Abstract
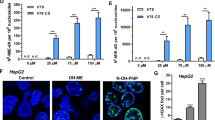
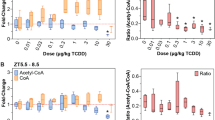
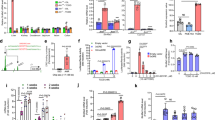
The intense interest in the metabolic fate of epoxidized xeno-biotics is due to several factors. For instance, epoxides are often intermediates in the lipophile to hydrophile conversions necessary for the excretion of olefinic and aromatic compounds by living systems1, and are widely encountered in man's diet from both natural and man–made sources. Some of these epoxidized compounds may alkylate proteins and nucleic acids and thus include some of the most potent cytotoxins, mutagens and carcinogens known2. In mammals, epoxides may rearrange, deoxygenate to olefins, react with glutathione to form conjugates, or be hydrolysed by water to yield 1,2–diols with or without enzymatic catalysis1,3,4,. The enzymes which catalyse the formation of diols are known as epoxide hydrolases (EC 3.3.2.3), and their subcellular distribution is the subject of this report. Early data showed that styrene oxide hydrolase activity was associated with the microsomal subcellular fraction5. Epoxide hydrolase activity was subsequently demonstrated on the nuclear6, Golgi apparatus and plasma membranes7, and in the cytosol of the cell8,9, leaving the mitochondria as the last major cellular organelle assumed to be devoid of epoxide hydrolase activity. We now report strong evidence for the occurrence of substantial epoxide hydrolase activity in the mitochondria.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Oesch, F. Xenobiotica 3, 305–340 (1973).
Miller, J. A. & Miller, E. C. in Chemical Carcinogenesis Pt A (eds Ts'o, P. O. P. & DiPaolo, V. A.) 61–85 (Dekker, New York, 1974).
Ivie, G. W. Science 191, 959–961 (1976).
Jakoby, W. B. Adv. Enzym. 46, 383–414 (1978).
Oesch, F., Jerina, D. M. & Daly, J. Biochim. biophys. Acta 227, 685–691 (1971).
Mukhtar, H., Elmamlouk, T. H. & Bend, J. R. Archs Biochem. Biophys. 192, 10–21 (1979).
Stasiecki, P., Oesch, F., Bruder, G., Jarasch, E.-D. & Franke, W. W. Eur. J. Cell Biol. 21, 79–92 (1980).
Gill, S. S., Hammock, B. D. & Casida, J. E. J. agric. Fd Chem. 22, 386–395 (1974).
Hammock, B. D., Gill, S. S., Stamoudis, V. & Gilbert, L. I. Comp. Biochem. Physiol. 53 B, 263–265 (1976).
Gill, S. S. & Hammock, B. D. Biochem. Pharmac. 29, 389–395 (1980).
Gill, S. S. & Hammock, B. D. Biochem. biophys. Res. Commun. 89, 965–971 (1979).
Jerina, D. M., Dansette, P. M., Lu, A. Y. H. & Levin, W. Molec. Pharmac. 13, 342–351 (1977).
Mullin, C. A. & Hammock, B. D. Analyt. Biochem. 106, 476–485 (1980).
Oesch, F. & Bentley, P. Nature 259, 53–55 (1976).
Leighton, F. et al. J. Cell Biol. 37, 482–513 (1968).
Sottocassa, G. L., Kuylenstierna, B., Ernster, L. & Bergstrand, A. J. Cell Biol. 32, 415–438 (1967).
Omura, T. & Sato, R. J. biol. Chem. 239, 2370–2378 (1964).
Shepherd, D. & Garland, P. B. Meth. Enzym. 13, 11–16 (1969).
Bergmeyer, H. U. Methods of Enzymatic Analysis Vols 1, 2 (Academic, New York, 1974).
Kalckar, H. M. J. biol. Chem. 167, 461–475 (1947).
Ames, B. N., McCann, J. & Yamasaki, E. Mutat. Res. 31, 347–364 (1975).
Hammock, B. D., Gill, S. S., Mumby, S. M. & Ota, K. in Molecular Basis of Environmental Toxicity (ed. Bhatnagar, R. S.) 229–272 (Ann Arbor Science, Michigan, 1980).
Allen, J. A. & Coombs, M. M. Nature 287, 244–245 (1980).
Backer, J. M. & Weinstein, I. B. Science 209, 297–299 (1980).
Sato, R., Atsuta, Y., Imai, Y., Taniguchi, S. & Okuda, R. Proc. natn. Acad. Sci. U.S.A. 74, 5477–5481 (1977).
Raw, I. Biochem. biophys. Res. Commun. 81, 1294–1297 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gill, S., Hammock, B. Epoxide hydrolase activity in the mitochondrial fraction of mouse liver. Nature 291, 167–168 (1981). https://doi.org/10.1038/291167a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/291167a0
This article is cited by
-
Cytosolic epoxide hydrolase in fetal and adult human liver
Archives of Toxicology (1983)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.