Abstract
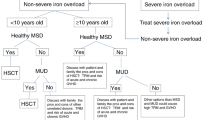
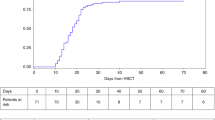
Autosomal recessive osteopetrosis (OP) is a disease characterized by osteoclast dysfunction, leading to multisystem morbidity and death of most affected children. Hematopoietic stem cell transplantation (HSCT) is the treatment of choice for OP, but this patient population is particularly prone to post-transplant complications and death after myeloablative conditioning. To determine the potential of achieving improved overall outcomes in these patients by decreasing pre-transplant mortality, we investigated engraftment and survival following a reduced intensity regimen including busulfan, fludarabine and total lymphoid irradiation. We report outcomes in 11 patients. All six patients who received a bone marrow or peripheral stem cell graft engrafted with >75% donor chimerism. In contrast, all five recipients of unrelated cord blood as a stem cell source for a first graft failed to demonstrate donor hematopoietic chimerism. The day 100 and 6-month mortality was low at 9%. One year after HSCT, six of 11 patients (55%) were surviving. Our data suggest that this regimen results in low peri-transplant mortality without compromising engraftment when a marrow or peripheral stem cell graft is used. An umbilical cord blood graft, however, should be used with caution for patients with OP when this or a similar reduced intensity regimen is used.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Teitelbaum SL, Coccia PF, Brown DM, Kahn AJ . Malignant osteopetrosis: a disease of abnormal osteoclast proliferation. Metab Bone Dis Relat Res 1981; 3: 99–105.
Tolar J, Teitelbaum SL, Orchard PJ . Osteopetrosis. N Engl J Med 2004; 351: 2839–2849.
Sobacchi C, Frattini A, Orchard P, Porras O, Tezcan I, Andolina M et al. The mutational spectrum of human malignant autosomal recessive osteopetrosis. Hum Mol Genet 2001; 10: 1767–1773.
Kornak U, Kasper D, Bosl MR, Kaiser E, Schweizer M, Schulz A et al. Loss of the ClC-7 chloride channel leads to osteopetrosis in mice and man. Cell 2001; 104: 205–215.
Kornak U, Schulz A, Friedrich W, Uhlhaas S, Kremens B, Voit T et al. Mutations in the a3 subunit of the vacuolar H(+)-ATPase cause infantile malignant osteopetrosis. Hum Mol Genet 2000; 9: 2059–2063.
Chalhoub N, Benachenhou N, Rajapurohitam V, Pata M, Ferron M, Frattini A et al. Grey-lethal mutation induces severe malignant autosomal recessive osteopetrosis in mouse and human. Nat Med 2003; 9: 399–406.
Sly WS, Hewett-Emmett D, Whyte MP, Yu YS, Tashian RE et al. Carbonic anhydrase II deficiency identified as the primary defect in the autosomal recessive syndrome of osteopetrosis with renal tubular acidosis and cerebral calcification. Proc Natl Acad Sci USA 1983; 80: 2752–2756.
Teitelbaum SL . Bone resorption by osteoclasts. Science 2000; 289: 1504–1508.
Coccia PF . Cells that resorb bone. N Engl J Med 1984; 310: 456–458.
Gerritsen EJ, Vossen JM, van Loo IH, Hermans J, Helfrich MH, Griscelli C et al. Autosomal recessive osteopetrosis: variability of findings at diagnosis and during the natural course. Pediatrics 1994; 93: 247–253.
Loria-Cortes R, Quesada-Calvo E, Cordero-Chaverri C . Osteopetrosis in children: a report of 26 cases. J Pediatr 1977; 91: 43–47.
Dorantes LM, Mejia AM, Dorantes S . Juvenile osteopetrosis: effects on blood and bone of prednisone and a low calcium, high phosphate diet. Arch Dis Child 1986; 61: 666–670.
Glorieux FH, Pettifor JM, Marie PJ, Delvin EE, Travers R, Shepard N . Induction of bone resorption by parathyroid hormone in congenital malignant osteopetrosis. Metab Bone Dis Relat Res 1981; 3: 143–150.
Key L, Carnes D, Cole S, Holtrop M, Bar-Shavit Z, Shapiro F et al. Treatment of congenital osteopetrosis with high-dose calcitriol. N Engl J Med 1984; 310: 409–415.
Key Jr LL, Ries WL, Rodriguiz RM, Hatcher HC . Recombinant human interferon gamma therapy for osteopetrosis. J Pediatr 1992; 121: 119–124.
Coccia PF, Krivit W, Cervenka J, Clawson C, Kersey JH, Kim TH et al. Successful bone-marrow transplantation for infantile malignant osteopetrosis. N Engl J Med 1980; 302: 701–708.
Eapen M, Davies SM, Ramsay NK, Orchard PJ . Hematopoietic stem cell transplantation for infantile osteopetrosis. Bone Marrow Transplant 1998; 22: 941–946.
Coccia PF . Hematopoietic cell transplantation for osteopetrosis. In: Blume KG, Forman SJ, Appelbaum FR (eds). Thomas' Hematopoietic Cell Transplantation, 3rd edn. Blackwell Publishing Ltd: Malden, MA, 2004, pp 1443–1454.
Gerritsen EJ, Vossen JM, Fasth A, Friedrich W, Morgan G, Padmos A et al. Bone marrow transplantation for autosomal recessive osteopetrosis. A report from the Working Party on Inborn Errors of the European Bone Marrow Transplantation Group. J Pediatr 1994; 125 (6 Part 1): 896–902.
Driessen GJ, Gerritsen EJ, Fischer A, Fasth A, Hop WC, Veys P et al. Long-term outcome of haematopoietic stem cell transplantation in autosomal recessive osteopetrosis: an EBMT report. Bone Marrow Transplant 2003; 32: 657–663.
Laughlin MJ, Eapen M, Rubinstein P, Wagner JE, Zhang MJ, Champlin RE et al. Outcomes after transplantation of cord blood or bone marrow from unrelated donors in adults with leukemia. N Engl J Med 2004; 351: 2265–2275.
Solh H, Da Cunha AM, Giri N, Padmos A, Spence D, Clink H et al. Bone marrow transplantation for infantile malignant osteopetrosis. J Pediatr Hematol Oncol 1995; 17: 350–355.
Steward CG, Pellier I, Mahajan A, Ashworth MT, Stuart AG, Fasth A et al. Severe pulmonary hypertension: a frequent complication of stem cell transplantation for malignant infantile osteopetrosis. Br J Haematol 2004; 124: 63–71.
Scharf SJ, Smith AG, Hansen JA, McFarland C, Erlich HA . Quantitative determination of bone marrow transplant engraftment using fluorescent polymerase chain reaction primers for human identity markers. Blood 1995; 85: 1954–1963.
Barker JN, Weisdorf DJ, DeFor TE, Blazar BR, McGlave PB, Miller JS et al. Transplantation of 2 partially HLA-matched umbilical cord blood units to enhance engraftment in adults with hematologic malignancy. Blood 2005; 105: 1343–1347.
Locatelli F, Beluffi G, Giorgiani G, Maccario R, Fiori P, Pession A et al. Transplantation of cord blood progenitor cells can promote bone resorption in autosomal recessive osteopetrosis. Bone Marrow Transplant 1997; 20: 701–705.
Jaing TH, Hou JW, Chen SH, Huang IA, Wang CJ, Lee WI . Successful unrelated mismatched cord blood transplantation in a child with malignant infantile osteopetrosis. Pediatr Transplant 2006; 10: 629–631.
Tsuji Y, Ito S, Isoda T, Kajiwara M, Nagasawa M, Morio T et al. Successful nonmyeloablative cord blood transplantation for an infant with malignant infantile osteopetrosis. J Pediatr Hematol Oncol 2005; 27: 495–498.
Nicholls BM, Bredius RG, Hamdy NA, Gerritsen EJ, Lankester AC, Hogendoorn PC et al. Limited rescue of osteoclast-poor osteopetrosis after successful engraftment by cord blood from an unrelated donor. J Bone Miner Res 2005; 20: 2264–2270.
Pozzi S, Lisini D, Podesta M, Bernardo ME, Sessarego N, Piaggio G et al. Donor multipotent mesenchymal stromal cells may engraft in pediatric patients given either cord blood or bone marrow transplantation. Exp Hematol 2006; 34: 934–942.
Wagner JE, Barker JN, DeFor TE, Baker KS, Blazar BR, Eide C et al. Transplantation of unrelated donor umbilical cord blood in 102 patients with malignant and nonmalignant diseases: influence of CD34 cell dose and HLA disparity on treatment-related mortality and survival. Blood 2002; 100: 1611–1618.
Wagner W, Wein F, Seckinger A, Frankhauser M, Wirkner U, Krause U et al. Comparative characteristics of mesenchymal stem cells from human bone marrow, adipose tissue, and umbilical cord blood. Exp Hematol 2005; 33: 1402–1416.
Erices A, Conget P, Minguell JJ . Mesenchymal progenitor cells in human umbilical cord blood. Br J Haematol 2000; 109: 235–242.
Bieback K, Kern S, Kluter H, Eichler H . Critical parameters for the isolation of mesenchymal stem cells from umbilical cord blood. Stem Cells 2004; 22: 625–634.
Kern S, Eichler H, Stoeve J, Kluter H, Bieback K . Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 2006; 24: 1294–1301.
Acknowledgements
This work was supported by Children's Cancer Research Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tolar, J., Bonfim, C., Grewal, S. et al. Engraftment and survival following hematopoietic stem cell transplantation for osteopetrosis using a reduced intensity conditioning regimen. Bone Marrow Transplant 38, 783–787 (2006). https://doi.org/10.1038/sj.bmt.1705533
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705533
Keywords
This article is cited by
-
TLI-based reduced-intensity conditioning hematopoietic SCT for children and adolescents with high-risk nonmalignant disorders
Bone Marrow Transplantation (2015)
-
From Marrow to Matrix: Novel Gene and Cell Therapies for Epidermolysis Bullosa
Molecular Therapy (2015)
-
Successful second haploidentical SCT in osteopetrosis
Bone Marrow Transplantation (2011)
-
Engraftment of unrelated cord blood after reduced-intensity conditioning regimen in children with refractory neuroblastoma: a feasibility trial
Bone Marrow Transplantation (2011)
-
Characterization and management of hypercalcemia following transplantation for osteopetrosis
Bone Marrow Transplantation (2010)