Abstract
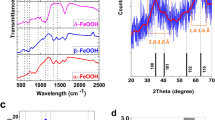
FOR the rusting of iron, oxygen, water, and a trace of acid are required. In the process oxygen is used up, also a certain amount of water, but the acid may be said to act as a catalyst. It is, in fact, regenerated when the ferrous salt first formed is oxidised to the ferric state, as was shown previously (Trans. Faraday Soc., 18, 310–317; 1923). This is at once evident when we consider that, on making solutions of ferric or ferrous salts less acid, ferric hydroxide is first precipitated, at pH. 3.5 to 5.5, and ferrous hydroxide comes down later, at pH. 5.1 and onwards, a trace being still in solution at pH 7.6. Oxidation, therefore, must result in a liberation of acid, accounted for by the ferric being hydrolysed to a greater extent than the ferrous salt.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ATKINS, W. Lecture Experiments on the Hydrogen Ion Concentration Changes in the Rusting of Iron. Nature 121, 615 (1928). https://doi.org/10.1038/121615a0
Issue Date:
DOI: https://doi.org/10.1038/121615a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.