Abstract
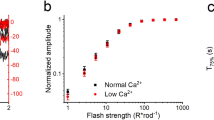
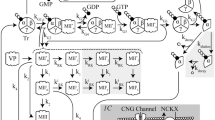
Visual excitation in retinal rod cells is mediated by a cascade that leads to the amplified hydrolysis of cyclic GMP (cGMP) and the consequent closure of cGMP-activated cation-specific channels in the plasma membrane1–3. Recovery of the dark state requires the resynthesis of cGMP, which is catalysed by guanylate cyclase, an axoneme-associated enzyme4–6. The lowering of the cytosolic calcium concentration (Cai) following illumination7–10 is thought to be important in stimulating cyclase activity11,12. This hypothesis is supported by the finding that the cGMP content of rod outer segments increases several-fold when Cai is lowered to less than 10 nM13–16. It is evident that cGMP and Cai levels are reciprocally controlled by negative feedback1,2. Guanylate cyclase from toad ROS is strongly stimulated when the calcium level is lowered from 10 μM to 10 nM, but only if they are excited by light17. We show here that the guanylate cyclase activity of unilluminated bovine rod outer segments increases markedly (5 to 20-fold) when the calcium level is lowered from 200 nM to 50 nM. This steep dependence of guanylate cyclase activity on the calcium level in the physiological range has a Hill coefficient of 3.9. Stimulation at low calcium levels is mediated by a protein that can be released from the outer segment membranes by washing with a low salt buffer. Calcium sensitivity is partially restored by adding the soluble extract back to the washed membranes. The highly cooperative activation of guanylate cyclase by the light-induced lowering of Cai is likely to be a key event in restoring the dark current after excitation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Pugh, E. N. Jr & Cobbs, W. H. Vision Res. 26, 1613–1643 (1986).
Stryer, L. A. Rev. Neurosci. 9, 87–119 (1986).
Baylor, D. A. Invest Opthalmol. Vis. Sci. 28, 34–49 (1987).
Krishnan, N., Fletcher, R. T., Chader, G. J. & Krishna, G. Biochim Biophys. Acta 523, 506–515 (1978).
Fleischman, D. & Denisevich, M. Biochemistry 18, 5060–5066 (1979).
Fleischman, D., Denisevich, M., Raveed, D. & Pannbacker, R. G. Biochim. Biophys. Acta 630, 176–186 (1980).
Yau, K.-W. & Nakatani, K. Nature 313, 579–582 (1985).
McNaughton, P. A., Cervetto, L. & Nunn, B. J. Nature 322, 261–263 (1986).
Miller, D. L. & Korenbrot, J. I. J. gen. Physiol. 90, 397–425 (1987).
Lamb, T. D., Matthews, H. R. & Torre, V. J. Physiol. 372, 315–349 (1986).
Hodgkin, A. L., McNaughton, P. A. & Nunn, B. J. J. Physiol. 358, 447–468 (1985).
Kondo, H. & Miller, W. H. Proc. natn. Acad. Sci. U.S.A. 85, 1322–1326 (1988).
Lipton, S. A. & Dowling, J. E. Curr. Top. Membranes Tramp. 15, 381–392 (1981).
Woodruff, M. L. & Fain, G. L. J. gen. Physiol. 80, 537–555 (1982).
Kilbride, P. J. gen. Physiol. 75, 457–465 (1980).
Lolley, R. N. & Racz, E. Vision Res. 22, 1481–1486 (1982).
Pepe, I. M., Panfoli, I. & Cugnoli, C. FEBS Lett. 203, 73–76 (1986).
Nakatani, K. & Yau, K.-W. J. Physiol. 395, 695–729 (1988).
Korenbrot, J. I. & Miller, D. L. Neurosci. Res. Suppl. 4, S11–S34 (1986).
Schnetkamp, P. P. M. J. Physiol. 373, 25–45 (1986).
Ames, A. III et al. J. biol. Chem. 261, 13034–13042 (1986).
Hodgkin, A. L. in Proc. First Retina Research Foundation Symp. (Portfolio, Woodlands Texas, in the press).
Robinson, P. R., Kawamura, S., Abramson, B. & Bownds, M. D. J. gen. Physiol. 76, 631–645 (1980).
Kawamura, S. & Bownds, M. D. J. gen. Physiol. 77, 571–591 (1981).
Grynkiewicz, G., Poenie, M. & Tsien, R. Y. J. biol. Chem. 260, 3440–3450 (1985).
Reysz, L. J., Carroll, A. G. & Jarrett, H. W. Analyt. Biochem. 166, 107–112 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koch, KW., Stryer, L. Highly cooperative feedback control of retinal rod guanylate cyclase by calcium ions. Nature 334, 64–66 (1988). https://doi.org/10.1038/334064a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/334064a0
This article is cited by
-
Chemical shift assignments of retinal guanylyl cyclase activating protein 5 (GCAP5) with a mutation (R22A) that abolishes dimerization and enhances cyclase activation
Biomolecular NMR Assignments (2023)
-
The structure of the native CNGA1/CNGB1 CNG channel from bovine retinal rods
Nature Structural & Molecular Biology (2022)
-
Biochemistry and physiology of zebrafish photoreceptors
Pflügers Archiv - European Journal of Physiology (2021)
-
Regulation of retinal membrane guanylyl cyclase (RetGC) by negative calcium feedback and RD3 protein
Pflügers Archiv - European Journal of Physiology (2021)
-
cAMP-Dependent Regulation of the Phototransduction Cascade in Cones
Neuroscience and Behavioral Physiology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.