Abstract
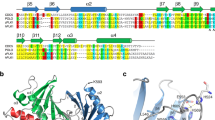
The Rho family GTPases, Cdc42, Rac and Rho, regulate signal transduction pathways via interactions with downstream effector proteins. We report here the solution structure of Cdc42 bound to the GTPase binding domain of αPAK, an effector of both Cdc42 and Rac. The structure is compared with those of Cdc42 bound to similar fragments of ACK and WASP, two effector proteins that bind only to Cdc42. The N-termini of all three effector fragments bind in an extended conformation to strand β2 of Cdc42, and contact helices α1 and α5. The remaining residues bind to switches I and II of Cdc42, but in a significantly different manner. The structure, together with mutagenesis data, suggests reasons for the specificity of these interactions and provides insight into the mechanism of PAK activation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jeanteur, P. Cytoskeleton and small G proteins (Springer, Berlin; 1999).
Burbelo, P.D., Drechsel, D. & Hall, A. J. Biol. Chem. 270, 29071–29074 (1995).
Thompson, G., Owen, D., Chalk, P.A. & Lowe, P.N. Biochemistry 37, 7885–7891 (1998).
Mott, H.R. et al. Nature 399, 384–399 (1999).
Abdul-Manan, N. et al. Nature 399, 379–383 (1999).
Cornilescu, G., Delaglio, F. & Bax, A. J. Biol. NMR 13, 289–302 (1999).
Guo, W., Sutcliffe, M.J., Cerione, R.S. & Oswald, R.E. Biochemistry 37, 14030–14037 (1998).
Leonard, D.A. et al. Biochemistry 36, 1173–1180 (1997).
Feltham, J.L. et al. Biochemistry 36, 8755–8766 (1997).
Hirshberg, M., Stockley, R.S., Dodson, G. & Webb, M.R. Nature Struct. Biol. 4, 147–152 (1997).
Owen, D, Mott, H.R., Laue, E.D. & Lowe, P.N. Biochemistry 39, 1243–1250 (2000).
Li, R. et al. J. Biol. Chem. 274, 29648–29654 (1999).
Manser, E. et al. Mol. Cell Biol. 17, 1129–1143 (1997).
Zhao, Z-S., Manser, E., Chen, X-Q, Chong, C., Leung, T. & Lim, L. Mol. Cell Biol. 18, 2153–2163 (1998).
Frost, J.A., Khokhlatchev, A., Stippec, S., White, M.A. & Cobb, M.H. J. Biol. Chem. 273, 28191–28198 (1998).
Kato, J., Kaziro, Y. & Satoh, T. Biochem. Biophys. Res. Commun. 268, 141–147 (2000).
Kim, A.S., Kakalis, L.T., Abdul-Manan, N., Liu, G.A. & Rosen, M.K. Nature 404, 151–158 (2000).
Huang, L., Hofer, F., Martin, G.S. & Kim, S-H. Nature Struct. Biol. 5, 422–426 (1998).
Nassar, N. et al. Nature 375, 554–560 (1995).
Ostermeier, C. & Brunger, A.T. Cell 96, 363–374 (1999).
Maesaki, R., Ihara, K., Shimizu, T., Kuroda, S., Kaibuchi, K. & Hakoshima, T. Mol. Cell 4, 793–803 (1999).
Vetter, I.R., Nowak, C., Nishimoto, T., Kuhlmann, J. & Wittinghofer, A. Nature 398, 39–46 (1999).
Edison, A.S., Abildgaard, F., Westler, W.M., Mooberry, E.S. & Markley, J.L. Methods Enzymol. 239, 3–79 (1994).
Wang, A.C. and Bax, A. J. Am. Chem. Soc. 118, 2483–2494 (1996).
Wang, A.C. and Bax, A. J. Am. Chem. Soc. 117, 1810–1813 (1995).
Zwahlen, C. et al. J. Am. Chem. Soc. 119, 6711–6721 (1997).
Brunger, A.T. et al. Acta Crystallogr. D 54, 905–921 (1998).
Nilges, M. & O'Donoghue, S.I. Prog. NMR Spec. 32, 107–139 (1998).
Nilges, M., Clore, G.M. & Gronenborn, A.M. FEBS Lett. 239, 129–136 (1988).
Barton, G.J. Protein Eng. 6, 37–40 (1993).
Kraulis, P.J. J. Appl. Crystallogr. 24, 946–950 (1991).
Merrit, E.A. and Murphy, M.E. Acta Crystallogr. D 50, 869–873 (1994).
Acknowledgements
We thank G. Thompson for the PAK(75–118) clone. A.M. is supported by a CASE award from the Medical Research Council and Glaxo-Wellcome. M.V. is supported by a grant from the Cambridge Commonwealth Trust (Cambridge Nehru Scholarship) and an Overseas Research Students (ORS) award. H.R.M. is an MRC Training Fellow. We acknowledge The European Commission for financial support. The Cambridge Centre for Molecular Recognition and the National 800 Facility are supported by the BBSRC and the Wellcome Trust.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morreale, A., Venkatesan, M., Mott, H. et al. Structure of Cdc42 bound to the GTPase binding domain of PAK. Nat Struct Mol Biol 7, 384–388 (2000). https://doi.org/10.1038/75158
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/75158
This article is cited by
-
Small molecules that allosterically inhibit p21-activated kinase activity by binding to the regulatory p21-binding domain
Experimental & Molecular Medicine (2016)
-
Juvenile myelomonocytic leukemia displays mutations in components of the RAS pathway and the PRC2 network
Nature Genetics (2015)
-
Mechanism of IRSp53 inhibition and combinatorial activation by Cdc42 and downstream effectors
Nature Structural & Molecular Biology (2014)
-
p21-Activated kinase 4 promotes prostate cancer progression through CREB
Oncogene (2013)
-
Inhibition of silibinin on migration and adhesion capacity of human highly metastatic breast cancer cell line, MDA-MB-231, by evaluation of β1-integrin and downstream molecules, Cdc42, Raf-1 and D4GDI
Medical Oncology (2012)