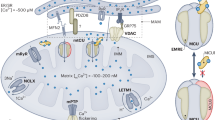
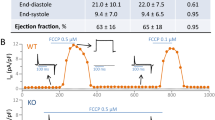
Abstract
In comparison to cation (K+, Na+ and Ca2+) channels, much less is currently known about the functional role of anion (Cl−) channels in cardiovascular physiology and pathophysiology. Over the past 15 years, various types of Cl− currents have been recorded in cardiac cells from different species including humans. All cardiac Cl− channels described to date may be encoded by five different Cl− channel genes: the PKA- and PKC-activated cystic fibrosis tansmembrane conductanceregulator (CFTR), the volume-regulated CIC-2 und CIC-3, and the Ca2+ - activated CLCA or Bestrophin. Recent studies using multiple approaches to examine the functional role of Cl− channels in the context of health and disease have demonstrated that Cl− channels might contribute to: 1) arrhythmogenesis in mycocardial injury; 2) Cardiac ischemic preconditioning; and 3) the adaptive remodeling of the heart during myocardial hypertrophy and heart failure. Therefore anion channels represent very attractive novel targets for therapeutic approaches to the treatment of heart diseases. Recent evidence suggests that Cl− channels, like cation channels, might function as a multiprotein complex or functional module. In the post-genome era, the emergence of functional proteomics has necessitated a new paradigm shift to the structural and functional assessment of integrated Cl− channel multiprotein complexes in the heart, which could provide new insight into our understanding of the underlying mechanisms responsible for heart disease and protection.
Similar content being viewed by others
Article PDF
References
Carmeliet E . Cardiac ionic currents and acute ischemia; from channels to arrhythmias. Physiol Rev 1999; 79: 917–1017.
Marban E . Cardiac channelopathies. Nature 2002; 415; 213–8.
Nattel S, Khairy P, Schram G . Arrhythmogenic ionic remodeling: adaptive responses with maladaptive consequences. Trends Cardiovasc Med 2001; 11: 295–301.
Nattel S . New ideas about atrial fibrillation 50 years on. Nature 2002; 415; 219–26.
Nattel S . Human genetics; lost anchors cost lives. Nature 2003; 421: 587–90.
Gross GJ, Peart JN . KATP channels and myocardial preconditioning; an update. Am J physiol Heart Circ Physiol 2003; 285: H921–30.
O'Rourke B . Evidence for mitochondrial K+ channels and their role in cardioprotection. Circ Res 2004: 94: 420–32.
Hume JR, Duan D, Collier ML, Yamazaki J, Horowitz B . Anion transport in heart, Physiol Rev 2000; 80: 31–81.
Baumgarten CM, Clemo HF . Swelling-activated chloride channels in cardiac physiology and pathophysiology. Prog Biophys Mol Biol 2003; 82: 25–42.
Harvey RD . Cardiac chloride currents. News Phys Sci 1996; 11: 175–81.
Hiraoka M, Kawano S, Hirano Y, Furukawa T . Role of cardiac chloride currents in changes in action potential characteristics and arrhythmias. Cardiovasc Res 1998; 40: 23–33.
Lang F, Busch GL, Ritter M, Volkl H, Waldegger S, Gulbins E, et al. Functional significance of cell volume regulatory mechanisms. Physiol Rev 1998; 78; 247–306.
Lehmann-Horn F, Jurkat-Rott K . Voltage-gated ion channels and hereditary disease. Physiol Rev 1999; 79: 1317–72.
Bahinski A, Nairn AC, Greengard P, Gadsby DC . Chloride conductance regulated by cyclic AMP-dependent protein kinase in cardiac myocytes. Nature 1989; 340: 718–21.
Harvey RD, Hume JR . Autonomic regulation of a chloride current in heart. Science 1989; 244; 983–5.
Nagel G, Hwang TC, Nastiuk KL, Nairn AC, Gadsby DC . The protein kinase A-regulated cardiac CL− channel resembles the cystic fibrosis transmembrane conductance regulator. Nature 1992; 360: 81–4.
Collier ML, Hume JR . Unitary chloride channels activated by protein kinase C in guinea pig ventricular myocytes. Circ Res 1995; 76: 317–24.
Walsh KB, Long KJ . Properties of a protein kinase C-activated chloride current in guinea pig ventricular myocytes. Circ Res 1994; 74: 121–9.
Duan D, Ye L, Britton F Miller LJ, Yamazaki J, Horowitz B, et al. Purinoceptor-coupled Cl− channels in mouse heart; a novel, alternative pathway for CFTR regulation. J Physiol 1999; 521 Pt 1: 43–56.
Levesque PC, Hume JR . ATP but not cAMP activates a chloride conductance in mouse ventricular myocytes. Cardiovasc Res 1995; 29; 336–43.
Yamamoto-Mizuma S, Wang GX, Hume JR . P2Y purinergic receptor regulation of CFTR chloride channels in mouse cardiac myocytes. J Physiol 2004; 556; 727–37.
Duan D, Ye L, Britton F, Horowitz B, Hume JR . A novel anionic inward rectifier in native cardiac myocytes. Circ Res 2000; 86: E63–E71.
Komukai K, Brette F, Orchard CH . Electrophysiological response of rat atrial myocytes to acidosis. Am J physiol Heart Circ Physiol 2002; 283: H715–24.
Komukai K, Brette F, Pascarel C, Orchard CH . Electrophysiological response of rat ventricular myocytes to acidosis. Am J Physiol Heart Circ Physiol 2002; 283: H412–22.
Duan D, Fermini B, Nattel S . Sustained outward current observed after l m1 inactivation in rabbit atrial myocytes is a novel Cl− current. Am J Physiol 1992; 263: H1967–71.
Duan D, Nattel S . Properties of single outwardly rectifying Cl− channels in heart. Circ Res 1994; 75: 789–95.
Duan D, Fermini B, Nattel S . Alpha-adrenergic control of volume-regulated Cl− currents in rabbit atrial myocytes. Characterization of a novel ionic regulatory mechanism. Circ Res 1995; 77; 379–93.
Duan D, Hume JR, Nattel S . Evidence that outwardly rectifying Cl− channels underline volume-regulated Cl− currents in heart. Circ Res 1997; 80: 103–13.
Duan D, Winter C, Cowley S, Hume JR, Horowitz B . Molecular identification of a volume-regulated chloride channel. Nature 1997; 390: 417–21.
Duan D, Cowley S, Horowitz B, Hume JR . A serine residue in CIC-3 links phosphorylation-dephosphorylation to chloride channel regulation by cell volume. J Gen Physiol 1999; 113: 57–70.
Duan D, Zhong J, Hermoso M, Satterwhite CM, Rossow CF, Hatton WJ, et al. Functional inhibition of native volume-sensitive outwardly rectifying anion channels in muscle cells and Xenopus oocytes by anti-CIC-3 antibody. J physiol 2001: 531: 437–44.
Hermoso M, Satterwhite CM, Andrade YN, Hidalgo J, Wilson SM, Horowitz B, et al. CIC-3 is a fundamental molecular component of volume-sensitive outwardly rectifying Cl− channels and volume regulation in HeLa cells and Xenopus laevis oocytes. J Biol Chem 2002; 277: 40066–74.
Wang GX, Hatton WJ, Wang GL, Zhong J, Yamboliev I, Duan D, et al. Functional effects of novel anti-CIC-3 antibodies on native volume-sensitive osmolyte and anion channels in cardiac and smooth muscle cells. Am J physiol Heart Circ Physiol 2003; 285: H1453–63.
Yamamoto-Mizuma S, Wang GX, Liu LL, Schegg K, Hatton WJ Duan D, et al. Altered properties of volume-sensitive osmolyte and anion channels (VSOACs) and membrane protein expression in cardiac and smooth muscle myocytes from Clcn3− mice J Physiol 2004; 557: 439–56.
Britton FC, Ohya S, Horowitz B, Greenwood IA . Comparison of the properties of CLCAI generated currents and I m1 in murine portal vein smooth muscle cells. J Physiol 2002; 539: 107–17.
Collier ML, Levesque PC, Kenyon JL, Hume JR . Unitary Cl− channels activated by cytoplasmic ca2+ in canine ventricular myocytes. Circ Res 1996: 78; 936–44.
Zygmunt AC, Gibbons WR . Properties of the calcium -activated chloride current in heart. J Gen Physiol 1992; 99: 391–414.
Xu Y . Dong PH, Zhang Z, Ahmmed GU, Chiamvimonvat N . Presence of a calcium-activated chloride current in mouse ventricular myocytes. Am J Physiol Heart Circ Physiol 2002; 283: H302–14.
Hartzell C, Putzier I, Arreola J . Calcium-activated chloride channels. Annu Rev physiol 2005; 67: 221–40.
Qu Z, Wei RW, Mann W, Hartzell HC . Two Bestrophins cloned from Xenopus Iaevis oocytes express Ca2+-activated Cl− currents. J Biol Chem 2003; 278: 49563–72.
Qu Z, Fischmeister R, Hartzell C . Mouse Betrophin-2 is a bona fide Cl− channel; identification of a residue important in a union binding and conduction. J Gen Physiol 2004; 123: 327–410.
Qu Z, Hartzell C . Determinants of anion permeation in the second transmembrane domain of the mouse Bestrophin-2 chloride channel. J Gen physiol 2004; 124: 371–82.
Baumgarten CM, Fozzard HA . Intracellular chloride activity in mammalian ventricular muscle. Am J physiol 1981; 241: C121–9.
Caille JP, Ruiz-Ceretti E, Schanne OF . Intracellular chloride activity in rabbit papillary muscle; effect of ouabain. Am J Physiol 1981; 240; C183.
Spitzer KW, Walker JL . Intracellular chloride activity in quiescent cat papillary muscle. Am J physiol 1980; 238; H487–93.
Vaughan-Jones RD . Non-passive chloride distribution in mammalian heart muscle; micro-electrode measurement of the intracellular chloride activity. J physiol (Lond) 1979; 295: 83–109.
Tomaselli GF, Beuckelmann DJ, Calkins HG, Berger RD, Kessler PD, Lawrence JH, et al. Sudden cardiac death in heart failure. The role of abnormal repolarization. Circulation 1994; 90: 1534–9.
Harvey RD, Clark CD, Hume JR . Chloride current in mammalian cardiac myocytes. Novel mechanism for autonomic regulation of action potential duration and resting membrane potential J Gen Physiol 1990; 95: 1077–102.
Matsuoka S, Ehara T, Noma A . Chloride-sensitive nature of the adrenaline-induced current in guinea-pig cardiac myocytes, J physiol (Lond) 1990; 425; 579–98.
Takano M, Noma A . Distribution of the Isoprenaline-induced chloride current in rabbit heart. Pflugers Arch 1992; 420: 223–6.
Yamawake N, Hirano Y, Sawanobori T, Hiraoka M . Arrhythmogenic effects of isoproterenol-activated Cl− current in guinea-pig ventricular myocytes. J Mol Cell Cardiol 1992; 24; 1047–58.
Ruiz PE, Ponce ZA, Schanne OF . Early action potential shortening in hypoxic hearts; role of chloride current(s) mediated by catecholamine release. J Mol Cell Cardiol 1996 28: 279–90.
Du XY, Sorota S . Cardiac swelling-induced chloride current depolarizes canine atrial myocytes. AM J Physiol 1997; 272; H1904–16.
Vandenberg JI, Bett GC, Powell T . Contribution of a swelling-activated chloride current to changes in the cardiac action potential. Am J Physiol 1997; 273: C541–7.
Tomaselli GF, Marban E . Electrophysiological remodeling in hypertrophy and heart failure cardiovasc. Res 1999; 42: 270–83.
Clemo HF, Stambler BS, Baumgarten CM . Swelling-activated chloride current is persistently activated in ventricular myocytes from dogs with tachycardia-induced congestive heart failure Circ Res 1999; 84: 157–65.
Hagiwara N, Masuda H, Shoda M, Irisawa H . Stretch-activated anion currents of rabbit cardiac myocytes. J Physiol (Lond) 1992; 456: 285–302.
Browe DM, Baumgarten CM . Strech of beta 1 integrin activates an outwardly rectifying chloride current via FAK and Sre in rabbit ventricular myocytes. J Gen Physiol 2003; 689–702.
Browe DM, Baumgarten CM . Angiotensin II (ATI) receptor and NADH oxidase regulate Cl− current elicited by β1 integrin stretch in rabbit ventricular myocytes. J Gen Physiol 2004; 124: 273–87.
Zygmunt AC, Gibbons WR . Calcium-activated chloride current in rabbit ventricular myocytes. Circ Res 1991; 68: 424–37.
Verkerk AO, Tan HL, Ravesloot JH . Ca2+ activated Cl− reduces transmural electrical heterogeneity within the rabbit left ventricle. Acta Physiol Scand 2004; 180: 239–47.
Zygmunt AC . Intracellular calcium activates a chloride current in canine ventricular myocytes. Am J Physiol 1994; 267: H1984–95.
January CT, Fozzard HA . Delayed afterdepolarizations in heart muscle: mechanisms and relevance. Pharmcol Rev 1988; 40: 219–27.
Verkerk AO, Veldkamp MW, Bouman LN, Van Ginneken AC . Calcium-activated Cl− current contributes to delayed afterdepolarizations in single Purkinje and ventricular myocytes. Circulation 2000; 101: 2639–44.
Verkerk AO, Veldkamp MW, de Jonge N . Wilders R, van Ginneken AC . Injury current modulates afterdepolarizatoin in single human ventricular cells. Cardiovasc Res 2000; 47; 124–32.
Verkerk AO, Veldkamp MW, Baartscheer A Schumacher CA, Klopping C, van Ginneken AC, et al. Ionic mechanism of delayed afterdepolarizations in ventricular cells isolated from human end-stage failing hearts. Circulation 2001; 104: 2728–33.
Verkerk AO, Wilders R, Coronel R, Ravesloot JH, Verheijek EE . Ionic remodeling of sinoatrial node cells by heart failure. Circulation 2003; 108: 760–6.
Wright AR, Rees SA . Targeting ischaemia-cell swelling and drug efficacy [published erratum appears in Trends Pharmacol Sci 1997; 18: 345]. Trends Pharmacol Sci 1997; 18: 224–8.
Frace AM, Maruoka F, Noma A . Control of the hyperpolarization-activated cation current by external anions in rabbit sinoatrial node cells. J Physiol (Lond) 1992; 453: 307–18.
Nakajima T, Sugimoto T, Kurachi Y . Effects of anions on the G Protein-mediated activation of the muscarinic K+ channel in the cardiac atrial cell membrane. Intracellular chloride inhibition of the GTPase activity of GK. J Gen Physiol 1992; 99: 665–82.
Diaz RJ, Losito VA, Mao GD, Ford MK, Backx PH, Wilson GJ . Chloride channel inhibition blocks the protection of ischemic preconditioning and hypo-osmotic stress in rabbit ventricular myocardium. Circ Res 1999; 84: 763–75.
Diaz RJ, Batthish M, Backx PH, Wilson GJ . Chloride channel inhibition does block the protection of ischemic preconditioning in myocardium. J Mol Cell Cardiol 2001; 33: 1887–9.
Batthish M, Diaz RJ, Zeng HP, Backx PH, Wilson GJ . Pharmacological preconditioning in rabbit myocardium is blocked by chloride channel inhibition. Cardiovasc Res 2002; 55; 660–71.
Heusch G, Liu GS, Rose J, Cohen MV, Downey JM . NO confirmation for a causal role of volume-regulated chloride channels in ischemic preconditioning in rabbits. J Mol Cell Cardiol 2000; 32: 2279–85.
Bozeat ND Dwyer L, Ye L, Yao T, Duan D . The role of CIC-3 chloride channels in early and late ischemic preconditioning in mouse heart. FASEB J 2005; 32; in press.
Mizoguchi K, Maeta H, Yamamoto A, Oe M, Kosaka H . Amelioration of myocardial global ischemia reperfusion injury with volume regulatory chloride channel inhibitors in vivo. Transplantation 2002; 73; 1185–93.
Chen Z, Chua CC, Ho YS, Hamdy RC, Chua BH . Overexpression of Bcl-2 attenuates apoptosis and protects against myocardial I/R injury in transgenic mice. Am J Physiol Heart Circ Physiol 2001; 280; H2313–20.
Lemonnier L, Shuba Y, Crepin A, Roudbaraki M, Slomianny C, Mauroy B, et al. Bcl-2-dependent modulation of swelling-activated CI current and CIC-3 expression in human prostate cancer epithelial cells. Cancer Res 2004; 64: 4841–8.
Wei L, Xiao AY, Jin c, Yang A, Lu ZY, Yu SP . Effects of chloride and potassium channel blockers on apoptotic cell shrinkage and apoptosis in cortical neurons. Pflugers Arch 2004; 448: 325–34.
Sheppard DN, Welsh MJ . Effect of ATP-sensitive K channel regulators on eystic fibrosis transmembrane conductance regulator chloride currents. J Gen Physiol 1992; 100: 573–91.
Sheppard DN, Welsh MJ . Effect of ATP sensitive K channel regulators n cystic fibrosis transmembrane conductance regulator chloride CL channels expressed in a murine cell line J Physiol (Lond) 1997; 503 ( Pt 2); 333–46.
Yamazaki J, Hume JR . Inhibitory effects of glibenclamide on cystic fibrosis transmembrane regulator, swelling-activated, Ca2+-activated Cl− channels in mammalian cardiac myocytes. Circ Res 1997: 81: 101–9.
Lochner A, Genade S, Tromp E, Podzuweit T, Moolman JA . Ischemic preconditioning and the beta-adrenergic signal transduction pathway. Circulation 1999; 100: 958–66.
Ping P, Song C, Zhang J, Guo Y, Cao X, Li RC, et al. Formation of protein Kinase C (epsilon)-Lek signaling modules confers cardioprotection. J Clin Invest 2002; 109; 499–507.
Yamazaki J, Britton F, Collier ML, Horowitz B Hume JR . Regulation of recombinant cardiac cystic fibrosis transmembrane conductance regulator chloride channels by protein kinase C. Biophys J 1999; 76: 1972–87.
Chen H, Liu LL, Ye LL, McGuckin C, Tamowski S, Scowen P, et al. Targeted inactivation of cystic fibrosis transmembrane conductance regulator chloride channel gene prevents ischemic preconditioning in isolated mouse heart. Circulation 2004; 110; 700–4.
Ye L, Ge ZD Liu L Murray K, Hatton WJ, Auchampach JA, et al. In vivo study of the protective role of CFTR chloride channels in ischemic preconditioning. FASEB J 2003; 174: A106.
Boujaoude I.C, Bradshaw-Wilder C, Mao C, Cohn J, Ogretmen B, Hannun YA, et al. Cystic fibrosis transmembrane regulator regulates uptake of sphingoid base phosphates and lysophosphatidic acid; modulation of cellular activity of sphingosine 1-phosphate. J Biol Chem 2001; 276: 35258–64.
Karliner JS . Lysophospholipids and the cardiovascular system. Biochim Biophys Acta 2001: 1582: 216–21.
Kogan L, Ramjeesingh M, Li C, Kidd JF, Wang Y, Leslie EM, et al. CFTR directly mediates nucleotide-regulated glutathione flue. EMBO J 2003; 22: 1981–9.
Barriere H, Poujeol C, Tauc M, Blasi JM, Counillon L, Poujcol P . CFTR modulates programmed cell death by decreasing intracellular PH in Chinese hamster lung fibroblast. Am J Physiol Cell Physiol 2001; 281: C810–24.
Valverde MA, Vazquez E, Munoz FJ, Nobles M, Delaney SJ, Wainwright BJ, et al. Murine CFTR channel and its role in regulatory volume decrease of small intestine crypts. Cell Physiol Biochem 2000; 10: 321–8.
Piper HM, Meuter K, Schafer C . Cellular mechanisms of ischemia reperfusion injury. Ann Thorac Surg 2003; 75: S644–8.
Piper HM, Abdallah Y, Schafer C . the first minutes of reperfusion; a window of opportunity for cardioprotection. Cardiovasc Res 2004; 61: 365–71.
Li GR, Du XL, Siow, YL, O K, Tse HF, Lau CP . Calcium-activated transient outward chloride current and phase I repolarization of swine ventricular action potential. Cardiovasc Res 2003; 58: 89–98.
Li GR Sun H, To J, Tse HF, Lau CP . Demonstration of calcium-activated transient outward chloride current and delayed rectifier potassium currents in Swine atrial myocytes. J Mol Cell Cardiol 2004: 36; 495–504.
Verkerk AO, Wilders R Zegers JG, van Borren MM, Ravesloot JH, Verheijck EE . Ca2+-activated CL current in rabbit sinoatrial mode cells. J Physiol 2002; 540: 105–17.
Zygmunt AC, Goodrow RJ, Weigel CM . INa(ca) and I Ca(ca) contribute to isoproterenol-induced delayed after depolarizations in midmyocardial cells. Am J Physiol 1998; 275: H1979–92.
Laser M, Willey CD, Jiang W, Cooper G, Menick DR, Zile MR, et al. Integrin activation and focal complex formation in cardeiac hypertrophy. J Biol Chem 2000; 275: 35624–30.
Weber KT, Sun Y, Guarda E . Structural remodeling in hypertensive heart disease and the role of hormones. Hypertension 1994: 23: 869–77.
Colucci WS . Molecular and cellular mechanisms of muyocardial failure. Am J Cardiol 1997; 80: 15L–25L.
Clemo HF, Stambler BS, Baumgarten CM . Swelling-activated chloride current is persistently activated in ventricular myocytes from dogs with tachycardia-induced congestive heart failure Circ Res 1999; 84; 157–65.
Clemo HF, Baumgarten CM . Protein Kinase C activation blocks I Cl and causes myocyte swelling in a rabbit congestive heart failure model. Circulation 1998: 98; 1–695.
Clemo HF, Danetz, JS, Baumgarten CM . Does CIC-3 modulate cardiac cell volume? Biophy J 1999; 76: A203.
Clemo HF, Rana J, Vaida AM, Tseng GN, Higgins RS, Baumgarten CM . Chronic activation of I Cl() in canine infarction model supreses inducibility of early afterdepolarizations. Circulation 2001; 104: II–624.
Patel DG, Higgins RS, Baumgarten CM . Swelling-activated Cl current, I Cl is chronically activated in diseased human atrial myocytes. Biophys J 2003: 84: 233a.
Benitah JP, Gomez AM, Delgado C, Lorente, P, Lederer, WJ . A chloride current components induced by hypertrophy in rat ventricular myocytes. Am J Physiol 1997; 272; H2500–6.
De Mello WC . Heart failure; how important is cellular sequestration? The role of the rennin-angiotensin-aldosterone system. J Mol Cell Cardiol 2004; 37: 431–8.
Liu L, McGuckin C, Hallon WJ, Dudan D . Disruption of Clan3gen in mice facilitates heart failure during pressure overload. J Gen Physiol 2003: 122: 76.
Liu L, Ye L, McGuckin C, Hatton WJ, Duan D . Disruption of Clean3 gene in mice facilitiates heart failure during pressure overload J Gen Physiol 2001; 532: 661–72.
Duffy SM, Leyland ML, Conley EC, Bradding P . Voltage-dependent and calcium-activated ion channels in the human must cell line HMC-1. J Leukoc Biol 2001; 70: 233–40.
Olsen ML, Schade S, Lyons SA, Amaral MD, Sontheimer H . Expression of voltage-gated chloride channels in human glioma cells. J Neurosci 2003; 23: 5572–82.
Paul S . Ventricular remodeling. Crit Care Nurs Clin North Am 2003; 15: 407–11.
Wong KR, Trezise AE, Crozatier B, Vandenberg JI . Loss of the normal epicardial to endocardial gradient of cftr mRNA expression in the hypertrophied rabbit left ventricle. Biochem Biophys Res Common 2000; 278: 144–9.
Davies WL, Vandenberg JI, Sayeed RA, Trezise AE . Post-transcriptional regulation of the cystic fibrosis gone in cardiac development and hypertrophy. Biochem Biophys Res Commun 2004; 319: 410–8.
Houser SR, Piacentino V III, Weisser J . Abnormalities of calcium cycling in the hypertrophied and failing heart. J Mol Cell Cardiol 2000; 32: 1595–607.
Elble RC, Pauli BU . Tumor suppression by a proapoptotic calcium activated chloride channel in mammary epithelium J boil Chem 2001; 276: 40510–7.
Verkerk AO, Tan HL, Krikels JH, Ravesloot JH . Role of Ca2+-activated Cl+ current during proarrhythmic early afterdepolarizations in sheep and human ventricular myocytes. Acta Physiol Scand 2003: 179: 143–8.
Moore RC, Lee IY, Silverman GL, Harrison PM, Strome R, Heinrich C, et al. Ataxia in prion protein (PrP)-deficient mice is associated with upregulation of the novel PrP-like protein doppel. J Mol Biol 1999; 292; 797–817.
London B . A knockout may not always be a knockout. Circulation 2000; 102: E122.
Jentsch TJ, Stein V, Weinreich F, Zdebik AA . Molecular structure and physiological function of chloride channels. Physiol Rev 2002; 82: 503–68.
Estevez R, Boettger T, Stein V, Birkenhager R, Otto E, Hilebrandt F et al. Barttin is a Cl− channel beta-subunit crucial for renal Cl− reabsorption and inner ear K secretion. Nature 2001; 414; 558–61.
Ardehali H, Chen Z, Ko Y, Mejia-Alvarez R, Marban E . Multiprotein complex containing succinate dehydrogenase confers mitochondrial ATP-sensitive K+ channel activity. Proc Natl Acad Sci USA 2004: 101: 11880–5.
Ping P, Song C, Zhang J, Guo Y, Cao X, Li RC, et al. Formation of protein kinase C(epsilon)-Lck signaling modules confers cardioprotection. J Clin Invest 2002; 109: 499–507.
Dutzler R, Campbell EB, Cadene M, Chait BT, Mackinnon R . X-ray structure of a CIC chloride channel at 3.0 A reveals the molecular basis of anion selectivity. Nature 2002; 415: 287–94.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by National Center for Research Resources (NCRR) P-20 RR-15581 and National Heart, Lung, and Blood Institute Grant (HL 63914).
Rights and permissions
About this article
Cite this article
Duan, Dy., Liu, L., Bozeat, N. et al. Functional role of anion channels in cardiac diseases. Acta Pharmacol Sin 26, 265–278 (2005). https://doi.org/10.1111/j.1745-7254.2005.00061.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00061.x
Keywords
This article is cited by
-
Association between serum chloride and in-hospital mortality in congestive heart failure with diabetes: Data from the MIMIC-IV database
Journal of Diabetes & Metabolic Disorders (2023)
-
Hypochloraemia in Patients with Heart Failure: Causes and Consequences
Cardiology and Therapy (2020)
-
Cardiac optogenetics: using light to monitor cardiac physiology
Basic Research in Cardiology (2017)
-
Impaired cardiac and peripheral hemodynamic responses to inhaled β2-agonist in cystic fibrosis
Respiratory Research (2015)
-
The ClC-3 chloride channels in cardiovascular disease
Acta Pharmacologica Sinica (2011)