Abstract
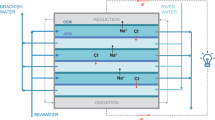
IN most experimental arrangements for electrodialysis using ion-exchange membranes, the ions of the electrolyte after being transported through the membranes are either discharged directly at the electrodes or recombine and neutralize each other's charge in an adjoining compartment. Thus, in industrial equipment for desalting brackish water, alternating cation- and anion-permeable membranes form a number of compartments between the electrodes. The saline water to be desalted is passed through every other compartment, and the ions from this water, after electrodialysis through the membranes, recombine in the remaining compartments to give a concentrated salt solution which is then discharged to waste1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kunin, R., Ion Exchange Resins, second ed., 229 (J. Wiley and Sons, New York, 1958).
Wood, T., Biochem. J., 62, 611 (1956).
Permaplex Ion-Selective Membranes, Technical Data (The Permutit Co., Ltd., London).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WOOD, T. Transport of Current during Fractionation by Electrodialysis through Ion-exchange Membranes. Nature 205, 1103–1104 (1965). https://doi.org/10.1038/2051103a0
Issue Date:
DOI: https://doi.org/10.1038/2051103a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.