Abstract
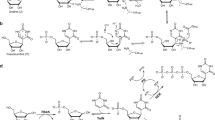
THE N-6 substituted adenines with a lipophilic radical show a biological activity of the kinetin type1,2. The ribosides of substituted adenine present an activity at least identical to that of the free bases2,3; this could be possibly attributed to their hydrolysis and the subsequent formation of the free bases. To test this hypothesis, the action of nucleoside phosphorylase from Escherichia coli was analysed on the ribosides of two kinins: 6-benzylaminopurine and 6-hexylaminopurine. The method used for the enzyme preparation was that of Paege and Schlenk4; the fraction of protein precipitated with ammonium sulphate between 60 and 80 per cent of saturation was retained and then dialysed. Preliminary tests on the hydrolysis of nucleosides were effected with inosine and adenosine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kuraishi, S., Sci. Papers Coll. Gen. Educ., Univ. Tokyo, 9, 67 (1959).
Strong, F. M., Topics Microbial. Chem. (John Wiley, New York, 1956).
Guern, J., and Hugon, E., C. R. Acad. Sci. (in the press).
Paege, L. M., and Schlenk, F., Arch. Biochem. Biophys., 40, 42 (1952).
Kalckar, H. M., J. Biol. Chem., 167, 429 (1947).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
TROUSLOT, M., GUERN, J. Enzyme Hydrolysis of 6-Benzylamino-9B D-Ribofuranosylpurine and 6-Hexylamino-9B D-Ribofuranosylpurine. Nature 213, 629–630 (1967). https://doi.org/10.1038/213629a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/213629a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.