Abstract
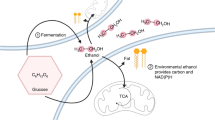
IT is generally accepted that the rate-limiting step in the metabolism of alcohol (ethanol) in man is the conversion of alcohol into acetaldehyde1–3. This reaction is believed to occur chiefly in the soluble cytoplasm of liver cells4,5, and is catalysed by alcohol dehydrogenase (ADH)6, NAD acting as hydrogen acceptor7,8. The part played by the interesting ethanol-oxidizing system recently discovered9 in the microsomes of liver cells may be significant, but requires further evaluation. The availability of unreduced NAD in the cell sap seems to be important10,11, because NADH competes with NAD for binding sites on the enzyme ADH and, in sufficient concentration, may inhibit the rate of ethanol dehydrogenation12. The NADH produced in the initial oxidative step in ethanol metabolism must therefore be continually reoxidized by various oxidation-reduction systems for the reaction to proceed13. In the cell sap of liver cells the reoxidation of NADH may be coupled to the reduction of pyruvate to lactate3,8, and to the reductive synthesis14, elongation and saturation of fatty acids; but perhaps the most important route for reoxidation of the NADH involves the mitochondrial flavoprotei–cytochrome electron transfer system coupled with phosphorylating oxidation15–17. The “carrier” of the hydrogen equivalents from NADH across the relatively impermeable mitochondrial membranes may be substances like alpha-glycero-phosphate17, beta-hydroxybutyrate18,19, malate and glutamate20, which serve to “shuttle” hydrogen from the extramitochondrial NADH, across the mitochondrial membranes, for oxidation in the intra-mitochondrial compartment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lundsgaard, E., Bull. Johns Hopkins Hosp., 63, 15 (1938).
Theorell, H., and Chance, B., Acta Chem. Scand., 5, 1127 (1951).
Westerfeld, W. W., Stotz, E., and Berg, R. L., J. Biol. Chem., 149, 237 (1943).
Nyberg, A., Schuberth, J., and Anggard, L., Acta Chem. Scand., 7, 1170 (1953).
Buttner, H., Biochem. Z., 341, 300 (1965).
Von Wartburg, J. P., Bethune, J. L., and Vallee, B. L., Biochemistry, 3, 1775 (1964).
Westheimer, F. H., Fisher, H. F., Conn, E. E., and Vennesland, B., J. Amer. Chem. Soc., 73, 2403 (1951).
Lundquist, F., Fugmann, U., Klaning, E., and Rasmussen, H., Biochem. J., 72, 409 (1959).
Lieber, C. S., and DeCarli, L. M., J. Clin. Invest., 47, 62a (1968).
Vitale, J. J., Hegsted, D. M., McGrath, H., Grable, E., and Zamcheck, N., J. Biol. Chem., 210, 753 (1954).
Isselbacher, K. J., and Greenberger, N. J., New Engl. J. Med., 270, 351 (1964).
Mahler, H. R., Baker, jun., R. H., and Shiner, jun., V. J., Biochemistry, 1, 47 (1962).
Smith, M. E., and Newman, H. W., J. Biol. Chem., 234, 1544 (1959).
Lieber, C. S., and Schmid, R., J. Clin. Invest., 40, 394 (1961).
Green, D. E., and Crane, F. L., in Proc. Intern. Symp. Enzyme Chem., 275 (Maruzen, Tokyo, 1958).
Lehninger, A. L., Harvey Lectures, 49, 176 (1953–54).
Lardy, H. A., Lee, Y. P., and Takemori, A. E., Ann. NY Acad. Sci., 86, 506 (1960).
Devlin, T. M., and Bedell, B. H., Biochim. Biophys. Acta, 36, 564 (1959).
Devlin, T. M., and Bedell, B. H., J. Biol. Chem., 235, 2134 (1960).
Chappell, J. B., Brit. Med. Bull., 24, 150 (1968).
Wordsworth, V. P., Brit. Med. J., i, 935 (1953).
Lieber, C. S., Ann. Rev. Med., 18, 35 (1967).
Pawan, G. L. S., and Hoult, W. H., Biochem. J., 87, 6P (1963).
Williams, L. A., Linn, R. A., and Zak, B., Clin. Chim. Acta, 3, 169 (1958).
Tygstrup, N., Winkler, K., and Lundquist, F., J. Clin. Invest., 44, 817 (1965).
Thieden, H. I. D., and Lundquist, F., Biochem. J., 102, 177 (1967).
Leuthardt, F., Testa, E., and Wolf, H. P., Helv. Chim. Acta, 36, 227 (1953).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PAWAN, G. Vitamins, Sugars and Ethanol Metabolism in Man. Nature 220, 374–376 (1968). https://doi.org/10.1038/220374a0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/220374a0
This article is cited by
-
Inhibition of ethanol absorption byRhodiola sachalinensis in rats
Archives of Pharmacal Research (1997)
-
Investigation of the properties of alcohol dehydrogenase from bean, rape, wheat and broad bean
Biologia Plantarum (1975)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.