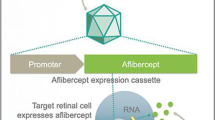
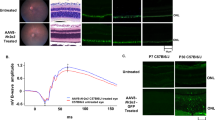
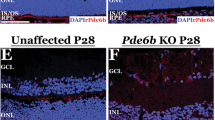
Abstract
The eye has unique advantages as a target organ for gene therapy of both inherited and acquired ocular disorders and offers a valuable model system for gene therapy. The eye is readily accessible to phenotypic examination and investigation of therapeutic effects in vivo by fundus imaging and electrophysiological techniques. Considerable progress has been made in the development of gene replacement therapies for retinal degenerations resulting from gene defects in photoreceptor cells (rds, RPGRIP, RS-1) and in retinal pigment epithelial cells (MerTK, RPE65, OA1) using recombinant adeno-associated virus and lentivirus-based vectors. Gene therapy also offers a potentially powerful approach to the treatment of complex acquired disorders such as those involving angiogenesis, inflammation and degeneration, by the targeted sustained intraocular delivery of therapeutic proteins. Proposals for clinical trials of gene therapy for early-onset retinal degeneration owing to defects in the gene encoding the visual cycle protein RPE65 have recently received ethical approval.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rolling F . Recombinant AAV-mediated gene transfer to the retina: gene therapy perspectives. Gene Ther 2004; 11 (Suppl 1): S26–S32.
Bainbridge JW, Mistry A, Schlichtenbrede FC, Smith A, Broderick C, de AM et al. Stable rAAV-mediated transduction of rod and cone photoreceptors in the canine retina. Gene Ther 2003; 10: 1336–1344.
Lotery AJ, Yang GS, Mullins RF, Russell SR, Schmidt M, Stone EM et al. Adeno-associated virus type 5: transduction efficiency and cell-type specificity in the primate retina. Hum Gene Ther 2003; 14: 1663–1671.
Weber M, Rabinowitz J, Provost N, Conrath H, Folliot S, Briot D et al. Recombinant adeno-associated virus serotype 4 mediates unique and exclusive long-term transduction of retinal pigmented epithelium in rat, dog, and nonhuman primate after subretinal delivery. Mol Ther 2003; 7: 774–781.
Acland GM, Aguirre GD, Bennett J, Aleman TS, Cideciyan AV, Bennicelli J et al. Long-term restoration of rod and cone vision by single dose rAAV-mediated gene transfer to the retina in a canine model of childhood blindness. Mol Ther 2005; 12: 1072–1082.
Min SH, Molday LL, Seeliger MW, Dinculescu A, Timmers AM, Janssen A et al. Prolonged recovery of retinal structure/function after gene therapy in an Rs1h-deficient mouse model of x-linked juvenile retinoschisis. Mol Ther 2005; 12: 644–651.
Challa P, Luna C, Liton PB, Chamblin B, Wakefield J, Ramabhadran R et al. Lentiviral mediated gene delivery to the anterior chamber of rodent eyes. Mol Vis 2005; 11: 425–430.
Loewen N, Leske DA, Cameron JD, Chen Y, Whitwam T, Simari RD et al. Long-term retinal transgene expression with FIV versus adenoviral vectors. Mol Vis 2004; 10: 272–280.
Cheng L, Toyoguchi M, Looney DJ, Lee J, Davidson MC, Freeman WR . Efficient gene transfer to retinal pigment epithelium cells with long-term expression. Retina 2005; 25: 193–201.
Tschernutter M, Schlichtenbrede FC, Howe S, Balaggan KS, Munro PM, Bainbridge JW et al. Long-term preservation of retinal function in the RCS rat model of retinitis pigmentosa following lentivirus-mediated gene therapy. Gene Ther 2005; 12: 694–701.
Miyazaki M, Ikeda Y, Yonemitsu Y, Goto Y, Sakamoto T, Tabata T et al. Simian lentiviral vector-mediated retinal gene transfer of pigment epithelium-derived factor protects retinal degeneration and electrical defect in Royal College of Surgeons rats. Gene Ther 2003; 10: 1503–1511.
Kostic C, Chiodini F, Salmon P, Wiznerowicz M, Deglon N, Hornfeld D et al. Activity analysis of housekeeping promoters using self-inactivating lentiviral vector delivery into the mouse retina. Gene Ther 2003; 10: 818–821.
Bainbridge JW, Stephens C, Parsley K, Demaison C, Halfyard A, Thrasher AJ et al. In vivo gene transfer to the mouse eye using an HIV-based lentiviral vector; efficient long-term transduction of corneal endothelium and retinal pigment epithelium. Gene Ther 2001; 8: 1665–1668.
Gruter O, Kostic C, Crippa SV, Perez MT, Zografos L, Schorderet DF et al. Lentiviral vector-mediated gene transfer in adult mouse photoreceptors is impaired by the presence of a physical barrier. Gene Ther 2005; 12: 942–947.
Molina RP, Ye HQ, Brady J, Zhang J, Zimmerman H, Kaleko M et al. A synthetic Rev-independent bovine immunodeficiency virus-based packaging construct. Hum Gene Ther 2004; 15: 865–877.
Bainbridge JW, Stephens C, Parsley K, Demaison C, Halfyard A, Thrasher AJ et al. In vivo gene transfer to the mouse eye using an HIV-based lentiviral vector; efficient long-term transduction of corneal endothelium and retinal pigment epithelium. Gene Ther 2001; 8: 1665–1668.
Poeschla EM . Non-primate lentiviral vectors. Curr Opin Mol Ther 2003; 5: 529–540.
Saenz DT, Loewen N, Peretz M, Whitwam T, Barraza R, Howell KG et al. Unintegrated lentivirus DNA persistence and accessibility to expression in nondividing cells: analysis with class I integrase mutants. J Virol 2004; 78: 2906–2920.
Ishikawa H, Takano M, Matsumoto N, Sawada H, Ide C, Mimura O et al. Effect of GDNF gene transfer into axotomized retinal ganglion cells using in vivo electroporation with a contact lens-type electrode. Gene Ther 2005; 12: 289–298.
Matsuda T, Cepko CL . Electroporation and RNA interference in the rodent retina in vivo and in vitro. Proc Natl Acad Sci U S A 2004; 101: 16–22.
Chalberg TW, Genise HL, Vollrath D, Calos MP . phiC31 integrase confers genomic integration and long-term transgene expression in rat retina. Invest Ophthalmol Vis Sci 2005; 46: 2140–2146.
Folliot S, Briot D, Conrath H, Provost N, Cherel Y, Moullier P et al. Sustained tetracycline-regulated transgene expression in vivo in rat retinal ganglion cells using a single type 2 adeno-associated viral vector. J Gene Med 2003; 5: 493–501.
Lebherz C, Auricchio A, Maguire AM, Rivera VM, Tang W, Grant RL et al. Long-term inducible gene expression in the eye via adeno-associated virus gene transfer in nonhuman primates. Hum Gene Ther 2005; 16: 178–186.
Smith JR, Verwaerde C, Rolling F, Naud MC, Delanoye A, Thillaye-Goldenberg B et al. Tetracycline-inducible viral interleukin-10 intraocular gene transfer, using adeno-associated virus in experimental autoimmune uveoretinitis. Hum Gene Ther 2005; 16: 1037–1046.
Bainbridge JW, Mistry A, Binley K, de AM, Thrasher AJ, Naylor S et al. Hypoxia-regulated transgene expression in experimental retinal and choroidal neovascularization. Gene Ther 2003; 10: 1049–1054.
Schlichtenbrede FC, da CL, Stephens C, Smith AJ, Georgiadis A, Thrasher AJ et al. Long-term evaluation of retinal function in Prph2Rd2/Rd2 mice following AAV-mediated gene replacement therapy. J Gene Med 2003; 5: 757–764.
Pawlyk BS, Smith AJ, Buch PK, Adamian M, Hong DH, Sandberg MA et al. Gene replacement therapy rescues photoreceptor degeneration in a murine model of Leber congenital amaurosis lacking RPGRIP. Invest Ophthalmol Vis Sci 2005; 46: 3039–3045.
Zeng Y, Takada Y, Kjellstrom S, Hiriyanna K, Tanikawa A, Wawrousek E et al. RS-1 Gene Delivery to an Adult Rs1h Knockout Mouse Model Restores ERG b-Wave with Reversal of the Electronegative Waveform of X-Linked Retinoschisis. Invest Ophthalmol Vis Sci 2004; 45: 3279–3285.
Smith AJ, Schlichtenbrede FC, Tschernutter M, Bainbridge JW, Thrasher AJ, Ali RR . AAV-Mediated gene transfer slows photoreceptor loss in the RCS rat model of retinitis pigmentosa. Mol Ther 2003; 8: 188–195.
Surace EM, Domenici L, Cortese K, Cotugno G, Di VU, Venturi C et al. Amelioration of both functional and morphological abnormalities in the retina of a mouse model of ocular albinism following AAV-mediated gene transfer. Mol Ther 2005; 12: 652–658.
Dejneka NS, Surace EM, Aleman TS, Cideciyan AV, Lyubarsky A, Savchenko A et al. In utero gene therapy rescues vision in a murine model of congenital blindness. Mol Ther 2004; 9: 182–188.
Narfstrom K, Katz ML, Ford M, Redmond TM, Rakoczy E, Bragadottir R . In vivo gene therapy in young and adult RPE65-/- dogs produces long-term visual improvement. J Hered 2003; 94: 31–37.
Narfstrom K, Katz ML, Bragadottir R, Seeliger M, Boulanger A, Redmond TM et al. Functional and structural recovery of the retina after gene therapy in the RPE65 null mutation dog. Invest Ophthalmol Vis Sci 2003; 44: 1663–1672.
Hennig AK, Ogilvie JM, Ohlemiller KK, Timmers AM, Hauswirth WW, Sands MS . AAV-mediated intravitreal gene therapy reduces lysosomal storage in the retinal pigmented epithelium and improves retinal function in adult MPS VII mice. Mol Ther 2004; 10: 106–116.
Gorbatyuk MS, Pang JJ, Thomas Jr J, Hauswirth WW, Lewin AS . Knockdown of wild-type mouse rhodopsin using an AAV vectored ribozyme as part of an RNA replacement approach. Mol Vis 2005; 11: 648–656.
Lingor P, Koeberle P, Kugler S, Bahr M . Down-regulation of apoptosis mediators by RNAi inhibits axotomy-induced retinal ganglion cell death in vivo. Brain 2005; 128: 550–558.
Buch PK, MacLaren RE, Duran Y, Balaggan KS, MacNeil A, Schlichtenbrede FC et al. In contrast to AAV-mediated CNTF expression, AAV-mediated GDNF expression ehnances gene replacement therapy in rodent models of retinal degeneration. Mol Ther (in press).
Schlichtenbrede FC, MacNeil A, Bainbridge JW, Tschernutter M, Thrasher AJ, Smith AJ et al. Intraocular gene delivery of ciliary neurotrophic factor results in significant loss of retinal function in normal mice and in the Prph2Rd2/Rd2 model of retinal degeneration. Gene Ther 2003; 10: 523–527.
Rota R, Riccioni T, Zaccarini M, Lamartina S, Gallo AD, Fusco A et al. Marked inhibition of retinal neovascularization in rats following soluble-flt-1 gene transfer. J Gene Med 2004; 6: 992–1002.
Rakoczy PE, Brankov M, Fonceca A, Zaknich T, Rae BC, Lai CM . Enhanced recombinant adeno-associated virus-mediated vascular endothelial growth factor expression in the adult mouse retina: a potential model for diabetic retinopathy. Diabetes 2003; 52: 857–863.
Deng WT, Yan Z, Dinculescu A, Pang J, Teusner JT, Cortez NG et al. Adeno-associated virus-mediated expression of vascular endothelial growth factor peptides inhibits retinal neovascularization in a mouse model of oxygen-induced retinopathy. Hum Gene Ther 2005; 16: 1247–1254.
Lai CC, Wu WC, Chen SL, Sun MH, Xiao X, Ma L et al. Recombinant adeno-associated virus vector expressing angiostatin inhibits preretinal neovascularization in adult rats. Ophthalmic Res 2005; 37: 50–56.
Gehlbach P, Demetriades AM, Yamamoto S, Deering T, Duh EJ, Yang HS et al. Periocular injection of an adenoviral vector encoding pigment epithelium-derived factor inhibits choroidal neovascularization. Gene Ther 2003; 10: 637–646.
Saishin Y, Silva RL, Saishin Y, Kachi S, Aslam S, Gong YY et al. Periocular gene transfer of pigment epithelium-derived factor inhibits choroidal neovascularization in a human-sized eye. Hum Gene Ther 2005; 16: 473–478.
Reich SJ, Fosnot J, Kuroki A, Tang W, Yang X, Maguire AM et al. Small interfering RNA (siRNA) targeting VEGF effectively inhibits ocular neovascularization in a mouse model. Mol Vis 2003; 9: 210–216.
Campochiaro PA . Ocular neovascularisation and excessive vascular permeability. Expert Opin Biol Ther 2004; 4: 1395–1402.
Verwaerde C, Naud MC, Delanoye A, Wood M, Thillaye-Goldenberg B, Auriault C et al. Ocular transfer of retinal glial cells transduced ex vivo with adenovirus expressing viral IL-10 or CTLA4-Ig inhibits experimental autoimmune uveoretinitis. Gene Ther 2003; 10: 1970–1981.
Broderick CA, Smith AJ, Balaggan KS, Georgiadis A, Buch PK, Trittibach PC et al. Local administration of an adeno-associated viral vector expressing IL-10 reduces monocyte infiltration and subsequent photoreceptor damage during experimental autoimmune uveitis. Mol Ther 2005; 12: 369–373.
van Adel BA, Kostic C, Deglon N, Ball AK, Arsenijevic Y . Delivery of ciliary neurotrophic factor via lentiviral-mediated transfer protects axotomized retinal ganglion cells for an extended period of time. Hum Gene Ther 2003; 14: 103–115.
Martin KR, Quigley HA, Zack DJ, Levkovitch-Verbin H, Kielczewski J, Valenta D et al. Gene therapy with brain-derived neurotrophic factor as a protection: retinal ganglion cells in a rat glaucoma model. Invest Ophthalmol Vis Sci 2003; 44: 4357–4365.
Zhou Y, Pernet V, Hauswirth WW, Di PA . Activation of the extracellular signal-regulated kinase 1/2 pathway by AAV gene transfer protects retinal ganglion cells in glaucoma. Mol Ther 2005; 12: 402–412.
Carlson EC, Liu CY, Yang X, Gregory M, Ksander B, Drazba J et al. In vivo gene delivery and visualization of corneal stromal cells using an adenoviral vector and keratocyte-specific promoter. Invest Ophthalmol Vis Sci 2004; 45: 2194–2200.
Acknowledgements
James Bainbridge is a Wellcome Advanced Fellow (Grant 074617/Z/04/Z); Mei Hong Tan is supported by the UK Department of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bainbridge, J., Tan, M. & Ali, R. Gene therapy progress and prospects: the eye. Gene Ther 13, 1191–1197 (2006). https://doi.org/10.1038/sj.gt.3302812
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302812
Keywords
This article is cited by
-
Retinal gene therapy: an eye-opener of the 21st century
Gene Therapy (2021)
-
Peripheral blood-derived monocytes show neuronal properties and integration in immune-deficient rd1 mouse model upon phenotypic differentiation and induction with retinal growth factors
Stem Cell Research & Therapy (2020)
-
Stem cell therapies for retinal diseases: from bench to bedside
Journal of Molecular Medicine (2020)
-
CAPN5 gene silencing by short hairpin RNA interference
BMC Research Notes (2014)
-
A point mutation in Semaphorin 4A associates with defective endosomal sorting and causes retinal degeneration
Nature Communications (2013)