Abstract
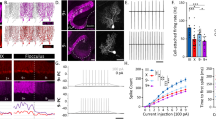
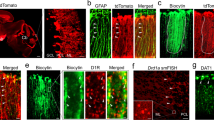
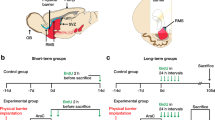
It has often been proposed that one way of replacing degenerating neurons in the brain is to implant embryonic neurons of the same type1. However, in the case of so-called 'point-to-point' systems, as opposed to the 'paracrine' systems which mainly involve local release of neurotransmitter, functional recovery requires a precise re-establishment of the missing circuitry. We recently showed that in one point-to-point system, the cerebellum of adult mice homozygous for the mutation Purkinje cell degeneration (pcd)2, missing Purkinje cells can be replaced by grafting cerebellar primordia from normal mouse embryos3,4. Here, we present studies of the cellular mechanisms underlying this successful replacement. Grafted Purkinje cells leave the graft to migrate along stereotyped pathways to their final position in the deficient molecular layer, where they receive synaptic contacts from adult host neurons. Both the detailed timetable and the precise cellular interactions observed are remarkably similar to those occurring during normal development. Our results suggest that the deficient molecular layer exerts a selective neurotropic effect on neurons of the missing category, and that the embryonic neurons are able to respond to this signal during a period defined by their own internal clock. We also raise the possibility that embryonic Purkinje cells can induce in adult neural cells a new type of plasticity, that of recreating a permissive microenvironment for the integration of embryonic neurons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dunnett, S. B. & Björklund, A. in Neural Grafting in the Mammalian CNS (eds Björklund, A. & Steveni, U.) 673–700 (Elsevier, Amsterdam, 1985).
Mullen, R. J., Eicher, E. M. & Sidman, R. L. Proc. natn. Acad. Sci. U.S.A. 73, 208–212 (1976).
Sotelo, C. Alvarado-Mallart, R. M. Proc. natn. Acad. Sci. U.S.A. 83, 1135–1139 (1986).
Soleto, C. & Alvarado-Mallart, R. M. Neuroscience 20, 1–22 (1987).
Mullen, R. J. Nature 270, 245–247 (1977).
Wassef, M., Simons, J., Tappaz, M. L. & Sotelo, C. Brain Res. 399, 125–135 (1986).
Wasserman, R. H. & Taylor, A. N. Science 152, 791–793 (1961).
Thomasset, M., Desplan, C. & Parkes, C. O. Eur. J. Biochem. 129, 519–524 (1983).
Legrand, Ch., Thomasset, M., Parkes, C. O., Clavel, M. C. & Rabié, A. Cell Tiss. Res. 233, 389–402 (1983).
Wassef, M., Zanetta, J. P., Brehier, A. & Sotelo, C. Devl Biol. 111, 129–137 (1985).
Ramón y Cajal, S. Trab. Lab. Invest. biol. Univ. Mad. 8, 63–135 (1910).
Miale, I. L. & Sidman, R. L. Expl Neurol. 4, 277–296 (1961).
Altman, J. & Bayer, S. Y. J. comp. Neurol. 231, 42–65 (1985).
Rakic, P. J. comp. Neurol. 141, 287–312 (1971).
Sotelo, C. Prog. Brain Res. 48, 149–170 (1978).
Ramón y Cajal, S. Histologie du système nerveux de l'homme et des vertébrés Vol. 2 (Maloine, Paris, 1911).
Larramendi, L. M. H. in Neurobiology of Cerebellar Evolution and Development (ed. Llinás, R.) 803–843 (American Medical Association, Chicago, 1969).
Trenkner, E., Smith, D. & Segil, N. J. Neuroscience 4, 2850–2855 (1984).
Meller, K. & Glees, P. in Neurobiology of Cerebellar Evolution and Development (ed. Llinás, R.) 783–801 (American Medical Association, Chicago, 1969).
Hendelman, W. J. & Aggerwal, A. S. J. comp. Neurol. 193, 1063–1079 (1980).
Pasteels, J. L. et al. Brain Res. 384, 294–303 (1986).
Stemberger, L. A., Hardy, P. H. Jr, Cuculis, J. J. & Meyer, H. G. J. Histochem. Cytochem. 18, 315–333 (1970).
Dupouey, P., Benjelloun, S. & Gomes, D. Devl Neurosci. 7, 81–93 (1985).
Palay, S. L. & Chan-Palay, V. Cerebellar Cortex Cytology and Organization (Springer, Berlin, 1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sotelo, C., Alvarado-Mallart, R. Embryonic and adult neurons interact to allow Purkinje cell replacement in mutant cerebellum. Nature 327, 421–423 (1987). https://doi.org/10.1038/327421a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/327421a0
This article is cited by
-
Consensus Paper: Strengths and Weaknesses of Animal Models of Spinocerebellar Ataxias and Their Clinical Implications
The Cerebellum (2022)
-
Task Force Paper On Cerebellar Transplantation: Are We Ready to Treat Cerebellar Disorders with Cell Therapy?
The Cerebellum (2019)
-
Embryonic Cerebellar Graft Morphology Differs in Two Mouse Models of Cerebellar Degeneration
The Cerebellum (2019)
-
Long-Term Development of Embryonic Cerebellar Grafts in Two Strains of Lurcher Mice
The Cerebellum (2018)
-
An expandable embryonic stem cell-derived Purkinje neuron progenitor population that exhibits in vivo maturation in the adult mouse cerebellum
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.