Abstract
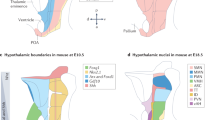
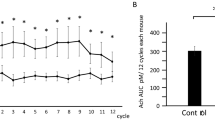
The Thy-1 differentiation antigen on the cell surfaces of mouse and rat thymocytes, lymphocytes1–4, epidermis5, developing skeletal muscle6, and brain1,3,4 has not yet been assigned a function. In rat and mouse brain, Thy-1 reaches adult levels at about 3 weeks of age from minimal or variably low levels at birth3,7–9. In vitro studies of fetal or newborn cerebellar neurons have shown that granule cells and some large multipolar cells become Thy-1 positive during culture10,11. All dorsal root ganglion neurones in culture express Thy-111,12. The antigen appears to be enriched in synaptosomal fractions13,14 but Thy-1 has recently been demonstrated on the surface and dendrites of microdissected neurons of adult rat brain15. In the brain Thy-1 has been thought to be a neuronal antigen, but some cerebellar astrocytes become Thy-1 positive after prolonged culture16. The transient or developed occurrence of the Thy-1 glycoprotein on selected cell surfaces would suggest that it mediates an event associated either with differentiation or with a differentiated function. The recent report by Dulbecco et al. that monoclonal hybridoma antibody against Thy-1.1 (aThy-1.1) blocks a differentiation event in culture of a rat mammary carcinoma cell line17 is, the first demonstration of a functional link between the molecule and a specific developmental process. We consider brain Thy-1 to be a differentiated adult expression, however, and we report here that injecting antibody to Thy-1 antigen into the rat hypothalamus caused selective inhibition of carbamyl choline induced drinking.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Reif, A. E. & Allen, J. M. V. J. exp. Med. 120, 413–433 (1964).
Raff, M. C. Immunology 19, 637–650 (1970).
Douglas, T. C. J. exp. Med. 136, 1054–1062 (1972).
Acton, R. T., Morris, R. J. & Williams, A. F. Eur. J. Immun. 4, 598–602 (1974).
Scheid, M., Boyse, E. A., Carswell, E. A. & Old, L. J. J. exp. Med. 135, 938 (1972).
Lesley, J. F. & Lennon, V. A. Nature 268, 163–165 (1977).
Schachner, M. & Hammerling, U. Brain Res. 73, 362–371 (1974).
Zwerner, R. K., Acton, R. T. & Seeds, N. W. Devl Biol. 60, 331–335 (1977).
Barclay, A. N. J. Neurochem. 32, 1249–1257 (19641979).
Mirsky, R. & Thompson, E. J. Cell 4, 95–101 (1975).
Fields, K. L. Curr. Topics dev. Biol. 13 (in the press).
Fields, K. L., Brockes, J. P., Mirsky, R. & Wendon, L. M. B. Cell 14, 43–51 (1978).
Stohl, W. & Gonatas, N. K. J. Immun. 119, 422–427 (1977).
Acton, R. T., Addis, J., Carl, G. F., McClain, L. D. & Bridgers, W. F. Proc. natn. Acad. Sci. U.S.A. 75, 1848–1852 (1979).
Barclay, A. N. & Hyden, H. J. Neurochem. 32, 1583–1586 (1979).
Pruss, R. Nature 280, 688–690 (1979).
Dulbecco, R., Bologna, M. and Unger, M. Proc. natn Acad. Sci. U.S.A. 76, 1846–1852 (1979).
Grossman, S. P. Science 132, 301–303 (1960).
Fisher, A. E. & Courey, J. N. Science 138, 691–693 (1962).
Slangen, J. L. & Miller, N. E. Physiol. Behav. 4, 543–552 (1969).
Leibowitz, S. F. Physiol. Behav. 14, 731–742 (1975).
Clark, E. A., Lake, P., Khorshidi, M. & Sunshine, G. Eur. J. Immun. (in the press).
Barclay, A. N., Letarte-Muirhead, M., Williams, A. F. & Faulkes, R. A. Nature 263, 563–567 (1976).
Williams, A. F., Barclay, A. N., Letarte-Muirhead, M. & Morris, R. J. Cold Spring Harb. Symp. quant. Biol. 41, 51–61 (1977).
McClain, L. D., Tomana, M. & Acton, R. T. Brain Res. 159, 161–171 (1978).
Barclay, A. N. & Hyden, H. J. Neurochem. 31, 1375–1391 (1978).
Haglid, K. G. et al. Brain Res. 168, 119–132 (19791979).
Schupf, N., Jaehn, B. & Williams, C. A. (in preparation).
Rapport, M. M., Graf, L., Huang, Y. & Yu, R. K. Fedn Proc. 37, 1655 (1978).
Goding, J. W. J. immun. Meth. 13, 215–226 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Williams, C., Barna, J. & Schupf, N. Antibody to Thy-1 antigen injected into rat hypothalamus selectively inhibits carbamyl choline induced drinking. Nature 283, 82–84 (1980). https://doi.org/10.1038/283082a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/283082a0
This article is cited by
-
Ability of some amino acids incorporated into protein to stimulate the thymus-dependent immune response
Bulletin of Experimental Biology and Medicine (1986)
-
Serological survey of T-lymphocyte differentiation antigens in wild mice
Immunogenetics (1985)
-
The distribution of Thy-1 antigen in the P.N.S. of the adult rat
Journal of Neurocytology (1983)
-
Effect of thymectomy and thymus polypeptide factor on instrumental reflex formation to food in rats
Bulletin of Experimental Biology and Medicine (1982)
-
Biological effects of antibodies to lymphocyte surface receptors
Springer Seminars in Immunopathology (1982)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.