Abstract
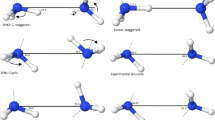
THE communication1 by C. K. and E. H. Ingold on the structure of N2O4 reviews the chemical and physical evidence in favour of the various structures proposed for N2O4. The authors conclude that the structure is  which is equivalent to the D2h. structure proposed by Sutherland2 on the basis of the spectroscopic data. Using the fundamentals proposed by Sutherland and the following molecular dimensions, namely, ∠ONO=120°, rNO = 1.15 A., rNN = 1.66 A.3, the entropy at 298° K. may be calculated by the usual methods of statistical thermodynamics4. In this way the entropy of translation for gaseous N2O4 at 298° K. is found to be 39.44 cal./deg./mole, and that due to vibration and rotation without taking torsion into account is 30.00 ± 0.3 cal./deg./mole. (The error of ± 0.3 E.U. in the rotational entropy is estimated by assuming that the error in each molecular dimension is ± 5 per cent.) The entropy observed by Giauque and Kemp5 was 72.73 cal./deg./mole. Thus the amount of entropy contributed by the degree of freedom associated with internal rotation is 3.29 ± 0.3 cal./deg./mole. Assuming the potential energy is of the form V = B/2 (1−cos 2π) the torsional frequency and potential barrier B are related to the moment of inertia about the torsional axis by ν = 1/2π [8B/I]1/2. Then if the potential barrier is considered to be so high that the contribution to the entropy is simply due to a torsional oscillation, then the torsional frequency is 108 ± 20 cm.−1, with a corresponding barrier of 8.5 ± 2 cal./mole.
which is equivalent to the D2h. structure proposed by Sutherland2 on the basis of the spectroscopic data. Using the fundamentals proposed by Sutherland and the following molecular dimensions, namely, ∠ONO=120°, rNO = 1.15 A., rNN = 1.66 A.3, the entropy at 298° K. may be calculated by the usual methods of statistical thermodynamics4. In this way the entropy of translation for gaseous N2O4 at 298° K. is found to be 39.44 cal./deg./mole, and that due to vibration and rotation without taking torsion into account is 30.00 ± 0.3 cal./deg./mole. (The error of ± 0.3 E.U. in the rotational entropy is estimated by assuming that the error in each molecular dimension is ± 5 per cent.) The entropy observed by Giauque and Kemp5 was 72.73 cal./deg./mole. Thus the amount of entropy contributed by the degree of freedom associated with internal rotation is 3.29 ± 0.3 cal./deg./mole. Assuming the potential energy is of the form V = B/2 (1−cos 2π) the torsional frequency and potential barrier B are related to the moment of inertia about the torsional axis by ν = 1/2π [8B/I]1/2. Then if the potential barrier is considered to be so high that the contribution to the entropy is simply due to a torsional oscillation, then the torsional frequency is 108 ± 20 cm.−1, with a corresponding barrier of 8.5 ± 2 cal./mole.
Similar content being viewed by others
Article PDF
References
Ingold, C. K., and Ingold, E. H., Nature, 159, 743 (1947).
Sutherland, G. B. B. M., Proc. Roy. Soc., A, 141, 342 535 (1933).
Maxwell, L. R., Mosley, V. M., and Denning, L. S., J. Chem. Phys., 2, 331 (1934).
Herzberg, G., “Infra-red and Raman Spectra”, 501 et seq. (New York: D. van Nostrand, 1945).
Giauque, W. F., and Kemp, J. D., J. Chem. Phys., 6, 40 (1938).
Pitzer, K. S., and Gwinn, W. D., J. Chem. Phys., 10, 428 (1942).
Longuet-Higgins, H. C., Nature, 153, 408 (1944).
Duchesne, J., C.R. Acad. Sci., Paris, 204, 1112 (1937).
See, for example, Klotz, I. M., J. Chem. Educ., 22 328 (1945).
McCollum, E. D., J. Amer. Chem. Soc., 49, 28 (1927).
Partington, J. R., and Shilling, W. G., “The Specific Heat of Gases”, 188 (London: Ernest Benn, 1924).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BERNSTEIN, H., BURNS, W. Potential Barrier in Dinitrogen Tetroxide. Nature 166, 1039 (1950). https://doi.org/10.1038/1661039a0
Issue Date:
DOI: https://doi.org/10.1038/1661039a0
This article is cited by
-
Dipole moment of nitrogen dioxide and dinitrogen dioxide—The structure of the dimer
Proceedings of the Indian Academy of Sciences - Section A (1964)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.