Abstract
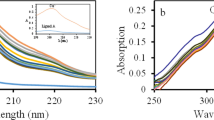
MERCURY electrodes used in electrochemistry are often prepared by plating mercury on solid electrodes. The formation of amalgams in these conditions has been in general disregarded. We have already shown1 that when gold or gold-plated electrodes are covered with mercury, there are electrode reactions different from those on pure mercury, owing to formation of intermetallic compounds.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kemula, W., Kublik, Z., and Galus, Z., Nature, 184, B.A.56 (1959).
Kemula, W., and Kublik, Z., Anal. Chim. Acta, 18, 104 (1958).
Gardiner, K. W., and Rogers, L. B., Anal. Chem., 25, 1393 (1953).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KEMULA, W., KUBLIK, Z. & GALUS, Z. Influence of Platinum in Mercury on the Mechanism of Electrode Reactions at the Mercury Electrode. Nature 184, 1795–1796 (1959). https://doi.org/10.1038/1841795a0
Issue Date:
DOI: https://doi.org/10.1038/1841795a0
This article is cited by
-
Einfluß der Versuchsbedingungen bei analytischen Bestimmungen mit Hilfe der square wave-Polarographie nach Vorelektrolyse an der stationären Elektrode
Fresenius' Zeitschrift für analytische Chemie (1962)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.