Key Points
-
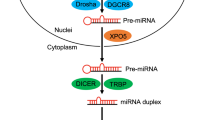
MicroRNAs (miRNAs) function as 21–24-nucleotide-long guides that regulate the expression of mRNAs containing complementary sequences.
-
Although plant miRNAs typically base-pair perfectly to target sites, animal miRNAs form imperfect duplexes with target sequences, complicating the ability to predict direct targets.
-
Numerous computational and experimental methods have been developed for studying miRNA target recognition and regulation.
-
In plants and animals, miRNAs usually repress target expression by inducing mRNA deadenylation and degradation or by inhibiting translation.
-
Many factors, including target site context, RNA-binding proteins and modifying enzymes, influence the ability of the miRNA complex to bind and regulate specific targets.
-
Base pairing between an miRNA and its target can influence the stability of the miRNA, resulting in increased miRNA levels in some cases and stimulated degradation in others.
-
Coding and non-coding RNAs can function as competing endogenous RNAs (ceRNAs) that bind miRNAs, sequestering them from binding and regulating other RNAs.
-
miRNA 'sponges' have been engineered to titrate specific miRNAs to study their functions in vivo.
-
The new understanding that miRNAs can both regulate and be regulated by target interactions raises many questions regarding the definition of a miRNA target and the functional outcome of miRNA targeting in vivo.
Abstract
MicroRNAs (miRNAs) have emerged as key gene regulators in diverse biological pathways. These small non-coding RNAs bind to target sequences in mRNAs, typically resulting in repressed gene expression. Several methods are now available for identifying miRNA target sites, but the mere presence of an miRNA-binding site is insufficient for predicting target regulation. Regulation of targets by miRNAs is subject to various levels of control, and recent developments have presented a new twist; targets can reciprocally control the level and function of miRNAs. This mutual regulation of miRNAs and target genes is challenging our understanding of the gene-regulatory role of miRNAs in vivo and has important implications for the use of these RNAs in therapeutic settings.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kozomara, A. & Griffiths-Jones, S. miRBase: integrating microRNA annotation and deep-sequencing data. Nucleic Acids Res. 39, D152–D157 (2011).
Bartel, D. P. MicroRNAs: target recognition and regulatory functions. Cell 136, 215–233 (2009).
Rigoutsos, I. New tricks for animal microRNAs: targeting of amino acid coding regions at conserved and nonconserved sites. Cancer Res. 69, 3245–3248 (2009).
Sayed, D. & Abdellatif, M. MicroRNAs in development and disease. Physiol. Rev. 91, 827–887 (2011).
Jackson, A. & Linsley, P. S. The therapeutic potential of microRNA modulation. Discov. Med. 9, 311–318 (2010).
Krol, J., Loedige, I. & Filipowicz, W. The widespread regulation of microRNA biogenesis, function and decay. Nature Rev. Genet. 11, 597–610 (2010).
Huntzinger, E. & Izaurralde, E. Gene silencing by microRNAs: contributions of translational repression and mRNA decay. Nature Rev. Genet. 12, 99–110 (2011).
Chen, X. Small RNAs and their roles in plant development. Ann. Rev. Cell Dev. Biol. 25, 21–44 (2009).
Yu, B. & Wang, H. Translational inhibition by microRNAs in plants. Prog. Mol. Subcell. Biol. 50, 41–57 (2010).
Addo-Quaye, C., Eshoo, T. W., Bartel, D. P. & Axtell, M. J. Endogenous siRNA and miRNA targets identified by sequencing of the Arabidopsis degradome. Curr. Biol. 18, 758–762 (2008).
German, M. A. et al. Global identification of microRNA-target RNA pairs by parallel analysis of RNA ends. Nature Biotech. 26, 941–946 (2008).
Llave, C., Xie, Z., Kasschau, K. D. & Carrington, J. C. Cleavage of scarecrow-like mRNA targets directed by a class of Arabidopsis miRNA. Science 297, 2053–2056 (2002).
Dai, X., Zhuang, Z. & Zhao, P. X. Computational analysis of miRNA targets in plants: current status and challenges. Brief. Bioinf. 12, 115–121 (2011).
Yekta, S., Shih, I. H. & Bartel, D. P. MicroRNA-directed cleavage of HOXB8 mRNA. Science 304, 594–596 (2004).
Doench, J. G. & Sharp, P. A. Specificity of microRNA target selection in translational repression. Genes Dev. 18, 504–511 (2004).
Reinhart, B. J. et al. The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature 403, 901–906 (2000).
Slack, F. J. et al. The lin-41 RBCC gene acts in the C. elegans heterochronic pathway between the let-7 regulatory RNA and the LIN-29 transcription factor. Mol. Cell 5, 659–669 (2000).
Vella, M. C., Choi, E. Y., Lin, S. Y., Reinert, K. & Slack, F. J. The C. elegans microRNA let-7 binds to imperfect let-7 complementary sites from the lin-41 3′UTR. Genes Dev. 18, 132–137 (2004).
Shin, C. et al. Expanding the microRNA targeting code: functional sites with centered pairing. Mol. Cell 38, 789–802 (2010).
Tay, Y., Zhang, J., Thomson, A. M., Lim, B. & Rigoutsos, I. MicroRNAs to Nanog, Oct4 and Sox2 coding regions modulate embryonic stem cell differentiation. Nature 455, 1124–1128 (2008).
Thomson, D. W., Bracken, C. P. & Goodall, G. J. Experimental strategies for microRNA target identification. Nucleic Acids Res. 39, 6845–6853 (2011).
Min, H. & Yoon, S. Got target? Computational methods for microRNA target prediction and their extension. Experimental Mol. Med. 42, 233–244 (2010).
Chi, S. W., Zang, J. B., Mele, A. & Darnell, R. B. Argonaute HITS-CLIP decodes microRNA–mRNA interaction maps. Nature 460, 479–486 (2009).
Hafner, M. et al. Transcriptome-wide identification of RNA-binding protein and microRNA target sites by PAR-CLIP. Cell 141, 129–141 (2010).
Leung, A. K. et al. Genome-wide identification of Ago2 binding sites from mouse embryonic stem cells with and without mature microRNAs. Nature Struct. Mol. Biol. 18, 237–244 (2011).
Zisoulis, D. G. et al. Comprehensive discovery of endogenous Argonaute binding sites in Caenorhabditis elegans. Nature Struct. Mol. Biol. 17, 173–179 (2010). References 23–26 report genome-wide analyses of Argonaute-binding sites in endogenous RNAs through CLIP experiments.
Baek, D. et al. The impact of microRNAs on protein output. Nature 455, 64–71 (2008).
Lim, L. P. et al. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 433, 769–773 (2005).
Selbach, M. et al. Widespread changes in protein synthesis induced by microRNAs. Nature 455, 58–63 (2008).
Behm-Ansmant, I. et al. mRNA degradation by miRNAs and GW182 requires both CCR4:NOT deadenylase and DCP1:DCP2 decapping complexes. Genes Dev. 20, 1885–1898 (2006).
Braun, J. E., Huntzinger, E., Fauser, M. & Izaurralde, E. GW182 proteins directly recruit cytoplasmic deadenylase complexes to miRNA targets. Mol. Cell 44, 120–133 (2011).
Chekulaeva, M. et al. miRNA repression involves GW182-mediated recruitment of CCR4-NOT through conserved W-containing motifs. Nature Struct. Mol. Biol. 18, 1218–1226 (2011).
Fabian, M. R. et al. miRNA-mediated deadenylation is orchestrated by GW182 through two conserved motifs that interact with CCR4-NOT. Nature Struct. Mol. Biol. 18, 1211–1217 (2011).
Giraldez, A. J. et al. Zebrafish miR-430 promotes deadenylation and clearance of maternal mRNAs. Science 312, 75–79 (2006).
Wu, L., Fan, J. & Belasco, J. G. MicroRNAs direct rapid deadenylation of mRNA. Proc. Natl Acad. Sci. USA 103, 4034–4039 (2006).
Humphreys, D. T., Westman, B. J., Martin, D. I. & Preiss, T. MicroRNAs control translation initiation by inhibiting eukaryotic initiation factor 4E/cap and poly(A) tail function. Proc. Natl Acad. Sci. USA 102, 16961–6 (2005).
Maroney, P. A., Yu, Y., Fisher, J. & Nilsen, T. W. Evidence that microRNAs are associated with translating messenger RNAs in human cells. Nature Struct. Mol. Biol. 13, 1102–1107 (2006).
Nottrott, S., Simard, M. J. & Richter, J. D. Human let-7a miRNA blocks protein production on actively translating polyribosomes. Nature Struct. Mol. Biol. 13, 1108–1114 (2006).
Olsen, P. H. & Ambros, V. The lin-4 regulatory RNA controls developmental timing in Caenorhabditis elegans by blocking LIN-14 protein synthesis after the initiation of translation. Dev. Biol. 216, 671–680 (1999).
Petersen, C. P., Bordeleau, M. E., Pelletier, J. & Sharp, P. A. Short RNAs repress translation after initiation in mammalian cells. Mol. Cell 21, 533–542 (2006).
Pillai, R. S. et al. Inhibition of translational initiation by Let-7 MicroRNA in human cells. Science 309, 1573–1576 (2005).
Seggerson, K., Tang, L. & Moss, E. G. Two genetic circuits repress the Caenorhabditis elegans heterochronic gene lin-28 after translation initiation. Dev. Biol. 243, 215–225 (2002).
Orom, U. A., Nielsen, F. C. & Lund, A. H. MicroRNA-10a binds the 5′UTR of ribosomal protein mRNAs and enhances their translation. Mol. Cell 30, 460–471 (2008).
Vasudevan, S., Tong, Y. & Steitz, J. A. Switching from repression to activation: microRNAs can up-regulate translation. Science 318, 1931–1934 (2007).
Mortensen, R. D., Serra, M., Steitz, J. A. & Vasudevan, S. Posttranscriptional activation of gene expression in Xenopus laevis oocytes by microRNA-protein complexes (microRNPs). Proc. Natl Acad. Sci. USA 108, 8281–8286 (2011).
Wu, E. et al. Pervasive and cooperative deadenylation of 3′UTRs by embryonic microRNA families. Mol. Cell 40, 558–570 (2010).
Hendrickson, D. G. et al. Concordant regulation of translation and mRNA abundance for hundreds of targets of a human microRNA. PLoS Biol. 7, e1000238 (2009).
Ingolia, N. T., Ghaemmaghami, S., Newman, J. R. & Weissman, J. S. Genome-wide analysis in vivo of translation with nucleotide resolution using ribosome profiling. Science 324, 218–223 (2009).
Guo, H., Ingolia, N. T., Weissman, J. S. & Bartel, D. P. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature 466, 835–840 (2010). References 48 and 49 describe the development and use of ribosome profiling experiments to study the mechanism of miRNA target regulation, leading to the conclusion that mRNA destabilization largely accounts for repression by the miRNA pathway.
Djuranovic, S., Nahvi, A. & Green, R. A parsimonious model for gene regulation by miRNAs. Science 331, 550–553 (2011).
Grimson, A. et al. MicroRNA targeting specificity in mammals: determinants beyond seed pairing. Mol. Cell 27, 91–105 (2007).
Gu, S., Jin, L., Zhang, F., Sarnow, P. & Kay, M. A. Biological basis for restriction of microRNA targets to the 3′ untranslated region in mammalian mRNAs. Nature Struct. Mol. Biol. 16, 144–150 (2009).
Fang, Z. & Rajewsky, N. The impact of miRNA target sites in coding sequences and in 3′UTRs. PLoS ONE 6, e18067 (2011).
Broderick, J. A., Salomon, W. E., Ryder, S. P., Aronin, N. & Zamore, P. D. Argonaute protein identity and pairing geometry determine cooperativity in mammalian RNA silencing. RNA 17, 1858–1869 (2011).
Saetrom, P. et al. Distance constraints between microRNA target sites dictate efficacy and cooperativity. Nucleic Acids Res. 35, 2333–2342 (2007).
Didiano, D. & Hobert, O. Molecular architecture of a miRNA-regulated 3′ UTR. RNA 14, 1297–1317 (2008).
Meisner, N. C. & Filipowicz, W. Properties of the regulatory RNA-binding protein HuR and its role in controlling miRNA repression. Adv. Exp. Med. Biol. 700, 106–123 (2010).
Bhattacharyya, S. N., Habermacher, R., Martine, U., Closs, E. I. & Filipowicz, W. Relief of microRNA-mediated translational repression in human cells subjected to stress. Cell 125, 1111–1124 (2006). This paper provides the first demonstration that an RNA-binding protein can reverse the repressive effect of a miRNA on a specific target.
Lebedeva, S. et al. Transcriptome-wide analysis of regulatory interactions of the RNA-binding protein HuR. Mol. Cell 43, 340–352 (2011).
Mukherjee, N. et al. Integrative regulatory mapping indicates that the RNA-binding protein HuR couples pre-mRNA processing and mRNA stability. Mol. Cell 43, 327–339 (2011).
Kim, H. H. et al. HuR recruits let-7/RISC to repress c-Myc expression. Genes Dev. 23, 1743–1748 (2009).
Kedde, M. et al. RNA-binding protein Dnd1 inhibits microRNA access to target mRNA. Cell 131, 1273–1286 (2007).
Mishima, Y. et al. Differential regulation of germline mRNAs in soma and germ cells by zebrafish miR-430. Curr. Biol. 16, 2135–2142 (2006).
Slack, F. J. & Ruvkun, G. A novel repeat domain that is often associated with RING finger and B-box motifs. Trends Biochem. Sci. 23, 474–475 (1998).
Meroni, G. & Diez-Roux, G. TRIM/RBCC, a novel class of 'single protein RING finger' E3 ubiquitin ligases. BioEssays 27, 1147–1157 (2005).
Rybak, A. et al. The let-7 target gene mouse lin-41 is a stem cell specific E3 ubiquitin ligase for the miRNA pathway protein Ago2. Nature Cell Biol. 11, 1411–1420 (2009).
Chatterjee, S. & Grosshans, H. Active turnover modulates mature microRNA activity in Caenorhabditis elegans. Nature 461, 546–549 (2009). This study is the first report of target interactions having a protective effect on miRNA stability.
Diederichs, S. & Haber, D. A. Dual role for argonautes in microRNA processing and posttranscriptional regulation of microRNA expression. Cell 131, 1097–1108 (2007).
Neumuller, R. A. et al. Mei-P26 regulates microRNAs and cell growth in the Drosophila ovarian stem cell lineage. Nature 454, 241–245 (2008).
Hammell, C. M., Lubin, I., Boag, P. R., Blackwell, T. K. & Ambros, V. nhl-2 modulates microRNA activity in Caenorhabditis elegans. Cell 136, 926–938 (2009).
Chu, C. Y. & Rana, T. M. Translation repression in human cells by microRNA-induced gene silencing requires RCK/p54. PLoS Biol. 4, e210 (2006).
Eulalio, A. et al. Target-specific requirements for enhancers of decapping in miRNA-mediated gene silencing. Genes Dev. 21, 2558–2570 (2007).
Schwamborn, J. C., Berezikov, E. & Knoblich, J. A. The TRIM–NHL protein TRIM32 activates microRNAs and prevents self-renewal in mouse neural progenitors. Cell 136, 913–925 (2009).
Chang, T. C. et al. Lin-28B transactivation is necessary for Myc-mediated let-7 repression and proliferation. Proc. Natl Acad. Sci. USA 106, 3384–9 (2009).
Qi, H. H. et al. Prolyl 4-hydroxylation regulates Argonaute 2 stability. Nature 455, 421–424 (2008).
Wu, C. et al. Hypoxia potentiates microRNA-mediated gene silencing through post-translational modification of Argonaute2. Mol. Cell. Biol. 31, 4760–4774 (2011).
Rudel, S. et al. Phosphorylation of human Argonaute proteins affects small RNA binding. Nucleic Acids Res. 39, 2330–2343 (2011).
Leung, A. K. et al. Poly(ADP-ribose) regulates stress responses and microRNA activity in the cytoplasm. Mol. Cell 42, 489–499 (2011).
Grishok, A. et al. Genes and mechanisms related to RNA interference regulate expression of the small temporal RNAs that control C. elegans developmental timing. Cell 106, 23–34 (2001).
Chatterjee, S., Fasler, M., Bussing, I. & Grosshans, H. Target-mediated protection of endogenous microRNAs in C. elegans. Dev. Cell 20, 388–396 (2011).
Ameres, S. L. et al. Target RNA-directed trimming and tailing of small silencing RNAs. Science 328, 1534–1539 (2010).
Wyman, S. K. et al. Post-transcriptional generation of miRNA variants by multiple nucleotidyl transferases contributes to miRNA transcriptome complexity. Genome Res. 21, 1450–1461 (2011).
Kai, Z. S. & Pasquinelli, A. E. MicroRNA assassins: factors that regulate the disappearance of miRNAs. Nature Struct. Mol. Biol. 17, 5–10 (2010).
Baccarini, A. et al. Kinetic analysis reveals the fate of a microRNA following target regulation in mammalian cells. Curr. Biol. 21, 369–376 (2011).
Cazalla, D., Yario, T. & Steitz, J. A. Down-regulation of a host microRNA by a Herpesvirus saimiri noncoding RNA. Science 328, 1563–1566 (2010). References 81, 84 and 85 demonstrated that some miRNA–target interactions result in destabilization of the miRNA.
Franco-Zorrilla, J. M. et al. Target mimicry provides a new mechanism for regulation of microRNA activity. Nature Genet. 39, 1033–1037 (2007). This paper provides the first example of an endogenous ncRNA acting as a sponge to limit the activity of a specific miRNA on other targets.
Ivashuta, S. et al. Regulation of gene expression in plants through miRNA inactivation. PLoS ONE 6, e21330 (2011).
Todesco, M., Rubio-Somoza, I., Paz-Ares, J. & Weigel, D. A collection of target mimics for comprehensive analysis of microRNA function in Arabidopsis thaliana. PLoS Genet. 6, e1001031 (2010).
Cesana, M. et al. A long noncoding RNA controls muscle differentiation by functioning as a competing endogenous RNA. Cell 147, 358–369 (2011).
Karreth, F. A. et al. In vivo identification of tumor- suppressive PTEN ceRNAs in an oncogenic BRAF-induced mouse model of melanoma. Cell 147, 382–395 (2011).
Poliseno, L. et al. A coding-independent function of gene and pseudogene mRNAs regulates tumour biology. Nature 465, 1033–1038 (2010). References 89 and 91 provide the first examples of pseudogenes and lncRNAs functioning as sponges to titrate miRNAs from protein-coding mRNAs with common miRNA target sites.
Salmena, L., Poliseno, L., Tay, Y., Kats, L. & Pandolfi, P. P. A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language? Cell 146, 353–358 (2011).
Seitz, H. Redefining microRNA targets. Curr. Biol. 19, 870–873 (2009). This paper develops the original hypothesis that the function of some miRNA target interactions is to sequester the miRNA, as opposed to repress expression of the target mRNA.
Sumazin, P. et al. An extensive microRNA-mediated network of RNA–RNA interactions regulates established oncogenic pathways in glioblastoma. Cell 147, 370–381 (2011).
Tay, Y. et al. Coding-independent regulation of the tumor suppressor PTEN by competing endogenous mRNAs. Cell 147, 344–357 (2011). References 90, 94 and 95 show that there is competition for miRNA activity among protein-coding mRNAs that contain target sites for a common miRNA, thus revealing an extensive network of ceRNAs that regulate miRNA effectiveness.
Vandewalle, C. et al. SIP1/ZEB2 induces EMT by repressing genes of different epithelial cell-cell junctions. Nucleic Acids Res. 33, 6566–6578 (2005).
Ponting, C. P., Oliver, P. L. & Reik, W. Evolution and functions of long noncoding RNAs. Cell 136, 629–641 (2009).
Ebert, M. S., Neilson, J. R. & Sharp, P. A. MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells. Nature Methods 4, 721–726 (2007).
Ebert, M. S. & Sharp, P. A. MicroRNA sponges: progress and possibilities. RNA 16, 2043–2050 (2010).
Rybak, A. et al. A feedback loop comprising lin-28 and let-7 controls pre-let-7 maturation during neural stem-cell commitment. Nature Cell Biol. 10, 987–993 (2008).
Mercer, T. R. et al. Expression of distinct RNAs from 3′ untranslated regions. Nucleic Acids Res. 39, 2393–2403 (2011).
Beitzinger, M., Peters, L., Zhu, J. Y., Kremmer, E. & Meister, G. Identification of human microRNA targets from isolated argonaute protein complexes. RNA Biol. 4, 76–84 (2007).
Easow, G., Teleman, A. A. & Cohen, S. M. Isolation of microRNA targets by miRNP immunopurification. RNA 13, 1198–1204 (2007).
Hendrickson, D. G., Hogan, D. J., Herschlag, D., Ferrell, J. E. & Brown, P. O. Systematic identification of mRNAs recruited to argonaute 2 by specific microRNAs and corresponding changes in transcript abundance. PLoS ONE 3, e2126 (2008).
Karginov, F. V. et al. A biochemical approach to identifying microRNA targets. Proc. Natl Acad. Sci. USA 104, 19291–19296 (2007).
Landthaler, M. et al. Molecular characterization of human Argonaute-containing ribonucleoprotein complexes and their bound target mRNAs. RNA 14, 2580–2596 (2008).
Zhang, L. et al. Systematic identification of C. elegans miRISC proteins, miRNAs, and mRNA targets by their interactions with GW182 proteins AIN-1 and AIN-2. Mol. Cell 28, 598–613 (2007).
Salles, F. J., Richards, W. G. & Strickland, S. Assaying the polyadenylation state of mRNAs. Methods 17, 38–45 (1999).
Jovanovic, M. et al. A quantitative targeted proteomics approach to validate predicted microRNA targets in C. elegans. Nature Methods 7, 837–842 (2010).
Wightman, B., Ha, I. & Ruvkun, G. Posttranscriptional regulation of the heterochronic gene lin-14 by lin-4 mediates temporal pattern formation in C. elegans. Cell 75, 855–862 (1993).
Jackson, R. J., Hellen, C. U. & Pestova, T. V. The mechanism of eukaryotic translation initiation and principles of its regulation. Nature Rev. Mol. Cell Biol. 11, 113–127 (2010).
Johnston, M. & Hutvagner, G. Post-translational modification of Argonautes and their role in small RNA mediated gene regulation. Silence 2, 5 (2011).
Pasquinelli, A. E. Molecular biology. Paring miRNAs through pairing. Science 328, 1494–1495 (2010).
Ebert, M. S. & Sharp, P. A. Emerging roles for natural microRNA sponges. Curr. Biol. 20, R858–R861 (2010).
Acknowledgements
Research in this laboratory is supported by the US National Institutes of Health (GM071654), Keck and Peter Gruber Foundations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Related links
Glossary
- miRNA-induced silencing complex
-
(miRISC). This is composed of the miRNA guide and the effector proteins, which minimally include Argonaute proteins. The complex recruits additional proteins to regulate the stability or translation of targeted mRNAs.
- Crosslinking immunoprecipitation
-
(CLIP). A method used to isolate and identify sequences that are bound by specific RNA-binding proteins. CLIP of microRNA complex proteins, such as Argonaute, has been used to detect microRNA-induced silencing complex (miRISC)-binding sites on genome-wide scales.
- GW182 proteins
-
Glycine–tryptophan repeat-containing proteins that interact with the microRNA-induced silencing complex (miRISC) to recruit proteins that mediate degradation or translational repression of target mRNAs. Also known as TNRC6A, TNRC6B and TNRC6C in vertebrates and AIN-1 and AIN-2 in Caenorhabditis elegans.
- Fragile X mental retardation syndrome-related protein 1
-
(FXR1). An RNA-binding protein that can regulate the translation or stability of bound transcripts.
- P bodies
-
Processing bodies, or P bodies, are cytoplasmic foci enriched for proteins that are involved in RNA degradation but that are devoid of translation factors. MicroRNAs, Argonaute and GW182 proteins also localize to P bodies, and silencing of some targets may occur in these structures.
- Competing endogenous RNAs
-
(ceRNAs). Bind miRNA complexes, titrating them from other target RNAs. Long non-coding RNAs, pseudogene RNAs and mRNAs can compete with other RNAs for access to specific miRNA complexes, effectively diluting their activity.
- Pseudogene
-
Relatives of protein-coding genes that can no longer be translated into functional gene products. In some cases, pseudogene RNAs are still transcribed but are then improperly processed or translated.
Rights and permissions
About this article
Cite this article
Pasquinelli, A. MicroRNAs and their targets: recognition, regulation and an emerging reciprocal relationship. Nat Rev Genet 13, 271–282 (2012). https://doi.org/10.1038/nrg3162
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg3162
This article is cited by
-
Serum MicroRNA profiles in chronic hepatitis C Egyptian patients before and after combined sofosbuvir and daclatasvir treatment
BMC Infectious Diseases (2024)
-
Effect and Mechanism of LIN28 on Ferroptosis in Mg2+-free Rat Hippocampal Neuron Model of Epilepsy
Neurochemical Research (2024)
-
Molecular functions of microRNAs in colorectal cancer: recent roles in proliferation, angiogenesis, apoptosis, and chemoresistance
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
Therapeutic Target MicroRNA Identification Based on Circular RNA Expression Signature After Intracerebral Hemorrhage
Molecular Neurobiology (2024)
-
Integrated analysis of microRNA and mRNA expression profiles in Preeclampsia
BMC Medical Genomics (2023)