Abstract
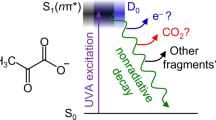
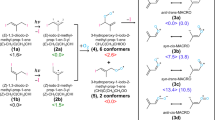
IN continuation of an investigation which has been reported elsewhere1, an attempt was made to produce free methyl radicals by the photo-decomposition of acetone. According to Norrish, Crone and Saltmarsh2, the radicals are produced by the following mechanism: (CH3)2CO + hv 2CH3 + CO which is based on the latter authors' observation that at 60° the main products are equal volumes of carbon monoxide and ethane.
Similar content being viewed by others
Article PDF
References
Proc. Leeds Phil. Soc., 3, 141 (1936).
J. Chem. Soc., 1456 (1934).
NATURE, 135, 307 (1935).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SPENCE, R., WILD, W. Mechanism of the Photo-Decomposition of Acetone. Nature 138, 206 (1936). https://doi.org/10.1038/138206b0
Issue Date:
DOI: https://doi.org/10.1038/138206b0
This article is cited by
-
Photodecomposition of Aldehydes and Ketones
Nature (1936)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.