Abstract
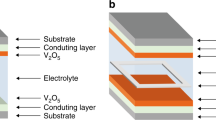
PHOTOCHROMIC materials1,2 change colour on absorption of light, whereas electrochromic materials3,4 change colour in response to an electrically induced change in oxidation state. Both classes of materials are being investigated for potential applications in displays, imaging devices and 'smart' windows5–8,15,16. Here we describe an alternative route to such applications, in which an electrochromic film and a photovoltaic film form the two electrodes of an electrochemical cell. The resulting structure exhibits photochromism, but unlike conventional photochromic films, the light-absorption process (in the photovoltaic film) is separate from the coloration process (in the electrochromic film): both may therefore be optimized individually. Moreover, as the coloration process in our cells requires an external electrical current between the two electrodes, the optical state of the cell—transparent, absorbing or, in the case of non-uniform illumination, patterned—can be stored when the circuit is open, or changed when the electrodes are connected.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dürr, H. & Bouas-Laurent, H. Photochromism: Molecules and Systems (Elsevier, Amsterdam, 1990).
Yao, J. N., Hashimoto, K. & Fujishima, A. Nature 355, 624–626 (1992).
Deb, S. K. Sol. Energy Mater. Sol. Cells 25, 327–338 (1992).
Green, M., Smith, W. C. & Weiner, J. A. Thin Solid Films 38, 89–100 (1976).
Granqvist, C. G. Solid State Mater. Sci. 16, 291–308 (1990).
Benson, D. K. & Branz, H. M. Sol. Energy Mater. Sol. Cells 39, 203–211 (1995).
Nagasu, M. & Koshida, N. Appl. Phys. Lett. 57, 1324–1325 (1990).
Deb, S. K. Appl. Opt. Suppl. 3, 192–195 (1969).
O'Regan, B. & Grätzel, M. Nature 353, 737–740 (1991).
Nazeeruddin, M. K. et al. J. Am. Chem. Soc. 115, 6382–6390 (1993).
Parkinson, B. A. & Spitler, M. T. Electrochim. Acta 37, 943–948 (1992).
Kay, A. & Grätzel, M. J. Phys. Chem. 97, 6272–6277 (1993).
Faughnan, B. W., Crandall, R. S. & Heyman, P. M. RCA Rev. 36, 177–197 (1975).
Bechinger, C. et al. J. Appl. Phys. 80, 1226–1232 (1996).
Bonhôte, P. et al. Chem. Commun. 1163–1164 (1996).
DeBerry, D. W. & Viehbeck, A. J. Electrochem. Soc. 130, 249–251 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bechinger, C., Ferrere, S., Zaban, A. et al. Photoelectrochromic windows and displays. Nature 383, 608–610 (1996). https://doi.org/10.1038/383608a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/383608a0
This article is cited by
-
First-principles analysis of how Cobalt doping affects the structural, electronic, and optical properties of α-MoO3
Indian Journal of Physics (2024)
-
A New Tripodal Zwitterionic Ligand-Based Cationic Manganese(II) Metal-Organic Framework: Synthesis, Characterization and Photochromic Property
Journal of Inorganic and Organometallic Polymers and Materials (2024)
-
Synthesis and study of photoelectrochromic and photocatalytic behavior of double-layer N-doped TiO2/Co3O4 configuration via DC reactive magnetron sputtering
Optical and Quantum Electronics (2024)
-
Optimization of Optical Modulation in Amorphous WO3 Thin Films
Electronic Materials Letters (2024)
-
Investigating the Properties of Triangle Terthiophene and Triphenylamine Configured Propeller-like Photochromic Dye with Ethyne Bridge
Journal of Fluorescence (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.