Abstract
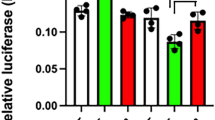
The pituitary hormones corticotropin (ACTH) and β-lipotropin (β-LPH) are formed from a large common precursor1–8. Recently, we have elucidated the whole primary structure of the bovine ACTH-β-LPH precursor (designated alternatively as preproopiocortin) by determining the nucleotide sequence of cloned DNA complementary to the mRNA coding for the precursor protein9. The amino acid sequence assigned has disclosed a characteristic repetitive structure of the ACTH-β-LPH precursor. The repetitive units of the precursor protein each contain a melanotropin (MSH) sequence (α,-β- or γ-MSH) as well as other peptide components such as β-endorphin and corticotropin-like intermediate lobe peptide (CLIP). The repetitive units as well as their peptide components are each bounded by paired basic amino acid residues, which apparently represent the sites of proteolytic processing. Several studies10 have confirmed the translational initiation site and protein structure assigned (see also ref. 11 and refs therein). In view of the recent knowledge about the organization of eukaryotic genes (see refs 12, 13 for reviews), it would be of particular interest to investigate the relationship between the repetitive structure of the ACTH-β-LPH precursor containing different functional components and the arrangement of the protein-coding sequence in its gene. We have now isolated and characterized bovine genomic DNA fragments encoding this precursor protein and have demonstrated that the protein sequence is encoded by two non-consecutive DNA segments. An intron (intervening sequence) of approximately 2.2 kilobase pairs separates the smaller exon (mRNA-coding sequence), which contains the gene sequence encoding the signal peptide, from the larger exon, which contains the gene sequence for most of the protein structure, including the known biologically active component peptides.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nakanishi, S. et al. Proc. natn. Acad. Sci. U.S.A. 73, 4319–323 (1976).
Nakanishi, S., Inoue, A., Taii, S. & Numa, S. FEBS Lett. 84, 105–109 (1977).
Taii, S., Nakanishi, S. & Numa, S. EUT. J. Biochem. 93, 205–212 (1979).
Kita, T., Inoue, A., Nakanishi, S. & Numa, S. Eur. J. Biochem. 93, 213–220 (1979).
Mains, R. E. & Eipper, B. A. J. biol. Chem. 251, 4115–4120 (1976).
Mains, R. E., Eipper, B. A. & Ling, N. Proc. natn. Acad. Sci. U.S.A. 74, 3014–3018 (1977).
Roberts, J. L. & Herbert, E. Proc. natn. Acad. Sci. U.S.A. 74, 4826–4830, 5300–5304 (1977).
Nakanishi, S. et al. Proc. natn. Acad. Sci. U.S.A. 75, 6021–6025 (1978).
Nakanishi, S. et al. Nature 278, 423–427 (1979).
Nakamura, M., Inoue, A., Nakanishi, S. & Numa, S. FEBS Lett. 105, 357–359 (1979).
Numa, S. & Nakanishi, S. Biochem. Soc. Trans. (in the press).
Crick, F. Science 204, 264–271 (1979).
Dawid, I. B. & Wahli, W. Devl Biol. 69, 305–328 (1979).
Southern, E. M. J. molec. Biol. 98, 503–517 (1975).
Benoist, C., O'Hare, K., Breathnach, R. & Chambon, P. Nucleic Acids Res. 8, 127–142 (1980).
Breathnach, R. et al. Proc. natn. Acad. Sci. U.S.A. 75, 4853–4857 (1978).
Cooper, C. W., Peng, T. -C., Obie, J. F. & Garner, S. C. Endocrinology 107, 98–107 (1980).
Blobel, G. & Dobberstein, B. J. Cell Biol. 67, 852–862 (1975).
Gannon, F. et al. Nature 278, 428–434 (1979).
Chang, A. C. Y., Cochet, M. & Cohen, S. N. Proc. natn. Acad. Sci. U.S.A. (in the press).
Polsky, F., Edgell, M. H., Seidman, J. G. & Leder, P. Analyt. Biochem. 87, 397–410 (1978).
Weinstock, R. et al. Proc. natn. Acad. Sci. U.S.A. 75, 1299–1303 (1978).
Jeffreys, A. J. & Flavell, R.A. Cell 12, 429–439 (1977).
Leder, P., Tiemeier, D. & Enquist, L. Science 196, 175–177 (1977).
Blattner, F. R. et al. Science 196, 161–169 (1977).
Benton, W. D. & Davis, R. W. Science 196, 180–182 (1977).
Lacy, E., Hardison, R. C., Quon, D. & Maniatis, T. Cell 18, 1273–1283 (1979).
Maxam, A. M. & Gilbert, W. Meth. Enzym. 65, 499–560 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakanishi, S., Teranishi, Y., Noda, M. et al. The protein-coding sequence of the bovine ACTH-β-LPH precursor gene is split near the signal peptide region. Nature 287, 752–755 (1980). https://doi.org/10.1038/287752a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/287752a0
This article is cited by
-
Epidermal growth factor: Is the precursor a receptor?
Nature (1985)
-
Isolation and structural organization of the human preproenkephalin B gene
Nature (1983)
-
One gene, several messages. From multifunctional proteins to endogenous opiates
Human Genetics (1983)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.