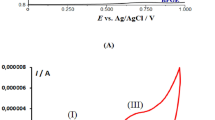
Abstract
IN supporting electrolytes generally used in polarography, for example, in solutions of potassium, lithium or ammonium salts or lithium hydroxide or in buffer solutions, no polarographic reduction wave of acetone can be observed. Only if electrolytes of tetra-alkylammonium are used can a reduction wave at a half-wave potential of about −2.1 V. (from saturated calomel electrode) be obtained1,2. After conversion of acetone to its hydrazone or phenyl-hydrazone, two reduction steps are obtained3, but none is suitable for analytical purposes; moreover, the height of these waves is not a linear function of concentration. Thus acetone is polarographically determined in an indirect way in acid sulphite solution, with which it reacts as other ketones and lowers the acid sulphite wave4.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Neiman, M. B., and Markhina, Z. V., Zavod. Lab., 13, 1174 (1947).
Stackelberg, M. v., and Stracke, W., Z.f. Elektrochem., 53, 118 (1949).
Lupton, J. M., and Lynch, C. C., J. Amer. Chem. Soc., 66, 697 (1944).
Heyrovský, J., in “Polarographie”, 368 (Springer, Vienna, 1941).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ZUMAN, P. Polarographic Behaviour of Acetone. Nature 165, 485–486 (1950). https://doi.org/10.1038/165485b0
Issue Date:
DOI: https://doi.org/10.1038/165485b0
This article is cited by
-
Indirect polarographic determination of glyoxylic acid
Pharmaceutical Chemistry Journal (1991)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.