Abstract
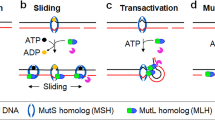
DNA mismatch repair corrects replication errors, thus reducing mutation rates and microsatellite instability. Genetic defects in this pathway cause Lynch syndrome and various cancers in humans. Binding of a mispaired or unpaired base by bacterial MutS and eukaryotic MutSα is well characterized. We report here crystal structures of human MutSβ in complex with DNA containing insertion-deletion loops (IDL) of two, three, four or six unpaired nucleotides. In contrast to eukaryotic MutSα and bacterial MutS, which bind the base of a mismatched nucleotide, MutSβ binds three phosphates in an IDL. DNA is severely bent at the IDL; unpaired bases are flipped out into the major groove and partially exposed to solvent. A normal downstream base pair can become unpaired; a single unpaired base can thereby be converted to an IDL of two nucleotides and recognized by MutSβ. The C-terminal dimerization domains form an integral part of the MutS structure and coordinate asymmetrical ATP hydrolysis by Msh2 and Msh3 with mismatch binding to signal for repair.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Iyer, R.R., Pluciennik, A., Burdett, V. & Modrich, P.L. DNA mismatch repair: functions and mechanisms. Chem. Rev. 106, 302–323 (2006).
Junop, M.S., Obmolova, G., Rausch, K., Hsieh, P. & Yang, W. Composite active site of an ABC ATPase: MutS uses ATP to verify mismatch recognition and authorize DNA repair. Mol. Cell 7, 1–12 (2001).
Habraken, Y., Sung, P., Prakash, L. & Prakash, S. ATP-dependent assembly of a ternary complex consisting of a DNA mismatch and the yeast MSH2–MSH6 and MLH1–PMS1 protein complexes. J. Biol. Chem. 273, 9837–9841 (1998).
Mendillo, M.L. et al. A conserved MutS homolog connector domain interface interacts with MutL homologs. Proc. Natl. Acad. Sci. USA 106, 22223–22228 (2009).
Habraken, Y., Sung, P., Prakash, L. & Prakash, S. Binding of insertion/deletion DNA mismatches by the heterodimer of yeast mismatch repair proteins MSH2 and MSH3. Curr. Biol. 6, 1185–1187 (1996).
Palombo, F. et al. hMutSbeta, a heterodimer of hMSH2 and hMSH3, binds to insertion/deletion loops in DNA. Curr. Biol. 6, 1181–1184 (1996).
Harfe, B.D. & Jinks-Robertson, S. Sequence composition and context effects on the generation and repair of frameshift intermediates in mononucleotide runs in Saccharomyces cerevisiae. Genetics 156, 571–578 (2000).
Surtees, J.A. & Alani, E. Mismatch repair factor MSH2–MSH3 binds and alters the conformation of branched DNA structures predicted to form during genetic recombination. J. Mol. Biol. 360, 523–536 (2006).
Marsischky, G.T., Filosi, N., Kane, M.F. & Kolodner, R. Redundancy of Saccharomyces cerevisiae MSH3 and MSH6 in MSH2-dependent mismatch repair. Genes Dev. 10, 407–420 (1996).
Herman, J.G. et al. Incidence and functional consequences of hMLH1 promoter hypermethylation in colorectal carcinoma. Proc. Natl. Acad. Sci. USA 95, 6870–6875 (1998).
de Wind, N. et al. HNPCC-like cancer predisposition in mice through simultaneous loss of Msh3 and Msh6 mismatch-repair protein functions. Nat. Genet. 23, 359–362 (1999).
Sia, E.A., Dominska, M., Stefanovic, L. & Petes, T.D. Isolation and characterization of point mutations in mismatch repair genes that destabilize microsatellites in yeast. Mol. Cell Biol. 21, 8157–8167 (2001).
Edelmann, W. et al. The DNA mismatch repair genes Msh3 and Msh6 cooperate in intestinal tumor suppression. Cancer Res. 60, 803–807 (2000).
Peltomäki, P. Lynch syndrome genes. Fam. Cancer 4, 227–232 (2005).
Haugen, A.C. et al. Genetic instability caused by loss of MutS homologue 3 in human colorectal cancer. Cancer Res. 68, 8465–8472 (2008).
Obmolova, G., Ban, C., Hsieh, P. & Yang, W. Crystal structures of mismatch repair protein MutS and its complex with a substrate DNA. Nature 407, 703–710 (2000).
Lamers, M.H. et al. The crystal structure of DNA mismatch repair protein MutS binding to a G x T mismatch. Nature 407, 711–717 (2000).
Warren, J.J. et al. Structure of the human MutSalpha DNA lesion recognition complex. Mol. Cell 26, 579–592 (2007).
Bowers, J., Sokolsky, T., Quach, T. & Alani, E. A mutation in the MSH6 subunit of the Saccharomyces cerevisiae MSH2–MSH6 complex disrupts mismatch recognition. J. Biol. Chem. 274, 16115–16125 (1999).
Drotschmann, K., Yang, W., Brownewell, F.E., Kool, E.T. & Kunkel, T.A. Asymmetric recognition of DNA local distortion. Structure-based functional studies of eukaryotic Msh2–Msh6. J. Biol. Chem. 276, 46225–46229 (2001).
Dowen, J.M., Putnam, C.D. & Kolodner, R.D. Functional studies and homology modeling of Msh2–Msh3 predict that mispair recognition involves DNA bending and strand separation. Mol. Cell Biol. 30, 3321–3328 (2010).
Lee, S.D., Surtees, J.A. & Alani, E. Saccharomyces cerevisiae MSH2–MSH3 and MSH2–MSH6 complexes display distinct requirements for DNA binding domain I in mismatch recognition. J. Mol. Biol. 366, 53–66 (2007).
Shell, S.S., Putnam, C.D. & Kolodner, R.D. Chimeric Saccharomyces cerevisiae Msh6 protein with an Msh3 mispair-binding domain combines properties of both proteins. Proc. Natl. Acad. Sci. USA 104, 10956–10961 (2007).
Kirkpatrick, D.T. & Petes, T.D. Repair of DNA loops involves DNA-mismatch and nucleotide-excision repair proteins. Nature 387, 929–931 (1997).
Sugawara, N., Paques, F., Colaiacovo, M. & Haber, J.E. Role of Saccharomyces cerevisiae Msh2 and Msh3 repair proteins in double-strand break-induced recombination. Proc. Natl. Acad. Sci. USA 94, 9214–9219 (1997).
Lyndaker, A.M. & Alani, E. A tale of tails: insights into the coordination of 3′ end processing during homologous recombination. Bioessays 31, 315–321 (2009).
van den Broek, W.J. et al. Somatic expansion behaviour of the (CTG)n repeat in myotonic dystrophy knock-in mice is differentially affected by Msh3 and Msh6 mismatch-repair proteins. Hum. Mol. Genet. 11, 191–198 (2002).
López Castel, A., Cleary, J.D. & Pearson, C.E. Repeat instability as the basis for human diseases and as a potential target for therapy. Nat. Rev. Mol. Cell Biol. 11, 165–170 (2010).
McMurray, C.T. Mechanisms of trinucleotide repeat instability during human development. Nat. Rev. Genet. 11, 786–799 (2010).
Tian, L. et al. Mismatch recognition protein MutSbeta does not hijack (CAG)n hairpin repair in vitro. J. Biol. Chem. 284, 20452–20456 (2009).
Hou, C., Chan, N.L., Gu, L. & Li, G.M. Incision-dependent and error-free repair of (CAG)(n)/(CTG)(n) hairpins in human cell extracts. Nat. Struct. Mol. Biol. 16, 869–875 (2009).
Panigrahi, G.B., Slean, M.M., Simard, J.P., Gileadi, O. & Pearson, C.E. Isolated short CTG/CAG DNA slip-outs are repaired efficiently by hMutSbeta, but clustered slip-outs are poorly repaired. Proc. Natl. Acad. Sci. USA 107, 12593–12598 (2010).
Wilson, T., Guerrette, S. & Fishel, R. Dissociation of mismatch recognition and ATPase activity by hMSH2–hMSH3. J. Biol. Chem. 274, 21659–21664 (1999).
Tian, L., Gu, L. & Li, G.M. Distinct nucleotide binding/hydrolysis properties and molar ratio of MutSalpha and MutSbeta determine their differential mismatch binding activities. J. Biol. Chem. 284, 11557–11562 (2009).
Owen, B.A.L., Lang, W.H. & McMurray, C.T. The nucleotide binding dynamics of human MSH2–MSH3 are lesion dependent. Nat. Struct. Mol. Biol. 16, 550–557 (2009).
Yang, W. Poor base stacking at DNA lesions may initiate recognition by many repair proteins. DNA Repair (Amst.) 5, 654–666 (2006).
Natrajan, G. et al. Structures of Escherichia coli DNA mismatch repair enzyme MutS in complex with different mismatches: a common recognition mode for diverse substrates. Nucleic Acids Res. 31, 4814–4821 (2003).
Mendillo, M.L., Putnam, C.D. & Kolodner, R.D. Escherichia coli MutS tetramerization domain structure reveals that stable dimers but not tetramers are essential for DNA mismatch repair in vivo. J. Biol. Chem. 282, 16345–16354 (2007).
Biswas, I. et al. Oligomerization of a MutS mismatch repair protein from Thermus aquaticus. J. Biol. Chem. 274, 23673–23678 (1999).
Hess, M.T., Mendillo, M.L., Mazur, D.J. & Kolodner, R.D. Biochemical basis for dominant mutations in the Saccharomyces cerevisiae MSH6 gene. Proc. Natl. Acad. Sci. USA 103, 558–563 (2006).
Alani, E., Sokolsky, T., Studamire, B., Miret, J.J. & Lahue, R.S. Genetic and biochemical analysis of Msh2p–Msh6p: role of ATP hydrolysis and Msh2p–Msh6p subunit interactions in mismatch base pair recognition. Mol. Cell Biol. 17, 2436–2447 (1997).
Calmann, M.A., Nowosielska, A. & Marinus, M.G. The MutS C terminus is essential for mismatch repair activity in vivo. J. Bacteriol. 187, 6577–6579 (2005).
Mazur, D.J., Mendillo, M.L. & Kolodner, R.D. Inhibition of Msh6 ATPase activity by mispaired DNA induces a Msh2(ATP)–Msh6(ATP) state capable of hydrolysis-independent movement along DNA. Mol. Cell 22, 39–49 (2006).
Drotschmann, K., Yang, W. & Kunkel, T.A. Evidence for sequential action of two ATPase active sites in yeast Msh2–Msh6. DNA Repair (Amst.) 1, 743–753 (2002).
Antony, E., Khubchandani, S., Chen, S. & Hingorani, M.M. Contribution of Msh2 and Msh6 subunits to the asymmetric ATPase and DNA mismatch binding activities of Saccharomyces cerevisiae Msh2–Msh6 mismatch repair protein. DNA Repair (Amst.) 5, 153–162 (2006).
Nakahara, M., Yokozaki, H., Yasui, W., Dohi, K. & Tahara, E. Identification of concurrent germ-line mutations in hMSH2 and/or hMLH1 in Japanese hereditary nonpolyposis colorectal cancer kindreds. Cancer Epidemiol. Biomarkers Prev. 6, 1057–1064 (1997).
Bjornson, K.P., Allen, D.J. & Modrich, P. Modulation of MutS ATP hydrolysis by DNA cofactors. Biochemistry 39, 3176–3183 (2000).
Gradia, S. et al. hMSH2–hMSH6 forms a hydrolysis-independent sliding clamp on mismatched DNA. Mol. Cell 3, 255–261 (1999).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Vagin, A. & Teplyakov, A. Molecular replacement with MOLREP. Acta Crystallogr. D Biol. Crystallogr. 66, 22–25 (2010).
Emsley, P., Lohkamp, B., Scott, W.G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Brünger, A.T. et al. Crystallography and NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Acknowledgements
We thank J. Jiang for collecting X-ray diffraction data of Loop4; A. Howard for help with data processing; T. Kunkel for the term 'isomerization'; R. Kolodner, E. Alani and J. Surtees for vital discussions; and D. Leahy for critical reading of the manuscript. The research of all authors was funded by the intramural research program of the National Institute of Diabetes and Digestive and Kidney Diseases, US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
S.G. conducted all experiments and collected X-ray data. W.Y. determined and refined the structures. S.G., M.G. and W.Y. contributed to the experimental design, data interpretation and manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 12577 kb)
Rights and permissions
About this article
Cite this article
Gupta, S., Gellert, M. & Yang, W. Mechanism of mismatch recognition revealed by human MutSβ bound to unpaired DNA loops. Nat Struct Mol Biol 19, 72–78 (2012). https://doi.org/10.1038/nsmb.2175
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2175
This article is cited by
-
Lynch syndrome, molecular mechanisms and variant classification
British Journal of Cancer (2023)
-
Development of a versatile nuclease prime editor with upgraded precision
Nature Communications (2023)
-
Tandem regulation of MutS activity by ATP and DNA during MMR initiation
Nature Structural & Molecular Biology (2022)
-
The selection process of licensing a DNA mismatch for repair
Nature Structural & Molecular Biology (2021)
-
Mispair-bound human MutS–MutL complex triggers DNA incisions and activates mismatch repair
Cell Research (2021)