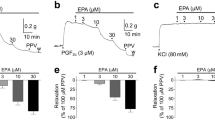
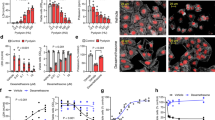
Abstract
ASPIRIN prevents prostaglandin (PG) generation by directly inhibiting the cyclo-oxygenase enzyme responsible for PG biosynthesis1–3. In addition, there is now conclusive evidence that anti-inflammatory steroids can also prevent PG generation4–13. Unlike the aspirin-like drugs, steroids have no anti-cyclo-oxygenase activity but exert their action by preventing the release from phospholipids of the fatty acid substrates required for PG biosynthesis4–9,12,13. We have shown that stimulation of thromboxane A2 (TXA2) release by agents such as histamine, 5-hydroxytryptamine and rabbit aorta contracting substance-releasing factor (RCS–RF) (but not arachidonic acid) is inhibited by anti-inflammatory steroids, and that their potency in this action closely parallels their anti-inflammatory activity12,13. Furthermore, their mechanism of action involves the inhibition of phospholipase A2 activity, and thus of arachidonate release within the lung12,13. In several other tissues, the mechanism of steroid hormone action depends on the combination of thesteroid with a cytosolic receptor protein, the translocation of this drug–receptor complex to the nucleus and the initiation of protein biosynthesis14–16. We now show that similar events occur in the guinea pig perfused lung before inhibition by steroids of phospholipase A2 activity (and thus TXA2 generation). We have discovered a steroid-induced factor which mimics the anti-phospholipase effects of these anti-inflammatory agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Vane, J. R. Nature new Biol. 231, 232–235 (1971).
Smith, J. B. & Willis, A. L. Nature new Biol. 231, 235–237 (1971).
Ferreira, S. H., Moncada, S. & Vane, J. R. Nature new Biol. 231, 237–239 (1971).
Gryglewski, R. J., Panczenko, B., Korbut, R., Grodzinska, A. & Oczekiewicz, A. Prostaglandins 10, 343–355 (1975).
Tashjian, A. H., Voekel, E. F., McDonough, J. & Levine, L. Nature 258, 739–741 (1976).
Kantrowitz, F., Robinson, D. W., McGuire, M. B. & Levine, L. Nature 258, 737–739 (1976).
Floman, N. & Zor, U. Invest. Opthal. visual Sci. 16, 69–73 (1977).
Hong, S-C. I. & Levine, L. Proc. natn. Acad. Sci. U.S.A. 73, 1730–1734 (1976).
Tam, S., Hong, S-C. L. & Levine, L. J. Pharm. exp. Ther. 203, 162–168 (1977).
Lewis, G. P. & Piper, P. J. Nature 254, 308–311 (1975).
Hammarstrom, S. et al. Science 197, 994–996 (1977).
Nijkamp, F. P., Flower, R. J., Moncada, S. & Vane, J. R. Nature 263, 479–482 (1976).
Blackwell, G. J., Flower, R. J., Nijkamp, F. P. & Vane, J. R. Br. J. Pharmac. 62, 79–89 (1978).
Buller, R. E. & O'Malley, B. W. Biochem. Pharmac. 25, 1–12 (1976).
Chan, L. & O'Malley, B. W. New Engl. J. Med. 294, 1372–1381 (1976).
Rousseau, G. G., Baxter, J. D. & Tomkins, G. M. J. molec. Biol. 67, 99–115 (1972).
Baxter, J. D. & Tomkins, G. M. Proc. natn. Acad. Sci. U.S.A. 68, 932–937 (1971).
Kaiser, N., Milholland, R. J., Turnell, R. W. & Rosen, F. Biochem. biophys. Res. Commun. 49, 516–521 (1972).
Reich, E. Cancer Res. 23, 1428–1441 (1963).
Vazquez, D. FEBS Lett. 40, S63–S84 (1974).
Piper, P. J. & Vane, J. R. Nature 233, 29–35 (1969).
Southern, A. L., Gordon, G. G., Yeh, H. S., Dunn, M. W. & Weinstein, B. I. Science 200, 1177–1178 (1978).
Tsurufuji, S. J. Pharm. Soc. Korea 21, 115–117 (1977).
Danon, A. & Assouline, G. Nature 273, 552–554 (1978).
Ferreira, S. H. & Vane, J. R. A. Rev. Pharmac. 14, 57–73 (1974).
Turner, S. R., Tainer, J. A. & Lynn, W. S. Nature 257, 680–681 (1975).
Kwon, T. W. & Olcott, H. S. Biochim. biophys. Acta 130, 528–531 (1966).
Bach, K. M., Brashler, J. R. & Gorman, R. R. Prostaglandins 14, 21–38 (1977).
Jakschick, B. A., Falkenhein, S. & Parker, C. W. Proc. natn. Acad. Sci. U.S.A. 74, 4577–4581 (1977).
Flower, R. J. Pharmac. Rev. 26, 33–67 (1974).
Drenth, J., Enzing, C. M., Kalk, K. H. & Vessies, J. C. A. in Advances in Prostaglandin and Thromboxane Research Vol. 3 (eds C. Galli et al.) 23–29 (Raven, New York, 1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
FLOWER, R., BLACKWELL, G. Anti-inflammatory steroids induce biosynthesis of a phospholipase A2 inhibitor which prevents prostaglandin generation. Nature 278, 456–459 (1979). https://doi.org/10.1038/278456a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/278456a0
This article is cited by
-
ANXA1 is identified as a key gene associated with high risk and T cell infiltration in primary sclerosing cholangitis
Human Genomics (2023)
-
Annexin A1 is a polarity cue that directs mitotic spindle orientation during mammalian epithelial morphogenesis
Nature Communications (2023)
-
Serratiopeptidase: a statistical approach towards enhancement of fermentation and biomass product recovery
Biomass Conversion and Biorefinery (2022)
-
Low Annexin A1 level in HTLV-1 infected patients is a potential biomarker for the clinical progression and diagnosis of HAM/TSP
BMC Infectious Diseases (2021)
-
Potential Adverse Effects of Dexamethasone Therapy on COVID-19 Patients: Review and Recommendations
Infectious Diseases and Therapy (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.