF . Psychosocial effects of unrelated bone marrow donation: experiences of the National Marrow Donor Program. Blood 1993; 81: 1947–1959.
Christopher KA . The experience of donating bone marrow to a relative. Oncol Nurs Forum 2000; 27: 693–700.
Simmons RG, Schimmel M, Butterworth VA . The self-image of unrelated bone marrow donors. J Health Soc Behav 1993; 34: 285–301.
Switzer GE, Goycoolea JM, Dew MA, Graeff EC, Hegland J . Donating stimulated peripheral blood stem cells vs bone marrow: do donors experience the procedures differently? Bone Marrow Transplant 2001; 27: 917–923.
Butterworth VA, Simmons RG, Schimmel M . When altruism fails: reactions of unrelated bone marrow donors when the recipient dies. Omega 1992; 26: 161–173.
Chang G, McGarigle C, Koby D, Antin JH . Symptoms of pain and depression in related marrow donors: changes after transplant. Psychosomatics 2003; 44: 59–64.
Switzer GE, Myaskovsky L, Goycoolea JM, Dew MA, Confer DL, King R . Factors associated with ambivalence about bone marrow donation among newly recruited unrelated potential donors. Transplantation 2003; 75: 1517–1523.
Switzer GE, Dew MA, Butterworth VA, Simmons RG, Schimmel M . Understanding donors' motivations: a study of unrelated bone marrow donors. Soc Sci Med 1997; 45: 137–147.
Switzer GE, Dew MA, Harrington DJ, Crowley-Matoka M, Myaskovsky L, Abress L et al. Ethnic differences in donation-related characteristics among potential hematopoietic stem cell donors. Transplantation 2005; 80: 890–896.
Pillay B, Lee SJ, Katona L, De Bono S, Warren N, Fletcher J et al. The psychosocial impact of haematopoietic SCT on sibling donors. Bone Marrow Transplant 2012; 47: 1361–1365.
Ings SJ, Balsa C, Leverett D, Mackinnon S, Linch DC, Watts MJ . Peripheral blood stem cell yield in 400 normal donors mobilised with granulocyte colony-stimulating factor (G-CSF): impact of age, sex, donor weight and type of G-CSF used. Br J Haematol 2006; 134: 517–525.
Vasu S, Leitman SF, Tisdale JF, Hsieh MM, Childs RW, Barrett AJ et al. Donor demographic and laboratory predictors of allogeneic peripheral blood stem cell mobilization in an ethnically diverse population. Blood 2008; 112: 2092–2100.
Platzbecker U, Bornhauser M, Zimmer K, Lerche L, Rutt C, Ehninger G et al. Second donation of granulocyte-colony-stimulating factor-mobilized peripheral blood progenitor cells: risk factors associated with a low yield of CD34+ cells. Transfusion 2005; 45: 11–15.
Miflin G, Charley C, Stainer C, Anderson S, Hunter A, Russell N . Stem cell mobilization in normal donors for allogeneic transplantation: analysis of safety and factors affecting efficacy. Br J Haematol 1996; 95: 345–348.
Spierings E, Kim YH, Hendriks M, Borst E, Sergeant R, Canossi A et al. Multicenter analyses demonstrate significant clinical effects of minor histocompatibility antigens on GvHD and GvL after HLA-matched related and unrelated hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013; 19: 1244–1253.
Kroger N, Zabelina T, de Wreede L, Berger J, Alchalby H, van Biezen A et al. Allogeneic stem cell transplantation for older advanced MDS patients: improved survival with young unrelated donor in comparison with HLA-identical siblings. Leukemia 2013; 27: 604–609.
Ayuk F, Zabelina T, Wortmann F, Alchalby H, Wolschke C, Lellek H et al. Donor choice according to age for allo-SCT for AML in complete remission. Bone Marrow Transplant 2013; 48: 1028–1032.
Kollman C, Howe CW, Anasetti C, Antin JH, Davies SM, Filipovich AH et al. Donor characteristics as risk factors in recipients after transplantation of bone marrow from unrelated donors: the effect of donor age. Blood 2001; 98: 2043–2051.
Anderlini P, Przepiorka D, Seong C, Smith TL, Huh YO, Lauppe J et al. Factors affecting mobilization of CD34+ cells in normal donors treated with filgrastim. Transfusion 1997; 37: 507–512.
Martino M, Callea I, Condemi A, Dattola A, Irrera G, Marcuccio D et al. Predictive factors that affect the mobilization of CD34(+) cells in healthy donors treated with recombinant granulocyte colony-stimulating factor (G-CSF). J Clin Apher 2006; 21: 169–175.
Chern B, McCarthy N, Hutchins C, Durrant ST . Analgesic infiltration at the site of bone marrow harvest significantly reduces donor morbidity. Bone Marrow Transplant 1999; 23: 947–949.
Pamphilon D, Siddiq S, Brunskill S, Doree C, Hyde C, Horowitz M et al. Stem cell donation-what advice can be given to the donor? Br J Haematol 2009; 147: 71–76.
Kodera Y, Yamamoto K, Harada M, Morishima Y, Dohy H, Asano S et al. PBSC collection from family donors in Japan: a prospective survey. Bone Marrow Transplant 2014; 49: 195–200.
Engelhardt M, Bertz H, Afting M, Waller CF, Finke J . High-versus standard-dose filgrastim (rhG-CSF) for mobilization of peripheral-blood progenitor cells from allogeneic donors and CD34(+) immunoselection. J Clin Oncol 1999; 17: 2160–2172.
Wolcott DL, Wellisch DK, Fawzy FI, Landsverk J . Psychological adjustment of adult bone marrow transplant donors whose recipient survives. Transplantation 1986; 41: 484–488.
Switzer GE, Dew MA, Magistro CA, Goycoolea JM, Twillman RK, Alter C et al. The effects of bereavement on adult sibling bone marrow donors' psychological well-being and reactions to donation. Bone Marrow Transplant 1998; 21: 181–188.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Billen, A., Madrigal, J. & Shaw, B. A review of the haematopoietic stem cell donation experience: is there room for improvement?. Bone Marrow Transplant 49, 729–736 (2014). https://doi.org/10.1038/bmt.2013.227
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2013.227
Keywords
This article is cited by
-
Basic characteristics and safety of donation in related and unrelated haematopoietic progenitor cell donors – first 10 years of prospective donor follow-up of Swiss donors
Bone Marrow Transplantation (2022)
-
Hematopoietic cell transplant nurse coordinators’ perceptions of related donor care: a European survey from the EBMT Nurses Group
Bone Marrow Transplantation (2020)
-
The potential of non-myeloablative heterochronous autologous hematopoietic stem cell transplantation for extending a healthy life span
GeroScience (2018)