Abstract
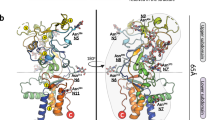
THE polyomaviruses are non-enveloped, icosahedrally symmetrical particles with circular double-stranded DNA genomes1,2. The outer shell of the virion contains 360 copies of viral protein VP1 (Mr ~42K) arranged in pentamers3. We report here the structure at 3.65 Å resolution of murine polyomavirus (‘polyoma’) complexed with an oligosaccharide receptor fragment. This structure has been determined using the previously described model of simian virus 40 (SV40) 4. Although very similar in structure to SV40, poly oma has interesting biological differences. Cell-surface N-ace-tyl neuraminic acid (sialic acid) is required for polyoma infectivity, but not for SV40. Polyoma attaches to the surface of susceptible cells by stereospecific recognition of oligosaccharides terminating in (α2,3)-linked sialic acid5,6. Studies of pathogenicity show that the specificity of viral binding to such oligosaccharides is an important determinant of the virus’ ability to establish a disseminated infection and to induce tumours in the natural host. The complex described here shows how polyoma recognizes the receptor fragment and how strains with different receptor specificities can distinguish between alternative ligands. The results also suggest an explanation for the large disparity in pathogenicity exhibited by strains differing in only one amino-acid residue of VP17,8.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Tooze, J. DNA Tumor Viruses (Cold Spring Harbor Laboratory Press, New York, 1981).
Eckhart, W. in Virology 2nd edn (ed. Fields, B. N.) 1593–1607 (Raven, New York, 1990).
Rayment, I., Baker, T. S., Caspar, D. L. D. & Murakami, W. T. Nature 295, 110–115 (1982).
Liddington, R. C., Yan, Y., Moulai, J., Sahli, R., Benjamin, T. L. & Harrison, S. C. Nature 354, 278–284 (1991).
Fried, H., Cahan, L. D. & Paulson, J. C. Virology 109, 188–192 (1981).
Cahan, L. D., Singh, R. & Paulson, J. C. Virology 130, 281–289 (1983).
Freund, R., Garcea, R. L., Sahli, R. & Benjamin, T. L. J. Virol. 65, 350–355 (1991).
Freund, R., Calderone, A., Dawe, C. J. & Benjamin, T. L. J. Virol. 65, 335–341 (1991).
Sabesan, S., Bock, K. & Paulson, J. C. Carbohydr. Res. 218, 27–54 (1991).
Poppe, L., Dabrowski, J., von der Lieth, C.-W., Numata, M. & Ogawa, T. Eur. J. Biochem. 180, 337–342 (1989).
Weis, W. et al. Nature 333, 426–431 (1988).
Sauter, N. K. et al. Biochemistry 31, 9609–9621 (1992).
Quiocho, F. A. Pure appl. Chem. 61, 1293–1306 (1989).
Wright, C. S. J. molec. Biol. 215, 635–651 (1990).
Crawford, L. V. In Fundamental Techniques in Virology (eds Habel, K. & Salzman, N. P.) 75–81 (Academic, New York, 1969).
CCP4, The SERC (UK) Collaborative Computing Project No. 4, a Suite of Programs for Protein Crystallography, distributed from Daresbury Laboratory, Warrington WA4 4AD, UK (1979).
Bricogne, G. Acta crystallogr. A32, 832–847 (1976).
Jones, T. A. Meth. Enzym. 115, 157–171 (1985).
Brünger, A. T. X-PLOR Version 3.1: A System for Crystallography and NMR (Yale Univ., 1992).
Brünger, A. T. Nature 355, 472–475 (1992).
Kraulis, P. J. J. appl. Crystallogr. 24, 946–950 (1991).
Jones, T. A., Zhou, J.-Y., Cowan, S. W. & Kjeldgaard, M. Acta crystallogr. A47, 110–119 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stehle, T., Yan, Y., Benjamin, T. et al. Structure of murine polyomavirus complexed with an oligosaccharide receptor fragment. Nature 369, 160–163 (1994). https://doi.org/10.1038/369160a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/369160a0
This article is cited by
-
Stress proteins: the biological functions in virus infection, present and challenges for target-based antiviral drug development
Signal Transduction and Targeted Therapy (2020)
-
Survey of molecular chaperone requirement for the biosynthesis of hamster polyomavirus VP1 protein in Saccharomyces cerevisiae
Archives of Virology (2016)
-
Construction of polyomavirus-derived pseudotype virus-like particles displaying a functionally active neutralizing antibody against hepatitis B virus surface antigen
BMC Biotechnology (2015)
-
Production of recombinant VP1-derived virus-like particles from novel human polyomaviruses in yeast
BMC Biotechnology (2015)
-
A cornucopia of human polyomaviruses
Nature Reviews Microbiology (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.