Abstract
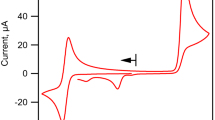
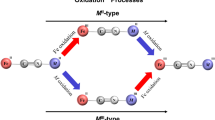
NEUTRAL solutions of K4Fe(CN)6 have been known to become alkaline when irradiated by ultra-violet light1–3. It has been suggested that photolysis of Fe(CN)6−4 first takes place to give Fe(CN)6−3 and (CN)−1, and the latter then hydrolyses, producing free alkali:  When the light source is cut off, all the three reactions are reversed and the original pH is restored unless irradiation has been far too prolonged2.
When the light source is cut off, all the three reactions are reversed and the original pH is restored unless irradiation has been far too prolonged2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Asperger, S., Trans. Farad. Soc., 48, 617 (1952).
MacDiarmid, A. G., and Hall, N. F., J. Amer. Chem. Soc., 75, 5204 (1953).
Yatsimirsky, K. B., and Vasil'ev, V. P., Instability Constants of Complex Compounds (Pergamon Press, 1960).
Martell, A. E., and Calvin, M., Chemistry of the Metal Chelate Compounds, 474 (Prentice Hall, Inc., New York, 1953).
Jortner, J., and Stein, G., J. Phys. Chem., 66, 1258 (1962).
Jortner, J., and Stein, G., J. Phys. Chem., 66, 1264 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MITRA, R., JAIN, D., BANNERJEE, A. et al. Role of Free Radicals in the Photo-oxidation of Fe+2 in Acidic Solutions of Ferrocyanide. Nature 200, 163–164 (1963). https://doi.org/10.1038/200163a0
Issue Date:
DOI: https://doi.org/10.1038/200163a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.