Abstract
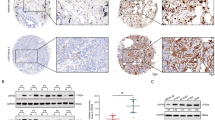
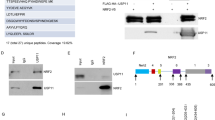
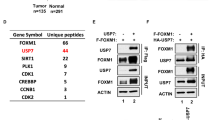
The oncoprotein c-Myc is frequently overexpressed in many cancers and is essential for cancer cell proliferation. Ubiquitin-proteasome-dependent degradation is one of the main ways in which cells control c-Myc abundance at a post-translational level. However, the underlying mechanism by which c-Myc is directly deubiquitinated is not fully understood. In this study, by screening ubiquitin-specific proteases (USPs) that may regulate c-Myc stability, we identified USP37 as a novel deubiquitinating enzyme (DUB) that stabilizes c-Myc via direct binding. The overexpression of USP37 markedly increases c-Myc abundance by blocking its degradation, whereas the depletion of USP37 promotes c-Myc degradation and reduces c-Myc levels. Further studies indicate that USP37 directly interacts with c-Myc and deubiquitinates c-Myc in a DUB activity-dependent manner. Functionally, USP37 regulates cell proliferation and the Warburg effect by regulating c-Myc levels. Clinically, USP37 is significantly upregulated in human lung cancer tissues, where its expression is positively correlated with c-Myc protein expression. Thus, our findings uncover a previously unrecognized role for USP37 in the regulation of c-Myc stability in lung cancer and suggest that USP37 might be a potential therapeutic target for the treatment of lung cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hershko A, Ciechanover A . The ubiquitin system. Annu Rev Biochem 1998; 67: 425–479.
Rotin D, Kumar S . Physiological functions of the HECT family of ubiquitin ligases. Nat Rev Mol Cell Biol 2009; 10: 398–409.
Amerik AY, Hochstrasser M . Mechanism and function of deubiquitinating enzymes. Biochim Biophys Acta 2004; 1695: 189–207.
Nijman SMB, Luna-Vargas MPA, Velds A, Brummelkamp TR, Dirac AMG, Sixma TK et al. A genomic and functional inventory of deubiquitinating enzymes. Cell 2005; 123: 773–786.
Sacco JJ, Coulson JM, Clague MJ, Urbé S . Emerging roles of deubiquitinases in cancer-associated pathways. IUBMB Life 2010; 62: 140–157 NA-NA.
Todi SV, Paulson HL . Balancing act: deubiquitinating enzymes in the nervous system. Trends Neurosci 2011; 34: 370–382.
Yuan J, Luo K, Zhang L, Cheville JC, Lou Z . USP10 regulates p53 localization and stability by deubiquitinating p53. Cell 2010; 140: 384–396.
Sheng Y, Saridakis V, Sarkari F, Duan S, Wu T, Arrowsmith CH et al. Molecular recognition of p53 and MDM2 by USP7/HAUSP. Nat Struct Mol Biol 2006; 13: 285–291.
Song MS, Salmena L, Carracedo A, Egia A, Lo-Coco F, Teruya-Feldstein J et al. The deubiquitinylation and localization of PTEN are regulated by a HAUSP-PML network. Nature 2008; 455: 813–817.
Zhang J, Zhang P, Wei Y, Piao HL, Wang W, Maddika S et al. Deubiquitylation and stabilization of PTEN by USP13. Nat Cell Biol 2013; 15: 1486–1494.
Lin Z, Yang H, Kong Q, Li J, Lee SM, Gao B et al. USP22 antagonizes p53 transcriptional activation by deubiquitinating Sirt1 to suppress cell apoptosis and is required for mouse embryonic development. Mol Cell 2012; 46: 484–494.
Liu J, Xia H, Kim M, Xu L, Li Y, Zhang L et al. Beclin1 controls the levels of p53 by regulating the deubiquitination activity of USP10 and USP13. Cell 2011; 147: 223–234.
Song MS, Salmena L, Carracedo A, Egia A, Lo-Coco F, Teruya-Feldstein J et al. The deubiquitinylation and localization of PTEN are regulated by a HAUSP–PML network. Nature 2008; 455: 813–817.
Dang CV, O’Donnell KA, Zeller KI, Nguyen T, Osthus RC, Li F . The c-Myc target gene network. Semin Cancer Biol 2006; 16: 253–264.
Larsson L-G, Henriksson MA . The Yin and Yang functions of the Myc oncoprotein in cancer development and as targets for therapy. Exp Cell Res 2010; 316: 1429–1437.
Soga T . Cancer metabolism: key players in metabolic reprogramming. Cancer Sci 2013; 104: 275–281.
DeBerardinis RJ, Lum JJ, Hatzivassiliou G, Thompson CB . The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab 2008; 7: 11–20.
Dang CV, Kim JW, Gao P, Yustein J . The interplay between MYC and HIF in cancer. Nat Rev Cancer 2008; 8: 51–56.
Oster SK, Ho CS, Soucie EL, Penn LZ . The myc oncogene: MarvelouslY complex. Adv Cancer Res 2002; 84: 81–154.
Salghetti SE, Young Kim S, Tansey WP . Destruction of Myc by ubiquitin‐mediated proteolysis: cancer‐associated and transforming mutations stabilize Myc. EMBO J 1999; 18: 717–726.
Welcker M, Orian A, Jin J, Grim JA, Harper JW, Eisenman RN et al. The Fbw7 tumor suppressor regulates glycogen synthase kinase 3 phosphorylation-dependent c-Myc protein degradation. Proc Natl Acad Sci USA 2004; 101: 9085–9090.
Von Der Lehr N, Johansson S, Wu S, Bahram F, Castell A, Cetinkaya C et al. The F-box protein Skp2 participates in c-Myc proteosomal degradation and acts as a cofactor for c-Myc-regulated transcription. Mol Cell 2003; 11: 1189–1200.
Hart M, Concordet J, Lassot I, Albert I, Del los Santos R, Durand H et al. The F-box protein β-TrCP associates with phosphorylated β-catenin and regulates its activity in the cell. Curr Biol 1999; 9: 207–211.
Popov N, Schülein C, Jaenicke LA, Eilers M . Ubiquitylation of the amino terminus of Myc by SCF [beta]-TrCP antagonizes SCFFbw7-mediated turnover. Nat Cell Biol 2010; 12: 973–981.
Paul I, Ahmed SF, Bhowmik A, Deb S, Ghosh MK . The ubiquitin ligase CHIP regulates c-Myc stability and transcriptional activity. Oncogene 2013; 32: 1284–1295.
Schwamborn JC, Berezikov E, Knoblich JA . The TRIM-NHL protein TRIM32 activates microRNAs and prevents self-renewal in mouse neural progenitors. Cell 2009; 136: 913–925.
Choi SH, Wright JB, Gerber SA, Cole MD . Myc protein is stabilized by suppression of a novel E3 ligase complex in cancer cells. Genes Dev 2010; 24: 1236–1241.
Adhikary S, Marinoni F, Hock A, Hulleman E, Popov N, Beier R et al. The ubiquitin ligase HectH9 regulates transcriptional activation by Myc and is essential for tumor cell proliferation. Cell 2005; 123: 409–421.
Popov N, Wanzel M, Madiredjo M, Zhang D, Beijersbergen R, Bernards R et al. The ubiquitin-specific protease USP28 is required for MYC stability. Nat Cell Biol 2007; 9: 765–774.
Welcker M, Clurman BE . FBW7 ubiquitin ligase: a tumour suppressor at the crossroads of cell division, growth and differentiation. Nat Rev Cancer 2008; 8: 83–93.
Welcker M, Orian A, Jin J, Grim JE, Harper JW, Eisenman RN et al. The Fbw7 tumor suppressor regulates glycogen synthase kinase 3 phosphorylation-dependent c-Myc protein degradation. Proc Natl Acad Sci USA 2004; 101: 9085–9090.
Yada M, Hatakeyama S, Kamura T, Nishiyama M, Tsunematsu R, Imaki H et al. Phosphorylation-dependent degradation of c-Myc is mediated by the F-box protein Fbw7. EMBO J 2004; 23: 2116–2125.
Herbst A, Salghetti SE, Kim SY, Tansey WP . Multiple cell-type-specific elements regulate Myc protein stability. Oncogene 2004; 23: 3863–3871.
Hsu PP, Sabatini DM . Cancer cell metabolism: Warburg and beyond. Cell 2008; 134: 703–707.
Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 2012; 490: 61–70.
Seo HR, Kim J, Bae S, Soh JW, Lee YS . Cdk5-mediated phosphorylation of c-Myc on Ser-62 is essential in transcriptional activation of cyclin B1 by cyclin G1. J Biol Chem 2008; 283: 15601–15610.
von der Lehr N, Johansson S, Wu S, Bahram F, Castell A, Cetinkaya C et al. The F-box protein Skp2 participates in c-Myc proteosomal degradation and acts as a cofactor for c-Myc-regulated transcription. Mol Cell 2003; 11: 1189–1200.
Kim SY, Herbst A, Tworkowski KA, Salghetti SE, Tansey WP . Skp2 regulates Myc protein stability and activity. Mol Cell 2003; 11: 1177–1188.
Huang X, Summers Matthew K, Pham V, Lill Jennie R, Liu J, Lee G et al. Deubiquitinase USP37 is activated by CDK2 to antagonize APCCDH1 and promote S phase entry. Mol Cell 2011; 42: 511–523.
Bianchi F, Nuciforo P, Vecchi M, Bernard L, Tizzoni L, Marchetti A et al. Survival prediction of stage I lung adenocarcinomas by expression of 10 genes. J Clin Invest 2007; 117: 3436–3444.
Yang WC, Shih HM . The deubiquitinating enzyme USP37 regulates the oncogenic fusion protein PLZF/RARA stability. Oncogene 2012; 32: 5167–5175.
Das CM, Taylor P, Gireud M, Singh A, Lee D, Fuller G et al. The deubiquitylase USP37 links REST to the control of p27 stability and cell proliferation. Oncogene 2012; 32: 1691–1701.
Li R, Wei J, Jiang C, Liu D, Deng L, Zhang K et al. Akt SUMOylation regulates cell proliferation and tumorigenesis. Cancer Res 2013; 73: 5742–5753.
Ning X, Hui L, Jian L, Rui W, Haiquan C, Jiquan C et al. Ubiquitin-specific protease 4 (USP4) targets TRAF2 and TRAF6 for deubiquitination and inhibits TNFalpha-induced cancer cell migration. Biochem J 2012; 441: 979–986.
Liao P, Wang W, Shen M, Pan W, Zhang K, Wang R et al. A positive feedback loop between EBP2 and c-Myc regulates rDNA transcription, cell proliferation, and tumorigenesis. Cell Death Dis 2014; 5: e1032.
Acknowledgements
We thank other members of the Wang laboratory for their assistance. This work was supported by grants from the National Basic Research Program of China (973 program 2010CB529704 and 2012CB910404), the National Natural Science Foundation of China (30800587, 30971521 and 31171338), the Science and Technology Commission of Shanghai Municipality (11DZ2260300) and the Doctoral Fund of Ministry of Education of China (20130076110022). PW is a scholar of the Shanghai Rising-Star Program from the Science and Technology Commission of Shanghai Municipality (09QA1401900 and 13QH1401), the Program for New Century Excellent Talents in University (NCET-10-0387) and the Dawn Program of Shanghai Education Commission (11SG27).
Author Contributions
JP, QD, JC and PW designed the research; JP, QD, JC, XW, TN, HL, JJ, TC, PW, XC and XY performed the experiments; JP, QD, JC, XW and LM analyzed the data; JP, JX and PW wrote the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Pan, J., Deng, Q., Jiang, C. et al. USP37 directly deubiquitinates and stabilizes c-Myc in lung cancer. Oncogene 34, 3957–3967 (2015). https://doi.org/10.1038/onc.2014.327
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.327
This article is cited by
-
USP43 stabilizes c-Myc to promote glycolysis and metastasis in bladder cancer
Cell Death & Disease (2024)
-
New insights in ubiquitin-dependent Wnt receptor regulation in tumorigenesis
In Vitro Cellular & Developmental Biology - Animal (2024)
-
MYC in liver cancer: mechanisms and targeted therapy opportunities
Oncogene (2023)
-
Deubiquitinases in cancer
Nature Reviews Cancer (2023)
-
Ubiquitin specific peptidase 37 and PCNA interaction promotes osteosarcoma pathogenesis by modulating replication fork progression
Journal of Translational Medicine (2023)