Abstract
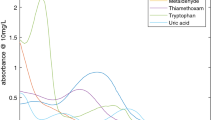
THE communication by T. R. Scott1 raises the point of the effect of bicarbonate in the hydrogen peroxide method for the absorptiometric determination of uranium. It should be pointed out that although this particular example had not been noted previously, it is one instance of two general effects which have not been stressed in the literature referring to this method. These are that the light absorption or optical density for a given concentration of uranium with excess peroxide present is dependent (a) upon the final pH. and (b) upon the particular alkali used (or if a mixture, upon the relative amounts); for example, we have shown that sodium hydroxide and sodium carbonate may give different results even at a given final pH. The accompanying graph illustrates these points.
Similar content being viewed by others
Article PDF
References
Scott, T. R., Nature, 163, 768 (1949).
Sandell, E. B., "Colorimetric Determination of Traces of Metals" (Interscience Publishers Inc., New York, 1944).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SMALES, A., FURBY, E. Absorptiometric Determination of Uranium. Nature 164, 579 (1949). https://doi.org/10.1038/164579a0
Issue Date:
DOI: https://doi.org/10.1038/164579a0
This article is cited by
-
Absorption Spectra of the Sodium Peruranates
Nature (1950)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.