Abstract
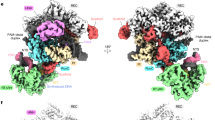
Angiogenin, an RNase A-family protein, promotes angiogenesis and has been implicated in cancer, neurodegenerative diseases, and epigenetic inheritance 1-10. Upon activation during cellular stress, angiogenin cleaves tRNAs at the anticodon loop, resulting in translation repression 11-15. The catalytic activity of isolated angiogenin, however, is very low, and the mechanisms of the enzyme activation and tRNA specificity have remained a puzzle 3,16-23. Here, we uncover these mechanisms using biochemical assays and cryogenic electron microscopy. Our work reveals that the cytosolic ribosome is the long-sought activator of angiogenin. A 2.8-Å resolution cryo-EM structure features angiogenin bound in the A site of the 80S ribosome. The C-terminal tail of angiogenin is rearranged by interactions with the ribosome to activate the RNase catalytic center, making the enzyme several orders of magnitude more efficient in tRNA cleavage. Additional 80S•angiogenin structures capture how tRNA substrate is directed by the ribosome into angiogenin’s active site, demonstrating that the ribosome acts as the specificity factor. Our findings therefore suggest that angiogenin is activated by ribosomes with a vacant A site, whose abundance increases during cellular stress24-27. These results may facilitate the development of therapeutics to treat cancer and neurodegenerative diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Information
This file contains the Supplementary Discussion, full descriptions for Supplementary Videos 1 and 2, Supplementary Fig. 1 and Supplementary references.
Supplementary Video 1
Cryo-EM maps and rigid-body fits of the subclasses of 80S•angiogenin•tRNAAla show different positions of tRNA (green) and P stalk (cyan, upper left corner) relative to angiogenin (magenta). This flexible tethering of the tRNA may allow cleavage at different anticodon positions on various tRNA species. The cryo-EM maps were low-pass filtered to 6 Å and are shown at 2.5 σ.
Supplementary Video 2
Cryo-EM maps and rigid-body fits of the subclasses of 80S•angiogenin•tRNAAla show different positions of tRNA (green) and P stalk (cyan, upper left corner) relative to the 60S subunit’s GTPase activation center. The cryo-EM maps were low-pass filtered to 6 Å and are shown at 2.5 σ.
Rights and permissions
About this article
Cite this article
Loveland, A.B., Koh, C.S., Ganesan, R. et al. Structural mechanism of angiogenin activation by the ribosome. Nature (2024). https://doi.org/10.1038/s41586-024-07508-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41586-024-07508-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.