Abstract
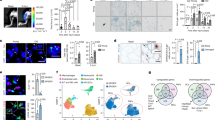
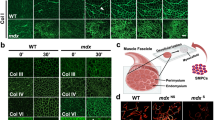
Age-related changes in the niche have long been postulated to impair the function of somatic stem cells. Here we demonstrate that the aged stem cell niche in skeletal muscle contains substantially reduced levels of fibronectin (FN), leading to detrimental consequences for the function and maintenance of muscle stem cells (MuSCs). Deletion of the gene encoding FN from young regenerating muscles replicates the aging phenotype and leads to a loss of MuSC numbers. By using an extracellular matrix (ECM) library screen and pathway profiling, we characterize FN as a preferred adhesion substrate for MuSCs and demonstrate that integrin-mediated signaling through focal adhesion kinase and the p38 mitogen-activated protein kinase pathway is strongly de-regulated in MuSCs from aged mice because of insufficient attachment to the niche. Reconstitution of FN levels in the aged niche remobilizes stem cells and restores youth-like muscle regeneration. Taken together, we identify the loss of stem cell adhesion to FN in the niche ECM as a previously unknown aging mechanism.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bentzinger, C.F., Wang, Y.X., Dumont, N.A. & Rudnicki, M.A. Cellular dynamics in the muscle satellite cell niche. EMBO Rep. 14, 1062–1072 (2013).
Gilbert, P.M. et al. Substrate elasticity regulates skeletal muscle stem cell self-renewal in culture. Science 329, 1078–1081 (2010).
Lv, H. et al. Mechanism of regulation of stem cell differentiation by matrix stiffness. Stem Cell Res. Ther. 6, 103 (2015).
Bonaldo, P. et al. Collagen VI deficiency induces early onset myopathy in the mouse: an animal model for Bethlem myopathy. Hum. Mol. Genet. 7, 2135–2140 (1998).
Bentzinger, C.F. et al. Fibronectin regulates Wnt7a signaling and satellite cell expansion. Cell Stem Cell 12, 75–87 (2013).
Urciuolo, A. et al. Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nat. Commun. 4, 1964 (2013).
Tierney, M.T. et al. Autonomous extracellular matrix remodeling controls a progressive adaptation in muscle stem cell regenerative capacity during development. Cell Rep. 14, 1940–1952 (2016).
Mann, C.J. et al. Aberrant repair and fibrosis development in skeletal muscle. Skelet. Muscle 1, 21 (2011).
D'Souza, D.M. et al. Diet-induced obesity impairs muscle satellite cell activation and muscle repair through alterations in hepatocyte growth factor signaling. Physiol. Rep. 3, e12506 (2015).
Sousa-Victor, P., García-Prat, L., Serrano, A.L., Perdiguero, E. & Muñoz-Cánoves, P. Muscle stem cell aging: regulation and rejuvenation. TEM 26, 287–296 (2015).
He, W.A. et al. NF-κB-mediated Pax7 dysregulation in the muscle microenvironment promotes cancer cachexia. J. Clin. Invest. 123, 4821–4835 (2013).
Fujimaki, S., Wakabayashi, T., Takemasa, T., Asashima, M. & Kuwabara, T. Diabetes and stem cell function. BioMed Res. Int. 2015, 592915 (2015).
Carlson, M.E. et al. Molecular aging and rejuvenation of human muscle stem cells. EMBO Mol. Med. 1, 381–391 (2009).
Watters, J.M., Clancey, S.M., Moulton, S.B., Briere, K.M. & Zhu, J.M. Impaired recovery of strength in older patients after major abdominal surgery. Ann. Surg. 218, 380–390 (1993).
Müller, M., Tohtz, S., Dewey, M., Springer, I. & Perka, C. Age-related appearance of muscle trauma in primary total hip arthroplasty and the benefit of a minimally invasive approach for patients older than 70 years. Int. Orthop. 35, 165–171 (2011).
Blau, H.M., Cosgrove, B.D. & Ho, A.T. The central role of muscle stem cells in regenerative failure with aging. Nat. Med. 21, 854–862 (2015).
Chakkalakal, J.V., Jones, K.M., Basson, M.A. & Brack, A.S. The aged niche disrupts muscle stem cell quiescence. Nature 490, 355–360 (2012).
Price, F.D. et al. Inhibition of JAK–STAT signaling stimulates adult satellite cell function. Nat. Med. 20, 1174–1181 (2014).
Tierney, M.T. et al. STAT3 signaling controls satellite cell expansion and skeletal muscle repair. Nat. Med. 20, 1182–1186 (2014).
Cosgrove, B.D. et al. Rejuvenation of the muscle stem cell population restores strength to injured aged muscles. Nat. Med. 20, 255–264 (2014).
Bernet, J.D. et al. p38 MAPK signaling underlies a cell-autonomous loss of stem cell self-renewal in skeletal muscle of aged mice. Nat. Med. 20, 265–271 (2014).
Sousa-Victor, P. et al. Geriatric muscle stem cells switch reversible quiescence into senescence. Nature 506, 316–321 (2014).
Gold, L. et al. Aptamer-based multiplexed proteomic technology for biomarker discovery. PLoS One 5, e15004 (2010).
Sakai, T. et al. Plasma fibronectin supports neuronal survival and reduces brain injury following transient focal cerebral ischemia but is not essential for skin-wound healing and hemostasis. Nat. Med. 7, 324–330 (2001).
Seibler, J. et al. Rapid generation of inducible mouse mutants. Nucleic Acids Res. 31, e12 (2003).
Siegel, A.L., Atchison, K., Fisher, K.E., Davis, G.E. & Cornelison, D.D. 3D time-lapse analysis of muscle satellite cell motility. Stem Cells 27, 2527–2538 (2009).
Brafman, D.A., Shah, K.D., Fellner, T., Chien, S. & Willert, K. Defining long-term maintenance conditions of human embryonic stem cells with arrayed cellular microenvironment technology. Stem Cells Dev. 18, 1141–1154 (2009).
Brafman, D.A. et al. Investigating the role of the extracellular environment in modulating hepatic stellate cell biology with arrayed combinatorial microenvironments. Integr. Biol. (Camb) 1, 513–524 (2009).
Moreno-Layseca, P. & Streuli, C.H. Signaling pathways linking integrins with cell cycle progression. Matrix Biol. 34, 144–153 (2014).
Sulzmaier, F.J., Jean, C. & Schlaepfer, D.D. FAK in cancer: mechanistic findings and clinical applications. Nat. Rev. Cancer 14, 598–610 (2014).
Raghavan, S., Bauer, C., Mundschau, G., Li, Q. & Fuchs, E. Conditional ablation of β1-integrin in skin. Severe defects in epidermal proliferation, basement membrane formation and hair follicle invagination. J. Cell Biol. 150, 1149–1160 (2000).
Lim, S.T. et al. Nuclear FAK promotes cell proliferation and survival through FERM-enhanced p53 degradation. Mol. Cell 29, 9–22 (2008).
Conboy, I.M. et al. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 433, 760–764 (2005).
Tigges, J. et al. The hallmarks of fibroblast aging. Mech. Ageing Dev. 138, 26–44 (2014).
Lanner, F. & Rossant, J. The role of FGF–Erk signaling in pluripotent cells. Development 137, 3351–3360 (2010).
Kim, S.H., Turnbull, J. & Guimond, S. Extracellular matrix and cell signaling: the dynamic cooperation of integrin, proteoglycan and growth factor receptor. J. Endocrinol. 209, 139–151 (2011).
Rozo, M., Li, L. & Fan, C.-M. Targeting β1-integrin signaling enhances regeneration in aged and dystrophic mice. Nat. Med. 10.1038/nm.4116 (2016).
García-Prat, L. et al. Autophagy maintains stemness by preventing senescence. Nature 529, 37–42 (2016).
Plath, T. et al. A novel function for the tumor suppressor p16INK4a: induction of anoikis via upregulation of the α5β1 fibronectin receptor. J. Cell Biol. 150, 1467–1478 (2000).
Lukjanenko, L., Brachat, S., Pierrel, E., Lach-Trifilieff, E. & Feige, J.N. Genomic profiling reveals that transient adipogenic activation is a hallmark of mouse models of skeletal muscle regeneration. PLoS One 8, e71084 (2013).
Srinivas, S. et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 1, 4 (2001).
Lepper, C., Conway, S.J. & Fan, C.M. Adult satellite cells and embryonic muscle progenitors have distinct genetic requirements. Nature 460, 627–631 (2009).
Schneider, C.A., Rasband, W.S. & Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Smyth, G.K. Linear models and empirical Bayes methods for assessing differential expression in microarray experiments. Stat. Appl. Genet. Mol. Biol. 3, e3 (2004).
Huang, W., Sherman, B.T. & Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4, 44–57 (2009).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Acknowledgements
We thank J. Ritchie for help with mouse husbandry. We are grateful to the NIHS community for fruitful discussions and support, in particular to the Aging and Stem Cells groups, and to E. Rolland and E. Baetge. We thank C. Poser for excellent technical assistance. ROSA26–CreERT2 mice were provided by F. Stewart (Technische Universität Dresden, Germany) as a kind gift to M.A.R. J.V.M. and M.J.J. were supported by a grant from the German Research Foundation (DFG) (grant MA–3975/2–1). M.A.R. holds the Canada Research Chair in Molecular Genetics and is supported by a grant from the Government of the Ontario Ministry of Research and Innovation (MRI) (grant ORF–RE05–084). M.R. and C.M.F. are supported by the National Institutes of Health grants (grants HD075345 and AR060042). C.F.B. is supported by the Fondation Suisse de Recherche sur les Maladies Musculaires (FSRMM).
Author information
Authors and Affiliations
Contributions
M.J.J., N.H., C.P.-C., M.R., S.K., M.S., L. Li, S.M., U.L., F.S., N.A.D., A.P., C.-M.F. and J.v.M., designed and conducted experiments, and analyzed results; E.M. and F.R. performed data analysis; G.J. provided support with high-content image acquisition and analysis; M.A.R. provided critical reagents, edited the manuscript and helped with data interpretation; R.F. provided critical reagents; D.H.W. helped with data acquisition and analysis; P.S. and P.D. helped with experimental design, data interpretation and editing of the manuscript; and L. Lukjanenko, J.N.F. and C.F.B. designed and conducted experiments, analyzed results and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
L. Lukjanenko, N.H., C.P.-C., E.M., S.K., G.J., S.M., F.R., U.L., F.S., A.P., P.S., P.D., J.N.F. and C.F.B. are employees of the Nestlé Institute of Health Sciences S.A., Switzerland.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1–3 (PDF 4843 kb)
Rights and permissions
About this article
Cite this article
Lukjanenko, L., Jung, M., Hegde, N. et al. Loss of fibronectin from the aged stem cell niche affects the regenerative capacity of skeletal muscle in mice. Nat Med 22, 897–905 (2016). https://doi.org/10.1038/nm.4126
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4126
This article is cited by
-
Melatonin promotes the development of the secondary hair follicles by regulating circMPP5
Journal of Animal Science and Biotechnology (2023)
-
Extracellular matrix: the critical contributor to skeletal muscle regeneration—a comprehensive review
Inflammation and Regeneration (2023)
-
Reconstructing the ageing extracellular matrix
Nature Reviews Bioengineering (2023)
-
Metabolomic signatures for the longitudinal reduction of muscle strength over 10 years
Skeletal Muscle (2022)
-
The jam session between muscle stem cells and the extracellular matrix in the tissue microenvironment
npj Regenerative Medicine (2022)