Abstract
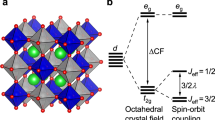
HOMONUCLEAR intervalcncc-transfer absorption of the first transition series cations has aroused considerable interest for two reasons1. First, it may give rise to intense colour which is not predicted by the addition of the colours of either of the individual ions. Second, it may lead in some chain structures to strong anisotropy in electronic conduction. A classic example is vivianite, Fe23+(H2O)8 [PO4]2: although colourless when fresh, upon partial oxidation of the transition metal the compound assumes a deep blue which is at its maximum intensity when the electric vector of polarised light is parallel to the axis of the two iron atoms in the octahedral edge-sharing dimer of the crystal structure. Extensive study on this and on other systems has led to a considerable body of literature1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Allen, G. C., and Hush, N. S., Progress in Inorganic Chemistry, 8, (edit by Cotton F. A.), 357 (Wiley, New York, 1967).
Moore, P. B., Amer. Mineral, 56, 1 (1971).
P. B., Lithos, 4, 93 (1971).
Larsen, E. S., and Berman, H., US geol. Surv. Bull., 848, 31 (1934).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MOORE, P. Evidence for a complete mixed valence solid solution series in Fe23+ (H2O)3 [PO4]2 (phosphoferrite) Fe33+(OH)3 [PO4]2 (kryzhanovskite). Nature 251, 305–306 (1974). https://doi.org/10.1038/251305a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/251305a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.