Abstract
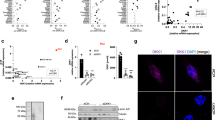
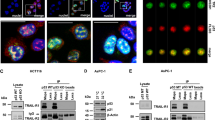
The TP53 tumor-suppressor gene is frequently mutated in human cancer. Missense mutations can add novel functions (gain-of-function, GOF) that promote tumor malignancy. Here we report that mutant (mut) p53 promotes tumor malignancy by suppressing the expression of a natural occurring anti-inflammatory cytokine, the secreted interleukin-1 receptor antagonist (sIL-1Ra, IL1RN). We show that mutp53 but not wild-type (wt) p53 suppresses the sIL-1Ra production in conditioned media of cancer cells. Moreover, mutp53, but not wtp53, binds physically the sIL-1Ra promoter and the protein–protein interaction with the transcriptional co-repressor MAFF (v-MAF musculoaponeurotic fibrosarcoma oncogene family, protein F) is required for mutp53-induced sIL-1Ra suppression. Remarkably, when exposed to IL-1 beta (IL-1β) inflammatory stimuli, mutp53 sustains a ready-to-be-activated in vitro and in vivo cancer cells’ response through the sIL-1Ra repression. Taken together, these results identify sIL-1Ra as a novel mutp53 target gene, whose suppression might be required to generate a chronic pro-inflammatory tumor microenvironment through which mutp53 promotes tumor malignancy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Aas T, Børresen AL, Geisler S, Smith-Sørensen B, Johnsen H, Varhaug JE et al. Specific P53 mutations are associated with de novo resistance to DOXorubicin in breast cancer patients. Nature Med 1996; 2: 811–814.
Lang GA, Iwakuma T, Suh YA, Liu G, Rao VA, Parant JM et al. Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell 2004; 199: 861–872.
Olive KP, Tuveson DA, Ruhe ZC, Yin B, Willis NA, Bronson RT et al. Mutp53 gain of function in two mouse models of Li-Fraumeni syndrome. Cell 2004; 119: 847–860.
Bossi G, Lapi E, Strano S, Rinaldo C, Blandino G, Sacchi A . Mutp53 gain of function: reduction of tumor malignancy of human cancer cell lines through abrogation of mutp53 expression. Oncogene 2006; 25: 304–309.
Weisz L, Zalcenstein A, Stambolsky P, Cohen Y, Goldfinger N, Oren M, Rotter V . Transactivation of the EGR1 gene contributes to mutp53 gain of function. Cancer Res 2004; 64: 8318–8327.
Bossi G, Marampon F, Maor-Aloni R, Zani B, Rotter V, Oren M et al. Conditional RNA interference in vivo to study mutp53 oncogenic gain of function on tumor malignancy. Cell Cycle 2008; 7: 1870–1879.
Smith Jr MF, Eidlen D, Brewer MT, Eisenberg SP, Arend WP, Gutierrez-Hartmann A . Human IL-1 receptor antagonist promoter. Cell type-specific activity and identification of regulatory regions. J Immunol 1992; 149: 2000–2007.
Jenkins JK, Drong RF, Shuck ME, Bienkowski MJ, Slightom JL, Arend WP et al. Intracellular IL-1 receptor antagonist promoter: cell type-specific and inducible regulatory regions. J Immunol 1997; 158: 748–755.
Dunn E, Sims JE, Nicklin MJ, O’Neill LA . Annotating genes with potential roles in the immune system: six new members of the IL-1 family. Trends Immunol 2001; 22: 533–536.
Schreuder H, Tardif C, Trump-Kallmeyer S, Soffientini A, Sarubbi E, Akeson A et al. A new cytokine-receptor binding mode revealed by the crystal structure of the IL-1 receptor with an antagonist. Nature 1997; 386: 194–200.
Apte RN, Voronov E . Is interleukin-1 a good or bad 'guy' in tumor immunobiology and immunotherapy? Immunol Rev 2008; 222: 222–241.
Zitvogel L, Kepp O, Galluzzi L, Kroemer G . ‘Inflammosome in carcinogenesis and anticancer immune responses’. Nat Immunol 2012; 13: 343–351.
Mantovani A, Allavena P, Sica A, Balkwill F . Cancer-related inflammation. Nature 2008; 454: 436–444.
Dinarello CA . Why not treat human cancer with Interleukin-1 blockade? Cancer Metastasis Rev 2010; 29: 317–329.
Dinarello CA, Simon A, van der Merr JWM . Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nature Rev 2012; 11: 633–652.
Gurtner A, Starace G, Norelli G, Piaggio G, Sacchi A, Bossi G . Mutp53-induced up-regulation of mitogen-activated protein kinase kinase 3 contributes to gain of function. J Biol Chem 2010; 285: 14160–14169.
Gabay C, Smith Jr MF, Eidlen D, Arend WP . Interleukin-1 receptor antagonist (IL-1Ra) is an acute-phase protein. J Clin Invest 1997; 99: 2930–2940.
Kim E, Deppert W . Transcriptional activities of mutp53: when mutations are more than a loss. J Cell Biochem 2004; 93: 878–886.
Quante T, Otto B, Brázdová M, Kejnovská I, Deppert W, Tolstonog GV . Mutp53 is a transcriptional co-factor that binds to G-rich regulatory regions of active genes and generates transcriptional plasticity. Cell Cycle 2012; 11: 3290–3303.
Scian MJ, Stagliano KE, Anderson MA, Hassan S, Bowman M, Miles MF et al. Tumor-derived p53 muts induce NF-kappaB2 gene expression. Mol Cell Biol 2005; 25: 10097–10110.
Di Agostino S, Strano S, Emiliozzi V, Zerbini V, Mottolese M, Sacchi A et al. Gain of function of mutp53: the mutp53/NF-Y protein complex reveals an aberrant transcriptional mechanism of cell cycle regulation. Cancer Cell 2006; 10: 191–202.
Zweidler-Mckay PA, Grimes HL, Flubacher MM, Tsichlis PN . Gfi-1 encodes a nuclear zinc finger protein that binds DNA and functions as a transcriptional repressor. Mol Cell Biol 1996; 16: 4024–4034.
Fischer A, Klattig J, Kneitz B, Diez H, Maier M, Holtmann B et al. Hey basic helix-loop-helix transcription factors are repressors of GATA4 and GATA6 and restrict expression of the GATA target gene ANF in fetal hearts. Mol Cell Biol 2005; 25: 8960–8970.
Kimura T, Ivell R, Rust W, Mizumoto Y, Ogita K, Kusui C et al. Molecular cloning of a human MAFF homologue, which specifically binds to the oxytocin receptor gene in term myometrium. Biochem Biophys Res Commun 1999; 264: 86–92.
Blank V, Small MAF . proteins in mammalian gene control: mere dimerization partners or dynamic transcriptional regulators? J Mol Biol 2008; 376: 913–925.
Elaraj DM, Weinreich DM, Varghese S, Puhlmann M, Hewitt SM, Carroll NM et al. The role of Interleukin 1 in growth and metastasis in human cancer xenograft. Clin Cancer Res 2006; 12: 1088–1096.
Puhlmann M, Weinreich DM, Farma JM, Carroll NM, Turner EM, Alexander Jr HR . Interleukin-1beta induced vascular permeability is dependent on induction of endothelial tissue factor (TF) activity. J Transl Med 2005; 3: 37.
Streicher KL, Willmarth NE, Garcia J, Boerner JL, Dewey TG, Ethier SP . Activation of a nuclear factor kappaB/interleukin-1 positive feedback loop by amphiregulin in human breast cancer cells. Mol Cancer Res 2007; 5: 847–861.
Wu S, Meeker WA, Wiener JR, Berchuck A, Bast Jr RC, Boyer CM . Transfection of ovarian cancer cells with tumor necrosis factor-alpha (TNF-alpha) antisense mRNA abolishes the proliferative response to interleukin-1 (IL-1) but not TNF-alpha. Gynecol Oncol 1994; 53: 59–63.
Li Y, Wang L, Pappan L, Galliher-Beckley A, Shi J . IL-1β promotes stemness and invasiveness of colon cancer cells through Zeb1 activation. Mol Cancer 2012; 11: 87.
Germano G, Allavena P, Mantovani A . Cytokines as a key component of cancer-related inflammation. Cytokine 2008; 43: 374–379.
Lewis AM, Varghese S, Xu H, Alexander HR . Interleukin-1 and cancer progression: the emerging role of inteleukin-1 receptor antagonist as a novel therapeutic agent in cancer treatment. J Transl Med 2006; 4: 48.
Saijo Y, Tanaka M, Miki M, Usui K, Suzuki T, Maemondo M et al. Proinflammatory cytokine IL-1h promotes tumor growth of Lewis lung carcinoma by induction of angiogenic factors: in vivo analysis of tumor-stromal interaction. J Immunol 2002; 169: 469–475.
Ulich TR, Guo K, Yin S, del Castillo J, Yi ES, Thompson RC et al. Endotoxin-induced cytokine gene expression in vivo. IV. Expression of interleukin-la/ß and interleukin-1 receptor antagonist mRNA during endotoxemia and during endotoxin-initiated local acute inflammation. Am J Pathol 1992; 141: 61–68.
Chirivi RG, Garofalo A, Padura IM, Mantovani A, Giavazzi R . Interleukin 1 receptor antagonist inhibits the augmentation of metastasis induced by interleukin 1 or lipopolysaccharide in a human melanoma/nude mouse system. Cancer Res 1993; 53: 5051–5054.
Skrzypczak M, Goryca K, Rubel T, Paziewska A, Mikula M, Jarosz D et al. Modeling oncogenic signaling in colon tumors by multidirectional analyses of microarray data directed for maximization of analytical reliability. PLoS ONE 2010; 5: pii e13091.
Notterman DA, Alon U, Sierk AJ, Levine AJ . Transcriptional gene expression profiles of colorectal adenoma, adenocarcinoma, and normal tissue examined by oligonucleotide arrays. Cancer Res 2001; 61: 3124–3130.
Muller PA, Vousden KH . p53 mutations in cancer. Nat Cell Biol 2013; 15: 2–8.
Freed-Pastor WA, Prives C . Mutp53: one name, many proteins. Genes Dev 2012; 26: 1268–1286.
Lujambio A, Akkari L, Simon J, Grace D, Tschaharganeh DF, Bolden JE et al. Non-cell-autonomous tumor suppression by p53. Cell 2013; 153: 449–460.
Bar J, Feniger-Barish R, Lukashchuk N, Shaham H, Moskovits N, Goldfinger N et al. Cancer cells suppress p53 in adjacent fibroblasts. Oncogene 2009; 28: 933–936.
Addadi Y, Moskovits N, Granot D, Lozano G, Carmi Y, Apte RN et al. p53 status in stromal fibroblasts modulates tumor growth in an SDF1-dependent manner. Cancer Res 2010; 70: 9650–9658.
Weisz L, Damalas A, Liontos M, Karakaidos P, Fontemaggi G, Maor-Aloni R et al. Mutp53 enhances nuclear factor kappaB activation by tumor necrosis factor alpha in cancer cells. Cancer Res 2007; 67: 2396–2401.
Cooks T, Pateras IS, Tarcic O, Solomon H, Schetter AJ, Wilder S et al. Mutant p53 prolongs NF-κB activation and promotes chronic inflammation and inflammation-associated colorectal cancer. Cancer Cell 2013; 23: 634–646.
D'Arcangelo D, Gaetano C, Capogrossi MC . Acidification prevents endothelial cell apoptosis by Axl activation. Circ Res 2002; 91: e4–e12.
Acknowledgements
We thank Dr S Bacchetti, Dr FL Graham and Dr G D’Orazi for critical reading of the manuscript; Dr C Gabay for providing pRa-1680-Luc vector; and Dr G Tolstonog for ENCODE in silico analyses on the sIL-1Ra promoter. This work was supported with grants from Associazione Italiana Ricerca sul Cancro (AICR) to GB (IG no. 8804), AG (MFAG no. 11752), PG (IG no. 13234), and AF (RC 3.5–2013). We thank the Proteomic Facility for Complex Potein Muxture (CPM) Analisys at ISS, Rome.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Ubertini, V., Norelli, G., D'Arcangelo, D. et al. Mutant p53 gains new function in promoting inflammatory signals by repression of the secreted interleukin-1 receptor antagonist. Oncogene 34, 2493–2504 (2015). https://doi.org/10.1038/onc.2014.191
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.191
This article is cited by
-
PPAR-γ alleviates the inflammatory response in TNF-α-induced fibroblast-like synoviocytes by binding to p53 in rheumatoid arthritis
Acta Pharmacologica Sinica (2023)
-
Mutant p53 in cancer: from molecular mechanism to therapeutic modulation
Cell Death & Disease (2022)
-
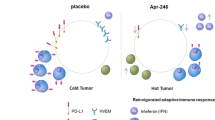
Mutant p53 gain of function mediates cancer immune escape that is counteracted by APR-246
British Journal of Cancer (2022)
-
Amino acid sensor GCN2 promotes SARS-CoV-2 receptor ACE2 expression in response to amino acid deprivation
Communications Biology (2022)
-
The complex role of tumor-infiltrating macrophages
Nature Immunology (2022)