Abstract
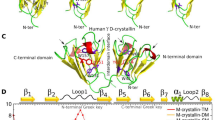
The three-dimensional structure of the eye lens protein, bovine γ-crystallin II, has been determined at 2.6 Å resolution. The protein has a two domain β-structure, folded into four remarkably similar ‘Greek key’ motifs, and shows the highest internal symmetry of any protein studied by X-ray analysis. Although the symmetrical structure seems very stable, the arrangement of the sulphydryl groups would allow intramolecular cross-linking leading to possible destabilization, and intermodular cross-linking leading to aggregation, both of which may be important in cataract formation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Van der Ouderaa, F. J., de Jong, W. W. & Bloemendal, H. Eur. J. Biochem. 39, 207–222 (1973).
Van der Ouderaa, F. J., de Jong, W. W., Hilderink, A. & Bloemendal, H. Eur. J. Biochem. 49, 157–168 (1974).
Spector, A. & Katz, E. J. biol. Chem. 240, 1979–1985 (1965).
Van Kleef, F. S. M., de Jong, W. W. & Hoenders, H. J. Nature 258, 264–266 (1975).
Siezen, R. J., Bindels, J. G. & Hoenders, H. J. Eur. J. Biochem. 91, 387–396 (1978).
Stauffer, J., Rothschild, C., Wandel, T. & Spector, A. Invest. Ophthalmol. Vis. Sci. 13, 135–146 (1974).
Herbrink, P., Van Westreenen, H. & Bloemendal, H. Expl Eye Res. 20, 541–548 (1975).
Asselbergs, F. A. M., Koopmans, M., Van Venrooij, W. J. & Bloemendal, H. Expl Eye Res. 28, 223–228 (1979).
Bloemendal, H. & Herbrink, P. Ophthal. Res. 6, 81–92 (1974).
Björk, I. Expl Eye Res. 9, 152–157 (1970).
Croft, L. R. Biochem. J. 128, 961–970 (1972).
Slingsby, C. & Croft, L. R. Expl Eye Res. 26, 291–304 (1978).
Driessen, H. P. C., Herbrink, P., Bloemendal, H. & de Jong, W. W. Expl Eye Res. 31, 243–246 (1980).
Philipson, B. T. & Fagerholm, P. P. Ciba Fdn Symp. 19, 45–63 (1973).
Jedziniak, J. A., Nicoli, D. F., Baram, H. & Benedek, G. B. Invest. Ophthalmol. Vis. Sci. 17, 51–57 (1978).
Spector, A. et al. Science 204, 1323–1326 (1979).
Anderson, E. I. & Spector, A. Expl Eye Res. 26, 407–417 (1978).
Takemoto, L. J. & Azari, P. Expl Eye Res. 23, 1–7 (1976).
Truscott, R. J. W. & Augusteyn, R. C. Expl Eye Res. 25, 139–148 (1977).
Spector, A. & Roy, D. Proc. natn. Acad. Sci. U.S.A. 75, 3244–3246 (1978).
Harding, J. J. & Dilley, K. J. Expl Eye Res. 22, 1–73 (1976).
Richardson, J. S. Nature 268, 495–500 (1977).
Björk, I. Expl Eye Res. 1, 145–154 (1961).
Van Dam, A. F. Expl Eye Res. 5, 255–266 (1966).
Björk, I. Expl Eye Res. 3, 254–261 (1964).
Carlisle, C. H., Lindley, P. F., Moss, D. S. & Slingsby, C. J. molec. Biol. 110, 417–419 (1977).
Blundell, T. L. et al. Acta crystallogr. 34, 3653–3657 (1978).
Tickle, I. J. Acta crystallogr. 31, 329–331 (1975).
North, A. C. T., Phillips, D. C. & Mathews, F. S. Acta crystallogr. 24, 351–359 (1968).
Blundell, T. L. & Johnson, L. N. Protein Crystallography (Academic, London, 1976).
Blow, D. M. & Crick, F. C. Acta crystallogr. 12, 794–802 (1959).
Richards, F. M. J. molec. Biol. 37, 225–230 (1968).
Jones, T. A. J. appl. Crystallogr. 11, 268–272 (1978).
Hermans, J. & McQueen, J. E. Acta crystallogr. 30, 730–739 (1974).
McLachlan, A. D. J. molec. Biol. 128, 49–80 (1979).
Walker, J. E., Wonacott, A. J. & Harris, J. I. Eur. J. Biochem. 108, 581–586 (1980).
Chirgadze, Iu. N., Nikonov, S. V., Garber, M. B. & Reshetnikova, L. S. J. molec. Biol. 110, 619–624 (1977).
Chirgadze, Iu. N., Sergeev, Iu. V., Fomenkova, N. P., Nikonov, S. V. & Lunin, V. Iu. Proc. Acad. Sci. USSR 250, 762–765 (1980).
Fahey, R. C., Hunt, J. S. & Windham, G. C. J. molec. Evol. 10, 155–160 (1977).
East, E. J., Chang, R. C. C., Yu, N. T. & Kuck, J. F. R. Jr. J. biol. Chem. 253, 1436–1441 (1978).
Askren, C. C., Yu, N. T. & Kuck, J. F. R. Expl Eye Res. 29, 647–654 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blundell, T., Lindley, P., Miller, L. et al. The molecular structure and stability of the eye lens: X-ray analysis of γ-crystallin II. Nature 289, 771–777 (1981). https://doi.org/10.1038/289771a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/289771a0
This article is cited by
-
Novel mutations in CRYGC are associated with congenital cataracts in Chinese families
Scientific Reports (2017)
-
Primary sequence contribution to the optical function of the eye lens
Scientific Reports (2014)
-
Explosive Expansion of βγ-Crystallin Genes in the Ancestral Vertebrate
Journal of Molecular Evolution (2010)
-
Evolutionary genetics: Seeing the light: the role of inherited developmental cascades in the origins of vertebrate lenses and their crystallins
Heredity (2006)
-
Vertebrate-like ??-crystallins in the ocular lenses of a copepod
Journal of Comparative Physiology A (2005)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.