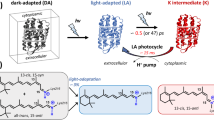
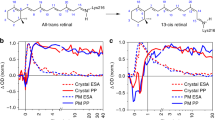
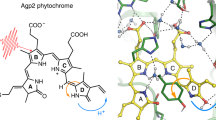
Abstract
The primary photochemical event in the two light-transducing pigments whose chromophore is retinal, rhodopsin or bacteriorhodopsin, is a source of controversy. It was originally proposed that the primary photoevent in the bleaching of rhodopsin is the photoisomerization of the chromophore from 11-cis to all-trans retinal1,2. Photochemical considerations suggested that a photoisomerization is the primary event in both rhodopsin and bacteriorhodopsin3–7. However, this description of bacteriorhodopsin's photochemistry has been questioned8–10. To elucidate this problem, we determined the isomeric conformation of retinal for two of the photolytic intermediates of bacteriorhodopsin, using a method that enables us to extract chromophores from the photocycle intermediates L and M at low temperatures (−74 °C), and have determined the isomeric conformation of the extracted retinals by HPLC11,12. Here we provide direct evidence that isomerization of the chromophore has taken place in two of the early photocycle intermediates (L and M) of bacteriorhodopsin.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kropf, A. & Hubbard, R. Ann. N.Y. Acad. Sci. 74, 266–280 (1958).
Yoshizawa, T. & Wald, G. Nature 197, 1279–1286 (1963).
Rosenfeld, T., Honig, B., Ottolenghi, M., Hurley, J. & Ebrey, T. G. Pure appl. Chem. 49, 341–351 (1977).
Hurley, J., Ebrey, T., Honig, B. & Ottolenghi, M. Nature 270, 540–542 (1977).
Hurley, J., Becher, B. & Ebrey, T. G. Nature 272, 87–88 (1978).
Honig, B., Ebrey, T., Callender, R. H., Dinur, U. & Ottolenghi, M. Proc. natn. Acad. Sci. U.S.A. 76, 2503–2507 (1979).
Mao, B., Ebrey, T. G. & Crouch, R. Biophys. J. 29, 247–250 (1980).
Applebury, M. L., Peters, K. S. & Rentzepis, P. M. Biophys. J. 23, 375–382 (1978).
Lewis, A. Proc. natn. Acad. Sci. U.S.A. 75, 549–553 (1978).
Warshel, A. Proc. natn. Acad. Sci. U.S.A. 75, 2558–2562 (1978).
Pettei, M. J., Yudd, A. P., Nakanishi, K., Henselman, R. & Stoeckenius, W. Biochemistry 16, 1955–1959 (1977).
Tsuda, M. & Ebrey, T. G. Biophys. J. 30, 149–158 (1980).
Stoeckenius, W., Lozier, R. & Bogomolni, R. Biochim. biophys. Acta 505, 212–278 (1979).
Oesterhelt, D. & Hess, B. Eur. J. Biochem. 37, 316–326 (1973).
Maeda, A., Iwasa, T. & Yoshizawa, T. J. Biochem. 82, 1599–1604 (1977).
Ohno, K., Takeuchi, Y. & Yoshida, M. J. Biochem. 82, 1177–1180 (1977).
Sperling, W., Carl, P., Rafferty, Ch.N. & Dencher, N. A. Biophys. struct. Mech. 3, 79–94 (1977).
Aton, B., Doukas, A. G., Callender, R. H., Becher, B. & Ebrey, T. G. Biochim. biophys. Acta 576, 424–428 (1979).
Tokunaga, F., Iwasa, T. & Yoshizawa, T. FEBS Lett. 72, 33–38 (1976).
Becher, B., Tokunaga, F. & Ebrey, T. G. Biochemistry 17, 2293–2300 (1978).
Iwasa, T., Tokunaga, F. & Yoshizawa, T. Biophys. struct. Mech. (in the press).
Aton, B., Doukas, A. G., Callender, R. H., Becher, B. & Ebrey, T. G. Biochemistry 16, 2995–2999 (1979).
Stockburger, M., Klosmann, W., Guttermann, H., Massing, G. & Peters, R. Biochemistry 18, 4886–4900 (1979).
Hubbard, R. J. Am. chem. Soc. 78, 4662–4665 (1965).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsuda, M., Glaccum, M., Nelson, B. et al. Light isomerizes the chromophore of bacteriorhodopsin. Nature 287, 351–353 (1980). https://doi.org/10.1038/287351a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/287351a0
This article is cited by
-
Opn5L1 is a retinal receptor that behaves as a reverse and self-regenerating photoreceptor
Nature Communications (2018)
-
Age-Dependent Differences in Recovered Visual Responses in Royal College of Surgeons Rats Transduced with the Channelrhodopsin-2 Gene
Journal of Molecular Neuroscience (2012)
-
Channelrhodopsins provide a breakthrough insight into strategies for curing blindness
Journal of Genetics (2009)
-
Photocycle of 13-cis-retinal in bacteriorhodopsin caused by destruction to the cooperative effect of purple membrane
Science in China Series B: Chemistry (1998)
-
A unifying concept for ion translocation by retinal proteins
Journal of Bioenergetics and Biomembranes (1992)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.