Abstract
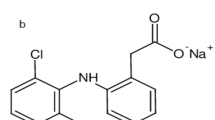
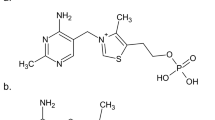
DURING a study of the reineckates of a series of clinically important basic compounds, the mechanism of the reaction between the bases and ammonium reineckate was studied. It was found that the formation of the complexes depends on the basicity of the compounds, and that under suitable pH conditions certain bases can be separated quantitatively from one another. A brief discussion of the reaction mechanism on which the separation is based is presented here, and a full report on this work will be published elsewhere.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Aycock, B. F., Eisenbraun, E. J., and Schrader, R. W., J. Amer. Chem. Soc., 73, 1351 (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KUM-TATT, L. Quantitative Separation of Nitrogenous Organic Bases using Ammonium Reineckate. Nature 182, 655–656 (1958). https://doi.org/10.1038/182655a0
Issue Date:
DOI: https://doi.org/10.1038/182655a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.