Abstract
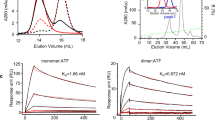
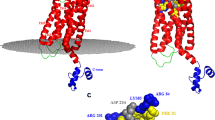
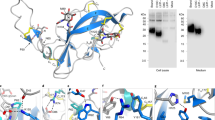
The urokinase receptor (uPAR) can recognize several ligands. The structural basis for this multiple ligand recognition by uPAR is unknown. This study reports the crystal structures of uPAR in complex with both urokinase (uPA) and vitronectin and reveal that uPA occupies the central cavity of the receptor, whereas vitronectin binds at the outer side of the receptor. These results provide a structural understanding of one receptor binding to two ligands.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Blasi, F. & Carmeliet, P. Nat. Rev. Mol. Cell Biol. 3, 932–943 (2002).
Llinas, P. et al. EMBO J. 24, 1655–1663 (2005).
Huai, Q. et al. Science 311, 656–659 (2006).
Wei, Y. et al. J. Biol. Chem. 269, 32380–32388 (1994).
Deng, G., Royle, G., Wang, S., Crain, K. & Loskutoff, D.J. J. Biol. Chem. 271, 12716–12723 (1996).
Gardsvoll, H. & Ploug, M. J. Biol. Chem. 282, 13561–13572 (2007).
Sidenius, N., Andolfo, A., Fesce, R. & Blasi, F. J. Biol. Chem. 277, 27982–27990 (2002).
Yuan, C. & Huang, M. Cell. Mol. Life Sci. 64, 1033–1037 (2007).
Madsen, C.D., Ferraris, G.M., Andolfo, A., Cunningham, O. & Sidenius, N. J. Cell Biol. 177, 927–939 (2007).
Zhou, A., Huntington, J.A., Pannu, N.S., Carrell, R.W. & Read, R.J. Nat. Struct. Biol. 10, 541–544 (2003).
Acknowledgements
This research is supported by grants from the US National Institutes of Health (HL08658) and the American Heart Association (0330089N) (to M.H.) and from the British Heart Foundation (to A.Z.). Data for this study were measured at beam lines ID19 and ID24 of Advanced Photon Sources and at beam line X29 of the the National Synchrotron Light Source.
Author information
Authors and Affiliations
Contributions
Q.H. and L.L. performed the experiments and solved the structures; M.H. refined the structures and wrote the paper; A.Z., A.P.M., G.C.P., J.C., D.E.S., B.F. and B.C.F. provided reagents and/or ideas to this work.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1, Supplementary Tables 1 and 2, and Supplementary Methods (PDF 1265 kb)
Rights and permissions
About this article
Cite this article
Huai, Q., Zhou, A., Lin, L. et al. Crystal structures of two human vitronectin, urokinase and urokinase receptor complexes. Nat Struct Mol Biol 15, 422–423 (2008). https://doi.org/10.1038/nsmb.1404
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1404
This article is cited by
-
Crystal structure and cellular functions of uPAR dimer
Nature Communications (2022)
-
Deficiency of Urokinase-Type Plasminogen Activator Receptor Is Associated with the Development of Perivascular Fibrosis in Mouse Heart
Bulletin of Experimental Biology and Medicine (2022)
-
Plasmin activity promotes amyloid deposition in a transgenic model of human transthyretin amyloidosis
Nature Communications (2021)
-
Molecular imaging of the urokinase plasminogen activator receptor: opportunities beyond cancer
EJNMMI Research (2020)
-
Therapeutics targeting the fibrinolytic system
Experimental & Molecular Medicine (2020)