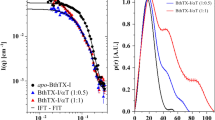
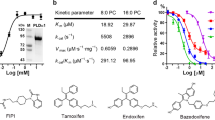
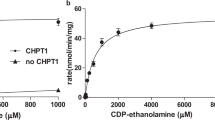
Abstract
PHOSPHOLIPASE A2 (EC 3.1.1.4) catalyses the hydrolysis of the fatty acid ester bond at the 2-position of 1,2 diacyl sn-phosphoglycerides, and has been purified from many sources, such as mammalian pancreas1,2, snake venom3 and bee venom4. In mammals, the enzyme is produced as a precursor, which trypsin can convert into active phospholipase A2 by splitting the peptide bond Arg 7–Ala 8, with the removal of the N-terminal heptapeptide from the molecule5. The activity of the different phospholipases A2 depends on the physical state of the substrate. The enzyme from snake venom is highly active when the substrate is tightly packed in a membrane. Mammalian phospholipases cannot attack membranes; they can degrade the lecithin molecules most effectively when these are more loosely packed in micelles. All phospholipases also have some activity towards substrates in a monomeric solution. The precursors from mammalian pancreas phospholipases are also active towards the monomeric substrate molecules, but inactive to substrates in micellar form6. The primary structure of many of the phospholipases A2 is known. They have a single polypeptide chain of approximately 130 amino acid residues and six or seven disulphide bridges7–10. There is a high degree of homology between all the enzymes. We report here the three-dimensional structure of one of these enzymes, prephospholipase A2 from porcine pancreas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
De Haas, G. H., Postema, N. M., Nieuwenhuizen, W. and Van Deenen, L. L. M. Biochim. biophys. Acta, 159, 103–117 (1968).
Dutilh, C. I., Van Doren, P. J., Verheul, F. E. A .M. and De Haas, G. H. Eur. J. Biochem., 53, 91–97 (1975).
Wells, M. A. Biochemistry, 10, 4074–4078 (1971).
Shipolini, R. A. et al., Eur. J. Biochem., 20, 459–468 (1971).
De Haas, G. H., Postema, N. M., Nieuwenhuizen, W. and Van Deenen, L. L. M. Biochim. biophys. Acta, 159, 118–129 (1968).
Pieterson, W. A., Vidal, J. C., Volwerk, J. J. and De Haas, G. H. Biochemistry 13, 1455–1460 (1974).
De Haas, G. H. et al., Biochim, biophys. Acta, 221, 31–53, 54–61 (1970).
Botes, D. P. and Viljoen, C. C. J. biol. Chem., 249, 3827–3835 (1974).
Joubert, J. J. Eur. J. Biochem., 52, 539–554 (1975).
Halpert, J. and Eaker, D. J. biol. Chem., 250, 6990–6997 (1975).
Volwerk, J. J., Pieterson, W. A. and De Haas, G. H. Biochemistry, 13, 1446–1454 (1974).
Pieterson, W. A., Volwerk, J. J. and De Haas, G. H. Biochemistry, 13, 1439–1445 (1974).
Cotton, F. A., Bier, C. J., Day, V. W., Hazen, E. E. and Larsen, S. Cold Spring Harb. Symp. quant. Biol., 36, 243–249 (1971).
Robertus, J. D., Kraut, J., Alden, R. A. and Birktoft, J. J. Biochemistry, 8, 4293–4303 (1972).
Henderson, R., Wright, C. S., Hess, G. P. and Blow, D. M. Cold Spring Harb. Symp. quant. Biol., 36, 63–70 (1971).
Drenth, J., Swen, H. M. and Kalk, K. H. Biochemistry, 15, 3731–3738 (1976).
Hartsuck, J. A. and Lipscomb, W. N., in The Enzymes, 3, third ed. (edit. by Boyer, P. D.), 1–56 (Academic, New York and London, 1971).
Matthews, B. W. et al., Nature new Biol., 238, 41–43 (1972).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DRENTH, J., ENZING, C., KALK, K. et al. Structure of porcine pancreatic prephospholipase A2. Nature 264, 373–377 (1976). https://doi.org/10.1038/264373a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/264373a0
This article is cited by
-
Tryptophan modification of phospholipase A2 enzymes and presynaptic neurotoxins from snake venoms
Journal of Protein Chemistry (1984)
-
Active site and catalytic mechanism of phospholipase A2
Nature (1981)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.