Abstract
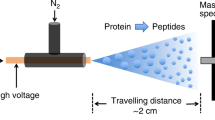
MOLECULAR analysis of complex biological structures and processes increasingly requires sensitive methods for protein sequencing. Electrospray mass spectrometry1 has been applied to the high-sensitivity sequencing of short peptides2, but technical difficulties have prevented similar success with gel-isolated proteins. Here we report a simple and robust technique for the sequencing of proteins isolated by polyacrylamide gel electro-phoresis, using nano-electrospray3,4 tandem mass spectrometry5,6. As little as 5ng protein starting material on Coomassie- or silver-stained gels can be sequenced. Multiple-sequence stretches of up to 16 amino acids are obtained, which identify the protein unambiguously if already present in databases or provide information to clone the corresponding gene. We have applied this method to the sequencing and cloning of a protein which inhibits the proliferation of capillary endothelial cells in vitroand thus may have potential antiangiogenic effects on solid tumours.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fenn, J. B., Mann, M., Meng, C. K., Wong, S. F. & Whitehouse, C. M. Science 246, 64–71 (1989).
Hunt, D. F. et al. Science 255, 1261–1263 (1992).
Wilm, M. S. & Mann, M. Int. J. Mass Spectrom. Ion Process. 136, 167–180 (1994).
Wilm, M. & Mann, M. Analyt. Chem. 68, 1–8 (1996).
Biemann, K. & Scoble, H. A. Science 237, 992–998 (1987).
Hunt, D. F., Yates, J. R., Shabanowitz, J., Winston, S. & Hauer, C. R. Proc. natn. Acad. Sci. U.S.A. 83, 6233–6237 (1986).
Mann, M. & Wilm, M. S. Analyt. Chem. 66, 4390–4399 (1994).
Fotsis, T. et al. Proc. natn. Acad. Sci. U.S.A. 90, 2690–2694 (1993).
Fotsis, T. et al. Nature 368, 237–239 (1994).
Walker, J. E. et al. J. molec. Biol. 226, 1051–1072 (1992).
Hunkapiller, M. et al. Nature 310, 105–111 (1984).
Erdjument-Bromage, H., Lui, M., Sabatini, D. M., Snyder, S. H. & Tempst, P. Protein Sci. 3, 2435–2446 (1994).
Matsudaira, P. (ed.) A Practical Guide to Protein and Peptide Purification for Microsequencing (Academic, San Diego, CA, 1993).
Roepstorff, P. & Fohlman, J. Biomed. Mass Spectrom. 11, 601 (1984).
Jenö, P., Mini, T., Hintermann, E. & Horst, M. Analyt. Biochem. 224, 451–455 (1995).
Rosenfeld, J., Capdevielle, J., Guillemot, J. C. & Ferrara, P. Analyt. Biochem. 203, 173–179 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wilm, M., Shevchenko, A., Houthaeve, T. et al. Femtomole sequencing of proteins from polyacrylamide gels by nano-electrospray mass spectrometry. Nature 379, 466–469 (1996). https://doi.org/10.1038/379466a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/379466a0
This article is cited by
-
TMX4-driven LINC complex disassembly and asymmetric autophagy of the nuclear envelope upon acute ER stress
Nature Communications (2023)
-
An ATR-PrimPol pathway confers tolerance to oncogenic KRAS-induced and heterochromatin-associated replication stress
Nature Communications (2023)
-
Affinity Selection from Synthetic Peptide Libraries Enabled by De Novo MS/MS Sequencing
International Journal of Peptide Research and Therapeutics (2022)
-
The splicing factor XAB2 interacts with ERCC1-XPF and XPG for R-loop processing
Nature Communications (2021)
-
Apoptosis-Inducing Active Protein from Marine Clam Donax variabilis on NSCLC Cells
International Journal of Peptide Research and Therapeutics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.