Abstract
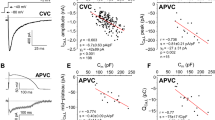
Repolarization of the action potential in squid axon1 and several types of neurones2–4 involves a voltage-activated potassium (K+) current. Voltage clamp analysis has demonstrated that this current has rapid activation kinetics1,3–5. In several neuronal types, the same technique has also revealed a slowly activated K+ current that is calcium (Ca2+)-sensitive3,5–10. This slow Ca2+-activated K+ current is the major current underlying the late, slower portion of the after-hyperpolarization following an action potential11–14. In several muscle types, fast, transient Ca2+-dependent K+ currents have been described15–17 which may contribute to repolarization of the action potential. Rapidly activating, Ca2+-dependent K+ currents have been observed in sympathetic neurones of the bullfrog and it has been suggested that they contribute to action potential repolarization of those neurones8,9,18. We have studied the membrane currents in bullfrog sympathetic neurones using voltage clamp methods and report here a transient outward current that appears to be composed of two separate currents. One of those currents is a transient, Ca2+-sensitive outward current as indicated by a significant reduction of the current by treatments that reduce or block Ca2+ entry (Mn2+, Cd2+, Co2+, Mg2+ or Ca2+ -free Ringer). Such treatments also decreased the rate of action potential repolarization. The results suggest that this current is involved in repolarization of the action potential and consequently may regulate Ca2+ entry into the neurone during spike activity.
This is a preview of subscription content, access via your institution
Access options
Similar content being viewed by others
References
Hodgkin, A. L. & Huxley, A. F. J. Physiol., Lond. 117, 500–544 (1952).
Aldrich, R. W., Getting, P. A. & Thompson, S. H. J. Physiol., Lond. 291, 531–544 (1979).
Barrett, E. F., Barrett, J. N. & Crill, W. E. J. Physiol., Lond. 304, 251–276 (1980).
Connor, J. A. & Stevens, C. F. J. Physiol., Lond. 213, 31–53 (1971).
Meech, R. W. & Standen, N. B. J. Physiol., Lond. 249, 211–239 (1975).
Thompson, S. H. J. Physiol., Lond. 265, 465–488 (1977).
MacDermott, A. B. & Weight, F. F. Fedn Proc. 39, 2074 (1980).
Constanti, A., Adams, P. R. & Brown, D. A. Soc. Neurosci. Abstr. 7, 14 (1981).
Weight, F. F. & MacDermott, A. B. in Physiology and Pharmacology of Epileptogenic Phenomena (eds Klee, M. R., Lux, H. D. & Speckmann, E. J.) 227–233 (Raven, New York, 1982).
Adams, P. R., Brown, D. A. & Constanti, A. in Physiology and Pharmacology of Epileptogenic Phenomena (eds Klee, M. R., Lux, H. D. & Speckmann, E. J.) 175–187 (Raven, New York, 1982).
Minota, S. Jap. J. Physiol. 24, 501–512 (1974).
Barrett, E. F. & Barrett, J. N. J. Physiol., Lond. 255, 737–774 (1976).
Busis, N. A. & Weight, F. F. Nature 263, 434–436 (1976).
McAfee, D. A. & Yarowsky, P. J. J. Physiol., Lond. 290, 507–523 (1979).
Vassort, G. J. Physiol., Lond. 252, 713–734 (1975).
Mounier, Y. & Vassort, G. J. Physiol., Lond. 251, 609–625 (1975).
Siegelbaum, S. A. & Tsien, R. W. J. Physiol., Lond. 299, 485–506 (1980).
Adams, P. R., Constanti, A., Brown, D. A. & Clark, R. B. Nature 296, 746–749 (1982).
MacDermott, A. B., Connor, E. A., Dionne, V. E. & Parsons, R. L. J. gen. Physiol. 75, 39–60 (1980).
Connor, J. A. & Stevens, C. F. J. Physiol., Lond. 213, 21–30 (1971).
Hagiwara, S. & Byerly, L. A. Rev. Neurosci. 4, 69–125 (1981).
Smith, S. J., MacDermott, A. B. & Weight, F. F. Neurosci. Abstr. 7, 15 (1981).
Coraboeuf, E. & Carmeliet, E. Pflügers Arch. ges. Physiol. 392, 352–359 (1982).
Aldrich, R. W., Getting, P. A. & Thompson, S. H. J. Physiol., Lond. 291, 507–530 (1979).
Hermann, A. & Gorman, A. L. F. J. gen. Physiol. 78, 87–110 (1981).
Meech, R. W. & Standen, N. B. J. Physiol., Lond. 249, 231–239 (1975).
Connor, J. A. J. Physiol., Lond. 286, 41–60 (1979).
Meech, R. W. Ann. Rev. Biophy. Bioengng 7, 1–18 (1978).
Armstrong, C. M. & Taylor, S. R. Biophys. J. 30, 473–488 (1980).
Eaton, D. C. & Brodwick, M. S. J. gen. Physiol. 75, 727–750 (1980).
Constanti, A., Adams, P. R. & Brown, D. A. Brain Res. 206, 244–250 (1981).
Kenyon, J. L. & Gibbons, W. R. J. gen. Physiol. 73, 117–138 (1979).
Schwindt, P. C. & Crill, W. E. J. Neurophysiol. 46, 1–16 (1981).
Adams, P. R. in Advances in Physiological Sciences Vol. 4 (ed. Salanki, J.) 135–138 (Pergamon, New York, 1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MacDermott, A., Weight, F. Action potential repolarization may involve a transient, Ca2+ -sensitive outward current in a vertebrate neurone. Nature 300, 185–188 (1982). https://doi.org/10.1038/300185a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/300185a0
This article is cited by
-
T-type channels buddy up
Pflügers Archiv - European Journal of Physiology (2014)
-
Somato-dendritic mechanisms underlying the electrophysiological properties of hypothalamic magnocellular neuroendocrine cells: A multicompartmental model study
Journal of Computational Neuroscience (2007)
-
A transient outward current dependent on external calcium in rat cerebellar granule cells
The Journal of Membrane Biology (1991)
-
A rapidly inactivating Ca2+-dependent K+ current in pheochromocytoma cells (PC12) of the rat
Pfl�gers Archiv European Journal of Physiology (1990)
-
Ca2+-activated K+ current generating the after-hyperpolarization in mammalian nonmyelinated nerve fibres
Pfl�gers Archiv European Journal of Physiology (1989)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.