Abstract
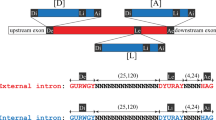
The discovery of intervening sequences (introns) in eukaryotic genes has raised questions about the origin and evolution of these sequences. Hypotheses concerning these topics usually consider the intron as a unit that could be lost or gained over time1–6, or as a region within which recombination can occur to facilitate the production of new proteins by exon shuffling7. Additional com-plexities are observed in introns of mitochondrial and chloroplast genes which contain secondary structures required for messenger RNA splicing8,9 and open-reading frames encoding proteins10,11. Here we describe differences in the organization of protein-coding sequences in the intron of the mitochondrial ND1 gene in two closely related species of Neurospora. These differences show that intron sequences involved in secondary structure formation and in protein coding can evolve as physically distinct elements. Indeed, the secondary structure elements of the ND1 intron can contain two different coding sequences located at two different positions within the intron.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sharp, P. A. Cell 42, 397–400 (1985).
Rogers, J. Nature 315, 458–459 (1985).
Doolittle, W. F. Nature 272, 581–582 (1978).
Darnell, J. E. Science 202, 1257–1260 (1978).
Cavalier-Smith, T. J. cell Sci. 34, 247–278 (1978).
Crick, F. Science 204, 264–271 (1979).
Gilbert, W. Nature 271, 501 (1978).
Michel F., Jacquier, A. & Dujon, B. Biochimie 64, 867–881 (1982).
Davies, R. W., Waring, R. B., Ray, J. A., Brown, T. A. & Scazzocchio, C. Nature 300, 719–724 (1982).
Borst, P. & Grivell, L. A. Nature 289, 439–440 (1981).
Kotylak, Z., Lazowska, J. & Slonimski, P. P. in Achievements and Perspectives of Mitochondrial Research. Vol. 2. (eds E. Quagliariello et al.) 1–20 (Elsevier, Amsterdam, 1985).
Burger, G. & Werner, S. J. molec. Biol. 185, 231–242 (1985).
Waring, R. B., Brown, T. A., Ray, J. A., Scazzocchio, C. & Davies, R. W. Proc. natn. Acad. Sci. U.S.A. 79, 6332–6336 (1982).
Hensgens, L. A., Bonen, L., deHaan, M., van der Horst, G. & Grivell, L. A. Cell 32, 379–389 (1983).
Merlos-Lange, A. M., Kanbay, F., Zimmer, M. & Wolf, K. Molec. gen. Genet. 206, 273–278 (1987).
Dujon, B., Colleaux, L., Jacquier, A., Michel, F. & Monteilhet, C. in Extrachromosomal Elements in Lower Eukaryotes (eds R. B. Wickner et al.) 5–28 (Plenum, New York, 1986).
Waring, R. B., Brown, T. A., Ray, J. A., Scazzocchio, C. & Davies, R. W. EMBO J. 3, 2121–2128 (1984).
Lang, B. F. EMBO J. 3, 2129–2136 (1984).
Michel, F. & Cummings, D. J. Curr. Genet. 10, 69–79 (1985).
de Samaroczy, M. & Bernardi, B. Gene 41, 1–22 (1986).
Coruzzi, G., Bonitz, S. G., Thalenfeld, B. E. & Tzagoloff, A. J. biol. Chem. 256, 12780–12787 (1981).
Michel, F. Curr. Genet. 8, 307–317 (1984).
Seraphin, B., Simon, M. & Faye, G. Nucleic Acids Res. 13, 3005–3014 (1985).
Burke, J. M. et al. Nucleic Acids Res. 15, 7217–7221 (1987).
Sanger, F., Nicklen, S. & Coulson, A. R. Proc. natn. Acad. Sci. U.S.A. 74, 5463–5467 (1977).
Mizusawa, S., Nishimura, S. & Seela, F. Nucleic Acids Res. 14, 1319–1324 (1986).
Mills, D. R. & Kramer, F. R. Proc. natn. Acad. Sci. U.S.A. 76, 2232–2235 (1979).
Tabor, S. & Richardson, C. C. Proc. natn. Acad. Sci. U.S.A. 84, 4767–4771 (1987).
Burke, J. & RajBhandary, U. L. Cell 31, 509–520 (1983).
Jacquier, A. & Dujon, B. Cell 41, 383–394 (1983).
Macreadie, I. G., Scott, M. R., Zinn, A. R. & Butow, R. A. Cell 41, 395–402 (1983).
Henikoff, S., Keene, M. A., Fechtel, K. & Fristrom, J. W. Cell 44, 33–42 (1986).
Chen, C., Malone, T., Beckendorf, S. K. & Davis, R. L. Nature 329, 721–725 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mota, E., Collins, R. Independent evolution of structural and coding regions in a Neurospora mitochondrial intron. Nature 332, 654–656 (1988). https://doi.org/10.1038/332654a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/332654a0
This article is cited by
-
The mtDNA rps3 locus has been invaded by a group I intron in some species of Grosmannia
Mycoscience (2012)
-
Interpretation of mtDNA RFLP variability among Aspergillus tubingensis isolates
Antonie van Leeuwenhoek (2007)
-
Multiple origins of fungal group I introns located in the same position of nuclear SSU rRNA gene
Journal of Molecular Evolution (1998)
-
A group-I intron in the mitochondrial small subunit ribosomal RNA gene of Sclerotinia sclerotiorum
Current Genetics (1995)
-
Homologous maturase-like proteins are encoded within the group I introns in different mitochondrial genes specifying Yarrowia lipolytica cytochrome c oxidase subunit 3 and Saccharomyces cerevisiae apocytochrome b
Current Genetics (1994)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.